Manuscript accepted on :26-09-2025
Published online on: 30-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Kasthuri NMK and Dr. Maha Jalal
Second Review by: Dr. Inamullah Khan and Dr. Fatimah Haitham Fathi
Final Approval by: Dr. Anton R Kiselev
Ni Wayan Bogoriani1* , I Gusti Ayu Putu Eka Pratiwi2
, I Gusti Ayu Putu Eka Pratiwi2 , Ni Wayan Mirawati3
, Ni Wayan Mirawati3 , Ida Ayu Raka Astiti Asih4
, Ida Ayu Raka Astiti Asih4 and Komang Tria Noviana Dewi5
and Komang Tria Noviana Dewi5
1Department of Chemistry, University of Udayana, Bali, Indonesia.
2Department. of Doctor Education, University of Udayana, Bali, Indonesia.
3Department of Chemistry, University of Udayana, Bali, Indonesia.
4Department. of Chemistry, University of Udayana, Bali, Indonesia.
5Department of Pharmacy, University of Udayana, Bali, Indonesia.
Corresponding Author E-mail:bogi_wayan@yahoo.com
DOI : https://dx.doi.org/10.13005/bpj/3250
Abstract
Obesity represents a metabolic disorder associated with complications including hyperglycemia, dyslipidemia, and insulin resistance. The present study sought to characterize the bioactive compounds in red ginger extract through Liquid Chromatography–Mass Spectrometry (LC-MS) and to assess its therapeutic potential on body weight, glucose, triglyceride (TG), and free fatty acid (FFA) levels in Wistar rats with induced obesity. Female Wistar rats were induced to develop obesity with a high-fat intake until they reached a Lee index > 0.3. The experimental animals were allocated into four groups: a negative control, a positive control (obese untreated), and two treatment groups that received oral red ginger extract at doses of 100 mg/kg BW and 200 mg/kg BW for a duration of four weeks. The results demonstrated that both doses of red ginger extract significantly (p < 0.05) reduced weight, glucose, profile lipid (TG, and FFA) levels compared with the obese group. Specifically, glucose levels were reduced by 58.65% and 62.86% at 100 mg/kg BW and 200 mg/kg BW, respectively. Triglyceride levels decreased by 62.39% and 65.46%, while free fatty acid levels were reduced by 24.08% and 43.23%, respectively. LC-MS analysis identified 11 bioactive compounds in red ginger extract, including gallic acid, ar-curcumin, α-zingiberene, naringenin, 6-shogaol, kaempferol, catechin, 6-gingerol, quercetin, 8-gingerol, and rutin. These findings indicate that the bioactive compounds in red ginger extract collectively provide a synergistic effect as a phytotherapeutic agent in the treatment of obesity and its metabolic complications.
Keywords
Blood Glucose; Free Fatty Acids; LC-MS; Red Ginger; Triglycerides
Download this article as:| Copy the following to cite this article: Bogoriani N. W, Pratiwi I. G. A. P. E, Mirawati N. Y, Asih I. A. R. A, Dewi K. T. N. Therapeutic Potential of Red Ginger (Zingiber officinale var. Rubrum) Bioactive Compounds in Obesity-Associated Metabolic Parameters of Wistar Rats. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Bogoriani N. W, Pratiwi I. G. A. P. E, Mirawati N. Y, Asih I. A. R. A, Dewi K. T. N. Therapeutic Potential of Red Ginger (Zingiber officinale var. Rubrum) Bioactive Compounds in Obesity-Associated Metabolic Parameters of Wistar Rats. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3L9E99t |
Introduction
Obesity is an escalating global health concern that is strongly linked to insulin resistance and metabolic disturbances, including hyperglycemia, hypertriglyceridemia, and increased free fatty acid levels. All of these factors increase the risk of metabolic syndrome and cardiometabolic diseases. Mechanistic studies and reviews suggest that ginger products and bioactive compounds can modulate immunometabolic pathways associated with adipogenesis, adipose tissue inflammation, and glucose homeostasis.1
Red ginger is a commonly consumed ginger variety in Indonesia and is reported to have a relatively high phenolic and vanilloid content (including gingerols and shogaols) compared to other varieties, potentially exhibiting potent antioxidant, anti-inflammatory, and metabolic activities. This makes red ginger an attractive candidate as a natural agent for addressing metabolic disorders associated with obesity. 2-3
Several main components of ginger (particularly 6-gingerol and 6-shogaol) have been studied in vivo and shown to reduce fat accumulation, body weight, serum triglyceride levels, and improve insulin/glucose tolerance in animal models of obesity. Reported mechanisms include inhibition of adipogenesis, regulation of lipid-metabolic gene expression, modulation of pro-inflammatory adipokine cytokines, activation of the Adenosine Monophosphate Activated Protein Kinase (AMPK) pathway, and changes in gut microbiota composition that contribute to metabolic improvements.4
Modern phytochemical analysis using techniques such as Liquid Chromatography–Mass Spectrometry allows the identification of bioactive compounds in red ginger extract, including gallic acid, naringenin, kaempferol, catechin, quercetin, rutin, and various gingerolsand shogaols, which collectively may exert synergistic effects on metabolic parameters (glucose, triglycerides, free fatty acids) through antioxidant, anti-inflammatory, and enzyme/lipid metabolism regulation mechanisms. Therefore, studies combining LC-MS profiles and metabolic parameter measurements in animal models of obesity are relevant to link the chemical components of the extract with the observed therapeutic effects.2
Accordingly, this study was designed on the premise that the bioactive constituents of red ginger extr/act (such as 6-gingerol, 6-shogaol, flavonoids, and phenolics) may contribute to weight reduction and improvements in glucose and lipid parameters (triglycerides and free fatty acids) in obesity-induced rats, with LC-MS employed to link the identified compounds to their observable pharmacological activities.2
Therefore, the present study seeks to assess the potential of bioactive compounds in red ginger extract to lower these parameters and explore its role as a natural therapeutic option for managing obesity and associated metabolic disorders.
Materials and Methods
Materials
Red ginger rhizome samples collected from the Batubulan area, Gianyar, Bali, distilled water, 96% methanol, twenty-four Wistar rats, high-fat intake (60% standard diet, 20% duck egg yolk, 20% lard), standard feed (CP550), Meyer’s reagent, NaOH, oxalic acid solids, magnesium powder, sulfuric acid, anhydrous acetic acid, FeCl3, concentrated HCl, dilute HCl, chloroform, PP indicator, Folin-Ciocelteau, distilled water.
Equipment
Erlenmeyer flasks, volumetric pipettes, evaporating cups, beakers, LC-MS (Shimadzu 8040), blender, rotary vacuum evaporator (Buchi type R 215), micropipettes, analytical balance, glucometer and test strip set, burette, clamps and stand, mouse cages, mouse drinking bottles, probes, and chemistry analyzer (ERBA-XL 100).
Methods
Sample Preparation
Red ginger rhizomes were prepared by cleaning the samples in running water and then draining them. The samples were cut into smaller pieces and air-dried. Next, the samples were blended until smooth and weighed on an analytical balance, then the water content was calculated.
Red Ginger Extraction
Red ginger powder (500 g) was macerated in 96% technical methanol at a ratio of 1:5 for 24 hours. The maceration was repeated three times for 24 hours. The extract was then filtered through filter paper to separate the filtrate from the pulp. A concentrated extract of red ginger rhizome was prepared by evaporating the solvent with a rotary vacuum evaporator.3,10
Phytochemical Testing
The flavonoid test on red ginger rhizome extract was performed by adding concentrated HCl and Mg powder to the extract. The flavonoid test was considered positive if the solution changed color to orange.
The saponin test was performed by adding methanol extract of red ginger rhizome in a test tube with distilled water in a 1:1 ratio, then shaking the mixture vigorously for one minute until foam formed. A positive result for saponins was indicated by the presence of foam that persisted for several minutes.
The alkaloid test on ginger rhizome extract was performed by adding Mayer’s reagent or dipotassium tetraiodomercurate to 1 ml of red ginger rhizome extract. The presence of alkaloids was confirmed by the appearance of a white precipitate.
The steroid and triterpenoid tests were performed by reacting the extract with Liebermann-Bouchard reagent, anhydrous acetic acid and concentrated sulfuric acid (H2SO4). The steroid test was considered positive if the extract changed color to blue-green. The triterpenoid test was considered positive if the extract changed color to red-purple.
The phenol test can be performed by adding Folin-Ciocalteau reagent to red ginger rhizome extract and then adding Na2CO3 powder to create an alkaline environment. This test is considered positive if the extract turns blue.
The tannin test is performed by reacting 1% FeCl3 with the sample. A positive tannin test is identified by observing the color change of the extract to blackish green.
LC-MS Analysis of Red Ginger
The concentrated extract of red ginger rhizome was purified using Solid Phase Extraction (SPE). First, a Sep-Pak C18 column (1 cc, 100 mg) was conditioned with 1 ml of an 80:20 acetonitrile-water mixture, followed by 0.5 ml of protein precipitation solution. Next, the column was eluted with 0.5 ml of an acetonitrile-water mixture, followed by 0.25 ml of a 200 mM ammonium formate solution in a 50:50 acetonitrile-methanol mixture. The final eluate was then mixed with 0.2 ml of an acetonitrile buffer solution (25:75) containing 25 mM ammonium formate at pH 4.5. The resulting mixture was filtered through a 0.45 µm cellulose acetate filter membrane, degassed, and injected into the LC-MS instrument. The obtained data were processed using LC-MS, and the chromatograms were analyzed using MassLynx software version 4.
Animal Treatment
With approval from the Animal Ethics Committee (No. B/203/UN14.2.9/PT.01.04/2024), twenty-four female Wistar rats were allocated into four groups and subjected to the following treatments:
K1: Negative (Normal) control group
K2: Positive (Obese) control group
K3: Obese + red ginger extract at 100 mg/kg bw
K4: Obese + red ginger extract at 200 mg/kg bw
Eighteen female rats were given a high-fat diet, and six rats were fed a standard diet. Red ginger rhizome extract was administered after the rats were declared obese, as measured by a Lee index value > 0.3. Body weight was measured weekly for four weeks. After four weeks, the rats were fasted for 10 hours to collect blood samples. Blood samples were obtained from the orbital cavity of rats using a capillary tube and transferred into a 3 mL syringe, then left to clot for 30 minutes at room temperature. Serum was isolated by centrifugation at 1000 rpm for 10 minutes, after which it was collected and stored in microcentrifuge tubes. Determining Glucose Levels
Blood glucose levels are measured using a glucometer. A blood sample is drawn, touched to the test strip on the glucometer, and then left to stand until the blood glucose reading is recorded. Normal fasting blood glucose levels in mice are 50-125 mg/dl; if the reading is greater than 125 mg/dl, the mouse is considered diabetic.11
Determining Triglyceride Levels
Serum triglyceride levels are measured using a chemistry analyzer, the ERBA XL-100 instrument. The reagents used to determine triglyceride levels are Sekisui Medical Triglyceride Kit reagents Lot No. R1: 105R10 and R2: 105R10.
Triglyceride levels are determined using a control run, placing 260 µl of Reagent R1 and 130 µL of Reagent R2 in the instrument according to the order indicated on the reagent tray. A 3.0 µL control and sample were placed in the instrument and measured at a wavelength of 600-700 nm. Afterward, a sample run was performed by inserting 3.0 µl of sample into the instrument. The instrument was then run, with the sample absorbance measured at a wavelength of 546 nm.11
Analysis of Free Fatty Acid Content
Free fatty acid levels were measured by alkalimetric titration after extraction using the Folch method. Extraction was carried out by dissolving 200 µl (0.2 ml) of blood serum in 10 ml of a neutral solution of methanol and chloroform (1:2) v/v. Water was then added to the mixture to achieve phase separation. The chloroform phase containing the lipids was separated from the other phase.
Two to three drops of phenolphthalein indicator were added to the sample, and the blood serum was titrated with 0.01 N NaOH solution. The titration was stopped when the solution changed color to a faint pink. The volume of NaOH was recorded, and the free fatty acid content was calculated in mmol/L.11
Data Analysis Using Statistics
Statistical analysis of the research data was performed using ANOVA in SPSS version 23.0 with a 95% confidence level. The procedure included several stages: data normality was first assessed using the Shapiro–Wilk test (suitable for sample sizes <30), with p > 0.05 indicating normal distribution. Homogeneity of variance was then evaluated using Levene’s test, where p > 0.05 signified uniform variance across groups. For datasets meeting both assumptions, a One-Way ANOVA was applied, followed by an LSD post hoc test to identify intergroup differences. When the assumptions were not met, Tamhane’s post hoc test was employed instead.
Results
Phytochemical Testing of Extract
Phytochemical screening outcomes of the concentrated red ginger rhizome extract are displayed in Table 1.
Table 1: Outcomes of phytochemical test on red ginger rhizome extract
| Metabolite | Parameter | Test Result |
| Flavonoids | Orange color | + |
| Saponins | Constant foam | + |
| Steroids | Dark green color | + |
| Triterpenoids | Brown ring | + |
| Alkaloids | Yellow precipitate | + |
| Phenolics | Blue color | + |
| Tannins | Blackish green color | + |
Description: + = present – = absent
LC-MS Analysis of Red Ginger Rhizome Extract
Compound content analysis of red ginger rhizome extract using LC-MS was performed by analyzing its mass spectrum and comparing it to the mass spectrum in the Chemspider database. Table 2 lists the eleven compounds identified in the crude extract of red ginger rhizome, namely gallic acid, α-curcumin, α-zingiberene, naringenin, 6-shogaol, kaempferol, catechin, 6-gingerol, quercetin, 8-gingerol, and rutin.
 |
Table 2: Compounds contained in red ginger rhizome extract |
Animal Treatment
Twenty-four female Wistar rats were fattened using a high-fat intake, which involved feeding them high-fat feed until their body weight reached obesity (Lee index > 0.3). Red ginger rhizome extract administration was associated not only with changes in glucose, triglyceride, and free fatty acid levels but also with a progressive decrease in body weight across the weeks. Changes in the rats’ body weight over the course of four weeks are displayed in Figure 1.
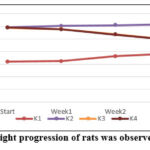 |
Figure 1: The body weight progression of rats was observed over a 4-week period |
Figure 1 and Table 3 show that four weeks of treatment with red ginger rhizome extract at doses of 100 and 200 mg/kg BW reduced the body weight of obese Wistar rats. Both doses produced comparable effects in lowering body weight, and notably, the extract’s anti-obesity activity helped restore the rats’ body weight toward the normal range.
Table 3: The average weight changes in Wistar rats were monitored for 4 weeks
| Group | K1 | K2 | K3 | K4 |
| Start (g) | 160.23 ± 1.03b,c,d | 295.45 ± 1.58a | 294.73 ± 3.31a | 295.98 ± 1.90a |
| Week -1(g) | 161.90 ± 1.39b,c,d | 301.67 ± 1.11a,c,d | 288.57 ± 1.64a,b | 287.89 ±1.40a,b |
| Week-2(g) | 181.26 ± 2.36b,c,d | 305.08 ± 1.03a,c,d | 268.18 ± 1.59a,b | 266.96 ± 1.79a,b |
| Week -3 (g) | 192.24 ± 1.94 b,c,d | 308.92 ± 1.06 a,c,d | 247.41 ± 1.48a,b | 246.36 ± 1.30a,b |
| Week-4 (g) | 213.15 ± 1.74 b,c,d | 312.35 ± 1.00 a,c,d | 218.47 ± 1.58a,b | 217.58 ± 1.33a,b |
Description: Mean ± SD (n=6) is shown, with differing superscript letters in a row reflecting significant differences (p<0.05). a = negative/normal control (K1), b = positive/obese control (K2), c = red ginger extract 100 mg/kgbw (K3), d = red ginger extract 200 mg/kgbw (K4)
Data Analysis from In vivo Test Results
At the end of the 4-week treatment period with red ginger extract, blood glucose, triglyceride, and free fatty acid levels were evaluated The data on blood glucose, triglyceride, and free fatty acid levels are illustrated in Figure 2 and Table 4.
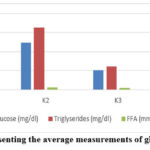 |
Figure 2: Graph presenting the average measurements of glucose, TG, and FFA. |
Table 4: Mean concentrations of glucose, TG, and FFA
| Group | K1 | K2 | K3 | K4 |
| Glucose (mg/dl) | 81.50 ± 1.05b,c,d | 246.17 ± 1.17a,c,d | 101,50 ± 1.05a,b,d | 91.00 ± 0.89a,b,c |
| Triglycerides (mg/dl) | 60.00 ± 1.79b,c,d | 325.67 ± 1.63a,c,d | 122.50 ± 1.05a,b,d | 112.50 ± 1.05a,b,c |
| Free fatty acids (mmol/L) | 8.53 ± 0.46b,d | 11.96 ± 0.99a,c,d | 9.08 ± 0.69b,d | 6.79 ± 0.52a,b,c |
Description: Mean ± SD (n=6) is shown, with differing superscript letters in a row reflecting significant differences (p<0.05). a = negative/normal control (K1), b = positive/obese control (K2), c = red ginger extract 100 mg/kgbw (K3), d = red ginger extract 200 mg/kgbw (K4).
Discussion
Phytochemical Testing
The phytochemical profile of red ginger rhizome extract (Table 1) demonstrated the presence of several secondary metabolites, namely flavonoids, saponins, steroids, triterpenoids, alkaloids, phenolics, and tannins. Research by Sururi et al. (2022) reported that red ginger contains flavonoids, saponins, alkaloids, and triterpenoids, which have antioxidant activity.3
LC-MS Analysis
As shown in Table 2, eleven compounds were detected in the crude extract of red ginger rhizome : gallic acid, α-curcumin, α-zingiberene, naringenin, 6-shogaol, kaempferol, catechin, 6-gingerol, quercetin, 8-gingerol, and rutin, which together may exert a synergistic effect against obesity and metabolic disorders.
Gallic acid has been reported to protect pancreatic cells by improving β-cell function by inhibiting caspase-9-related cellular apoptosis.12 Gallic acid can induce nuclear transcription, leading to differentiation and insulin sensitivity in adipocytes.13 Kaempferol and 6-shogaol have also been found to protect pancreatic β-cells by preventing apoptosis in pancreatic β-cells and activating autophagy, which helps β-cell survival.14 Rutin also plays a role in reducing carbohydrate absorption, facilitating glucose uptake in tissues, and inducing insulin secretion by β-cells.15
Furthermore, curcumin can act as an antidiabetic by reducing oxidative stress in diabetic patients, thereby reducing diabetes complications. Adab et al. (2019) reported that curcumin was effective in lowering excess body weight, triglyceride levels, and blood glucose.16
Another mechanism that influences these three parameters is by inhibiting enzymes in the body’s metabolism. The reduction in blood glucose levels is mediated through the inhibition of α-glucosidase and α-amylase enzymes. These enzymes function as key regulators in the pathways of carbohydrate metabolism and glucose production. The inhibition of these enzymes allows for controlled glucose production and protection against hyperglycemic states. Compounds that have this mechanism include catechin, quercetin, naringenin, and rutin.17 The presence of free hydroxyl groups in catechin is closely associated with its effectiveness in inhibiting α-glucosidase.18Catechins further act to improve insulin resistance, suppress oxidative stress, regulate mitochondrial performance, and reduce glucose levels in the blood. Furthermore, quercetin can activate the AMPK pathway and increase GLUT4 translocation, which plays a role in increasing glucose uptake in skeletal muscle cells. This increased glucose uptake in muscle cells can prevent insulin resistance.19 Naringenin contributes to better glucose regulation through its effect on liver enzymes that govern gluconeogenesis and glycolysis.20
Unlike other compounds, 6-gingerol can activate the GLP-1-mediated insulin secretion pathway and regulate insulin granule exocytosis, thereby enhancing glucose-stimulated insulin secretion.21 Furthermore, this compound can also increase glycogen synthase 1 activity, thereby increasing glycogen storage in skeletal muscle. 8-gingerol, a structurally similar compound, has also been suggested to lower glucose, triglyceride, and free fatty acid levels because of its antioxidant capacity. Antioxidants play a crucial role in reducing the risk of diabetes complications due to their ability to combat oxidative stress. According to Lian et al. (2024), 8-gingerol significantly reduced the elevation of reactive oxygen species (ROS).22
Animal Treatment
According to Table 3 and Figure 1, administration of red ginger rhizome extract at 100 mg/kg bw (K3) and 200 mg/kg bw (K4) for 4 weeks significantly reduced the body weight of obese Wistar rats relative to the obese control group (p < 0.05). This outcome is attributed to the combined action of secondary metabolites with anti-obesity activity. Supporting evidence is provided by Bogoriani et al. (2025), who demonstrated that obese mice receiving lidah mertua leaf extract containing bioactive compounds also underwent significant weekly weight loss at the same dosage.24
Table 3 and Figure 1 further demonstrate that red ginger rhizome extract at 100 and 200 mg/kg bw produced no significant difference, indicating that both doses were nearly equally effective in reducing the body weight of Wistar rats. This weight loss can occur due to decreased levels of triglycerides or fat in the body. Rampengan et al. (2024) reported that red ginger has anti-obesity properties containing rutin and curcumin compounds, which were obtained with the same compound content as this study.23 The difference in compound content found in red ginger rhizome extract is caused by differences in the growing location of the red ginger plant and the solvent used in the extraction.
In Vivo Data Analysis
Table 4 and Figure 2 demonstrate that oral administration of red ginger extract for four weeks at doses of 100 mg/kg (K3) and 200 mg/kg bw (K4) significantly (p < 0.05) in obese rats group. The positive control group exhibited marked elevations in glucose (246.17 mg/dL), triglycerides (325.67 mg/dL), and free fatty acids (11.96 mmol/L), confirming obesity-related metabolic disturbances. In contrast, treatment with red ginger extract lowered glucose to 101.50 mg/dL (K3) and 91.00 mg/dL (K4), triglycerides to 122.50 mg/dL (K3) and 112.50 mg/dL (K4), and free fatty acids to 9.08 mmol/L (K3) and 6.79 mmol/L (K4).
The reduction in these parameters indicates that the bioactive compounds in red ginger play a role in improving glucose and lipid metabolism. Gingerol and shogaol, the main components, are known to increase insulin sensitivity, inhibit lipogenesis, and enhance fatty acid oxidation. Furthermore, the anti-inflammatory properties of these compounds are thought to play a role in suppressing systemic inflammation that occurs in obesity.1-2,4-5
Figure 2 and Table 4 demonstrate that administration of red ginger rhizome extract for 4 weeks significantly (p < 0.05) lowered glucose, and profile lipid compared to the obese group. Glucose levels declined by 58.65% at 100 mg/kg bw and 62.86% at 200 mg/kg bw, compared to a 66.89% increase in the obese control group, confirming the antihyperglycemic potential of the extract. Similarly, triglyceride levels decreased by 62.39% and 65.46% at the respective doses, relative to an 81.58% elevation in obese controls, suggesting that lowering triglycerides contributes to body weight reduction. Free fatty acids were also reduced by 24.08% and 43.23% at 100 and 200 mg/kg bw, respectively, whereas the obese control group showed a 28.69% increase. Notably, the 200 mg/kg bw treatment exhibited superior efficacy in lowering free fatty acids compared to both the 100 mg/kg bw dose and the normal control. Administration of red ginger extract for 4 weeks in obese Wistar rats demonstrated stronger effects on glucose and triglyceride reduction compared to lidah mertua leaf extract at equivalent doses and treatment duration, whereas its impact on free fatty acids was comparatively lower. For lidah mertua leaf extract, decreases at 100 mg/kg bw were observed at 44.12% for glucose, 45.66% for triglycerides, and 53.36% for free fatty acids, while treatment at 200 mg/kg bw reduced these parameters by 45.53%, 48.79%, and 63.82%, respectively.24 These findings confirm that the bioactive compounds in red ginger extract play a crucial role in its activity. The novelty of this research lies in the identification of 11 bioactive compounds through LC-MS, which act synergistically and highlight the extract’s potential as a phytotherapeutic candidate for managing metabolic disorders.
The decrease in blood glucose levels in Wistar rats can occur due to the content of secondary metabolite compounds in red ginger rhizome extract which can help in inhibiting carbohydrate metabolism enzymes, namely α-glucosidase and α-amylase enzymes. This was reported by23 Rampengan et al. (2024), where in their research, ethanol and hexane extracts of red ginger help in inhibiting metabolic enzymes, namely α-amylase and α-glucosidase. Compounds that potentially have this mechanism are ar-curcumin, rutin, 6-shogaol, catechin, and quercetin.18,19,25 Other compounds such as gallic acid, naringenin, and 6-gingerol reduce blood glucose levels by increasing GLUT4 (glucose transporter 4),20 while kaempferol can increase insulin sensitivity.14
Triglyceride levels can decrease due to inhibition of lipolysis, thereby lowering lipid levels in the body. This decrease in triglyceride levels also affects free fatty acid levels in the body, as triglycerides are the main building block for free fatty acid formation. This decrease in triglyceride levels in the body will cause weight loss in obese Wistar rats due to the reduction in stored fat. Free fatty acids are also reduced due to the influence of secondary metabolites that can reduce lipogenesis, thus preventing free fatty acid biosynthesis in the body.4
Red ginger contains 11 bioactive compounds identified through LC-MS analysis, namely gallic acid, ar-curcumin, α-Zingiberene, naringenin, 6-shogaol, kaempferol, catechin, 6-gingerol, quercetin, 8-gingerol, and rutin. These compounds have reported anti-obesity, hypoglycemic and hypolipidemic activities via AMPK activation, inhibition of adipogenesis, modulation of mitochondrial function/thermogenesis (PGC-1α), pancreatic lipase inhibition and gut-microbiome effects.2,4-9,13-23, 25-26
Conclusion
Red ginger rhizome extract contains 11 bioactive compounds identified through LC-MS analysis, namely gallic acid, ar-curcumene, α-Zingiberene, naringenin, 6-shogaol, kaempferol, catechin, 6-gingerol, quercetin, 8-gingerol, and rutin, which collectively provide synergistic effects as phytotherapy candidates for the treatment of obesity and related metabolic disorders.
Acknowlegement
The author would like to thank Udayana of University for granting the research work. Special appreciation is extended to the Department of Chemistry, Udayana University, for providing access to laboratory facilities. The author is also deeply grateful to Udayana University’s Flagship Research 2024 and the Institute for Research and Community Service (LPPM) of Udayana University for funding this study.
Funding Source
The author(s) received financial support for the research, authorship, and/or publication of this article through Udayana University’s 2024 Flagship Research Program, administered by the Institute for Research and Community Service (LPPM) Udayana University with research assignment agreement number : B/255.320/UN14.4.A/PT.01.03/2024.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This study involves an experiment on animals. Research using test animals has been approved by the Animal Ethics Committee of the Faculty of Veterinary Medicine, Udayana University with Animal Ethics approval (number; B/203/UN14.2.9/PT.01.04/2024).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Ni Wayan Bogoriani: Conceptualization, Methodology, Writing – Original Draft.
- I Gusti Ayu Putu Eka Pratiwi: Data Collection, Analysis, Writing – Review & Editing.
- Ida Ayu Raka Astiti Asih: Data collection from LCMS analysis.
- Ni Wayan Mirawati: Data collection from free faty acid analysis, and writing
- Komang Tria Noviana Dewi: Data collection from glucose, trigliseride analysis, writing
References
- Preciado-Ortiz M.E., Gembe-Olivarez G., Erika Martínez-López , Rivera-Valdés J.J. Immunometabolic Effects of Ginger (Zingiber officinale Roscoe) Supplementation in Obesity: A Comprehensive Review. Molecules. 2025; 30(14), 2933: 1-29. https://doi.org/10.3390/molecules30142933
CrossRef - Zhang S, Kou X, Zhao H, Mak K.K, Balijepalli M.K, Pichika M.R. Zingiber officinale rubrum: Red Ginger’s Medicinal Uses Review. Molecules. 2022; 27(3), 775:1-31. https://doi.org/10.3390/molecules27030775
CrossRef - Sururi M, Bogoriani N.W, Asih, I.A.R.A. Characterization and Test of Antioxidant Activity of Red Ginger Extract (Zingiber officinale Rubrum) in Vitro. Cakra Kimia (Indonesian E-Journal of Applied Chemistry).2022; 10(2): 60-70.
- Hong K.H, Um M.Y, Ahn J, Ha T.Y. 6-Gingerol Ameliorates Adiposity and Inflammation in Adipose Tissue in High Fat Diet-Induced Obese Mice: Association with Regulating of Adipokines.2023; 15(15), 3457;1-12.https://doi.org/10.3390/nu15153457
CrossRef - Sang Y, Wang X, Wang Metabonomics and the gut microbiome analysis of the effect of 6-shogaol on improving obesity. Food Chemistry.2023; 404 ( Part B):134734. https://doi.org/10.1016/j.foodchem. 2022.134734
CrossRef - Mashhadi N.S, Ghiasvand R, Askari G, Hariri M, Darvishi l, Mofid M.R. Anti-oxidative and anti-inflammatory effects of ginger in health and physical activity: review of current of current evidence. International Journal of Preventive Medicine. 2013; 4 (Suppl 1): S36-S42
- Mahluji S, Attari V.E, Mobasseri M, Payahoo L, Ostadrahimi A, Golzari S.E.J. Effects of ginger (Zingiber officinale) on plasma glucose level, HbA1c and insulin sensitivity in type 2 diabetic patients. International Journal of Food Sciences and Nutrion. 2013; 64(6): 682-686. https://doi.org/10.3109/09637486.2013.
CrossRef - Safithri M, Kurniawati A. Formula of Piper crocatum, Cinnamomum burmanii, and Zingiber officinale extracts as a functional beverage for diabetics. Food Res. J. 2016; 23:(3):1123-1130.
- Masniah M, Rezi J, Faisal A.P. Isolation of Active Compounds and Activity Test of Red Ginger Extract (Zingiber officinales) as an Immunomodulator. Jurnal Riset Kefarmasian Indonesia. 2021; 3(2) : 77–91
CrossRef - Bogoriani N.W, Ariati K, Pratiwi I G.A.P.E. Potency of Balinese Kecombrang (Etlingera elatior) Extract. As Antioxidant Against The Activity of Superoxide (SOD), Glutathione (GSH) And Fatty liver in Obese rats. Dismutase. Biomedical & Pharmacology Journal. 2022; 15(1): 337-344 DOI: https://dx.doi.org/10.13005/ bpj/2372
CrossRef - Sukadana I M, Bogoriani N.W, Ariani M. Compounds in the stem of Etlingera elatior can reduce the levels of free Fatty Acid and Blood Glucose in Obesity Wistar Rats. Research J. Pharm. And Tech. 2023; 16(10): 4530-36. doi:10.52711/0974-360X.2023.00738
CrossRef - Xu Y, Tang G, Zhang C, Wang N, Feng Y. Gallic Acid and Diabetes Mellitus: Its Association with Oxidative Stress. Molecules. 2021; 26: 7115.
CrossRef - Kahkeshani N, Farzaei F, Fotouhi M, Alavi S.S. Pharmacological ects of gallic acid in health and diseases: A mechanistic review. J. Basic Med. Sci. 2019; 22: 225–237.
- Yang Y, Chen Z, Zhao X, et al. Mechanisms of Kaempferol in the treatment of diabetes: A comprehensive and latest review. Fronts Endocrinol. 2022;13:990299
CrossRef - Mechchate H, Es-safi I, Haddad H, Bekkari H, Grafov A, Bousta D. Combination of Catechin, Epicatechin, and Rutin:optimization of a novel complete antidiabetic formulation using a mixture design approach. The Journal of Nutritional Biochemistry. 2020; 88:108520
CrossRef - Adab Z, Eghtesadi S, Vafa M.R, et al. Effect of Turmeric on Glycemic Status, Lipid Profile, hs-CRP, and Total Antioxidant Capacity in Hyperlipidemic Type 2 Diabetes Mellitus Patients. Phytother Research: PTR. 2019; 33:1173–81.
CrossRef - Maradesha T, Patil S.M, Phanindra B, et al. Multiprotein Inhibitory Effect of Dietary Polyphenol Rutin from Whole Green Jackfruit Flour Targeting Different Stages of Diabetes Mellitus: Defining a Bio-Computational Stratagem. Separations. 2022; 9(262): 1-26
CrossRef - Mrabiti H.N, Jaradat N, Fichtali I, et al. A. Separation, Identification, and Antidiabetic Activity of Catechin Isolated from Arbutus unedo L. Plant Roots. 2018; 7(31): 1-9
CrossRef - Dhanya R, Arya A.D, Nisha P, Jayamurthy P. Quercetin, a Lead Compound Against Type 2 Diabetes Ameliorates Glucose Uptake via AMPK Pathway in Skeletal Muscle Cell Line. Frontiers in Pharmacology.2017; 8:336
CrossRef - Singh A.K, Raj V, Keshari A.K, et al. Isolated mangiferin and naringenin exert antidiabetic effect via PPARγ/GLUT4 dual agonistic action with strong metabolic regulation. Biol. Interact. 2018; 280:33–44
CrossRef - Wang K, Kong L, Wen X, et al. The Positive Effect of 6-Gingerol on High-Fat Diet and StreptozotocinInduced Prediabetic Mice: Potential Pathways and Underlying Mechanisms. Nutrients. 2023;15:824
CrossRef - Lian P, Huan Z, Wang Y, Yao H, Han S, Ge X. Protective effect of 8-Gingerol, a potent constituent of ginger, on acute lung injury following hemorrhagic shock in rats. Exp Anim. 2024; 73(4):446-457. doi: 10.1538/expanim.23-0178.
CrossRef - Rampengan D.D.C.H, Syahputra R.A, Halim P, et al. Red Ginger Confers Antioxidant Activity, Inhibits Lipid and Sugar Metabolic Enzymes, and Downregulates miR-21/132 Expression. Journal of Agriculture and Food Research. 2024; 18: 101526
CrossRef - Bogoriani N.W, Pratiwi IG.A.P.E, Bawa I G.A.G, Komang Tria Noviana Dewi K.T.N, Fudholi A. Effect of Intake of Lidah Mertua (Sansevieria trifasciata Laurentii) Leaf Extract on Free Fatty Acid, Glucose and Triglyceride Levels in Obese Wistar Rats and Compound Identification by LC-MS/MS. International Journal of Design & Nature and Ecodynamics. 2025; 20 (7): 1533-1540. https://doi.org/10.18280/ijdne.200710
CrossRef - Jiao W, Sang Y, Wang X, Wang S. Effects of 6-Shogaol on Glucose Uptake and Intestinal Barrier Integrity in Caco-2 Cells. 2023; 12: 503
CrossRef - Tanaka M, Sato A, Kishimoto Y, Mabashi-Asazuma H, Kondo K, Iida K. Gallic Acid Inhibits Lipid Accumulation via AMPK Pathway and Suppresses Apoptosis and Macrophage-Mediated Inflammation in Hepatocytes. Nutrients. 2020; 12, 1479: 1-12. doi:10.3390/nu12051479
CrossRef







