Manuscript accepted on :15-09-2025
Published online on: 26-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Neeta Rai
Second Review by: Dr. Abeer Gatea
Final Approval by: Dr. Eman Refaat Youness
Ibrahim Abbas Atiyah , Seree Niyomdecha
, Seree Niyomdecha and Dania Cheaha*
and Dania Cheaha*
Department of Biology, Division of Biological Science, Faculty of Science, Prince of Songkla University (PSU), Hatyai, Songkhla, Thailand
Corresponding Author E-mail: dania.c@psu.ac.th
DOI : https://dx.doi.org/10.13005/bpj/3264
Abstract
Major depressive disorder is associated with disrupted limbic network dynamics and impaired behavioral flexibility. While selective serotonin reuptake inhibitors (SSRIs) like fluoxetine are commonly used to treat depression, the acute neural circuit alteration underlying their therapeutic effects remains unclear. The purpose of this research was to examine the effects of acute fluoxetine administration (20 mg/kg) on cross-frequency interactions and local field potential (LFP) activity in the dorsal hippocampus (dHIP) and nucleus accumbens (NAc) during stress-induced behavioral despair. LFPs from the NAc and dHIP were simultaneously recorded, and depressive-like behavior was evaluated in mice using the tail suspension test (TST). The effects of fluoxetine on changes in brain oscillations and circuit synchronization were assessed using power spectral density (PSD) and phase–amplitude coupling (PAC) studies. Results showed fluoxetine significantly reduced immobility during the TST, indicative of rapid antidepressant-like effects. In the NAc, fluoxetine enhanced delta, alpha, beta, and both low and high gamma power, while in the dHIP, it increased gamma power and suppressed delta, theta, and alpha activity. PAC analysis revealed changes of delta/theta–gamma coupling in both regions, with a selective decrease in theta–low gamma coupling in the dHIP. These electrophysiological changes corresponded with improved behavioral engagement and consistent with enhanced monoaminergic modulation and circuit-level plasticity. In conclusion, acute fluoxetine induces rapid reorganization of limbic oscillatory dynamics, characterized by increased gamma activity and enhanced cross-frequency coupling. These findings provide evidence for gamma-band oscillations and PAC as candidate biomarkers of early antidepressant response and support the view that fluoxetine's therapeutic effects involve fast, network-level reconfiguration of mood-relevant circuits.
Keywords
Depression; Dorsal Hippocampus; Local Field Potential; Nucleus Accumbens; Phase-Amplitude Coupling; Tail Suspension Test
Download this article as:| Copy the following to cite this article: Atiyah I. A, Niyomdecha S, Cheaha D. Acute Fluoxetine Induces Rapid Reorganization of Limbic Oscillatory Dynamics: Gamma Oscillations and Cross-Frequency Coupling as Correlates of Antidepressant-Like Behavior. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Atiyah I. A, Niyomdecha S, Cheaha D. Acute Fluoxetine Induces Rapid Reorganization of Limbic Oscillatory Dynamics: Gamma Oscillations and Cross-Frequency Coupling as Correlates of Antidepressant-Like Behavior. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4gIcVmg |
Introduction
The neurobiological underpinnings of depression are complex and multifaceted, with considerable evidence pointing towards dysregulation within brain reward circuits.1,2 A hallmark symptom of depression is anhedonia, differentiated by a diminished capacity to feel pleasure (experience) and/or a reduced interest in activities that were once rewarding. 3 This impairment in reward processing is not merely a subjective feeling but is increasingly understood to stem from dysfunctional neural mechanisms. 4,5 Key among these mechanisms is the aberrant functioning of the nucleus accumbens (NAc), a major ventral striatal component essential for motivated behaviors, reward anticipation, and the processing of reward-related information, often associated with “wanting” or incentive salience.6,7 The hippocampus (HIP) is crucial for memory formation and emotional regulation.8
In depressed individuals, studies have consistently shown blunted activity in the NAc and other striatal regions in response to anticipated or received rewards, suggesting a deficit in the brain’s ability to process positive stimuli and drive motivated behavior.9 This hypoactivity is thought to involve aberrations in the mesolimbic dopamine pathway, a key circuit for reward.10
Pharmacological interventions for depression often target monoaminergic systems, with selective serotonin reuptake inhibitors (SSRIs) like fluoxetine being a first-line treatment.11 While the clinical efficacy of fluoxetine in ameliorating depressive symptoms is well-established, its acute neurophysiological effects on specific brain regions and their contribution to rapid changes in mood-related behaviors remain less understood. Neural oscillations, representing rhythmic electrical activity in the brain, are thought to be fundamental to information processing and communication within neural circuits.4 Beyond the power of individual frequency bands, the intricate coordination between different oscillatory rhythms, known as phase-amplitude coupling (PAC), is increasingly recognized as a crucial mechanism for integrating information across distinct neural processes.12 In complex cognitive and emotional processes, such as reward processing, PAC—where the phase of a low-frequency rhythm modulates the amplitude of high oscillations—is hypothesized to assist organize neuronal firing into functional assemblies and increasing communication between different brain areas.12–14 The PAC plays a critical role in organizing temporal dynamics of neuronal ensembles and may be disrupted in stress-related conditions.12,15 Alterations in these oscillatory dynamics, including PAC, have been observed in various neuropsychiatric disorders, including depression, and may serve as crucial biomarkers for disease state and treatment response, particularly in the context of reward circuit dysfunction. 16
The tail-suspension test (TST) is a commonly used preclinical model to assess antidepressant-like activity in rodents. This test measures the immobility time of mice suspended by their tails, with a reduction in immobility often interpreted as an antidepressant-like effect. Although the TST is sensitive to a variety of antidepressant compounds, the precise neural mechanisms underlying the behavioral changes observed in this model, particularly concerning the acute modulation of reward circuit activity, are not fully elucidated.
The purpose of this study is to characterize changes in PAC and assess the impact of a single acute dosage of fluoxetine on the NAc and dHIP neural oscillatory activity during the TST. We combined behavioral assessments with spectral power and PAC analyses to test the hypothesis that fluoxetine rapidly modulates cross-frequency coupling in a manner predictive of its antidepressant effects. By identifying frequency-specific electrophysiological changes associated with acute fluoxetine action, we aim to uncover early network signatures of antidepressant response and provide mechanistic insight into how serotonergic modulation impacts stress-related circuit function.
Materials and Methods
Animals
Male C57BL/6 adult mice (Nomura Siam International Co., Ltd.) weighing 35–40 g were kept separately in Prince of Songkla University’s animal house facility. The climate-controlled living area was 23 ± 2 °C, had a relative humidity of 55 ± 10%, and had a 12-hour light/dark cycle, with lights turning on at 07:00 and off at 19:00. Mice were provided with irradiated food pellets and distilled water without limits. To reduce stress, an acclimatization period of seven days was instituted before the experiments began.
Drugs and Chemicals
Fluoxetine hydrochloride (Flutine, Merck, Thailand) was prepared using distilled water. Following the day of the behavioral test and LFP recording, a single oral dosage of 20 mg/kg was administered. Our lab’s empirical data showing this particular dosage’s effectiveness in modifying neuronal activity in the NAc and dHIP,17,18 brain parts involved in depressive-like phenotypes. The 20 mg/kg dose was selected over lower, ineffective doses (e.g., 10 mg/kg), which prior studies indicate are insufficient to elevate extracellular dopamine or its metabolites in the NAc.19 Furthermore, this dosage has been shown to enhance oscillatory activity and potentiate dopaminergic and serotonergic signaling within these limbic structures,20 aligning with established mechanisms of fluoxetine action. In order to accomplish our main goal of producing strong, measurable alterations in LFP dynamics within the targeted neuronal circuits, this higher dosage had to be used. This high, acute dose was chosen to elicit robust signals for mechanistic study and may not mirror effects of chronic, clinical dosing.
Experimental Procedure
The overall experimental design is outlined in Figure 1. In brief, all animals underwent stereotaxic surgery for the implantation of recording electrodes in the NAc and dHIP, alongside a reference electrode positioned on the cerebellum. A 14-day postoperative recovery interval was provided. To minimize stress associated with the recording setup, mice were then habituated for three days (3 hours per day) by being placed individually in a transparent glass arena lined with clean bedding. On the day of the experiment, a 15-minute baseline LFP recording was first obtained from each mouse in this home cage-like environment. Subjects were then randomly allocated into two experimental cohorts (n=7/group) receiving an oral administration of either distilled water (DW, vehicle control) or fluoxetine (20 mg/kg; Flu20). Sixty minutes post-treatment, each mouse underwent the TST, during which both behavioral performance and LFP signals were recorded simultaneously for a 6-minute duration. All experimental sessions were carried out between 09:00 and 16:00 hours under standard housing conditions.
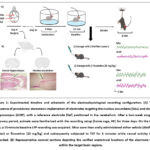 |
Figure 1: Experimental timeline and schematic of the electrophysiological recording configuration. |
Intracranial electrode implantation surgery
The surgical protocol for electrode implantation followed a previously established method.18 In short, the process was carried out as follows. Atropine sulfate (0.01 mg/kg) was given subcutaneously to mice to decrease bronchial secretions. Subsequently, xylazine hydrochloride (16 mg/kg; Sigma-Aldrich International, Switzerland) and zoletil (tiletamine-zolazepam, 50 mg/kg; Vibac AH, USA) were coadministered intraperitoneally to produce deep anesthesia. Each mouse’s head was stabilized by ear bars once it was positioned inside the stereotaxic apparatus. The scalp was cleansed and given a local anesthetic (20 mg/ml lidocaine) after it had been shaved at the surgery location. The sagittal incision made in the midline was made to reveal the surface of the skull. Bregma is used as the primary reference point for identifying stereotaxic coordinates according to a standard mouse brain atlas.21
At the determined locations for the target structures, small craniotomies were drilled. Silver wire electrodes (A-M Systems, USA) were lowered into the dHIP (DV -1.5 mm, ML +2.0 mm, AP -2.0 mm) and the NAc (DV -4.5 mm, ML +1.0 mm, AP +1.3 mm). On the surface of the cerebellum, a ground electrode (AP -6.5 mm, DV -2.0 mm) and an independent reference electrode were placed. To attach the assembly to the skull, stainless-steel anchor screws and dental acrylic cement were utilized (Unifast Trad, GC Dental Industrial Corp., Japan).
Postoperative Care and Histology
Following surgery, animals were kept in separate housing for at least 14 days while they recovered. Mice underwent a three-day course of subcutaneous injections to facilitate their recovery: carprofen (10 mg/kg; Best Equipment Center, Ltd., Thailand) for analgesia and ampicillin (100 mg/kg; General Drug House, Ltd., Thailand) for infection prevention. All electrodes’ anatomical locations were confirmed histologically at the end of the tests. Mice were transcardially perfused with saline and then 4% phosphate-buffered paraformaldehyde after being put to death by an intraperitoneal (i.p.) thiopental sodium overdose (100 mg/kg). The excised brains were coronally sectioned at 7 μm, post-fixed, and stained with H&E. By comparing the sections with a conventional mouse brain atlas, the positions of the electrode tips within the NAc and dHIP were verified (Figure 1B).
Tail Suspension Test (TST) Behavioral Analysis
The TST was used as a measure of despair-like behavior, with testing performed simultaneously alongside LFP recordings. For the test, adhesive tape is used to hang each mouse by its tail, positioned about 1 cm away from the end of the tail. The point of suspension was set 50 cm above the base of the apparatus. The full test duration was 6 minutes; however, to account for initial hyperactivity and adaptation, the first 2 minutes of each session were omitted from subsequent behavioral analysis.22 Immobility is defined as a complete cessation of active limb or body movements, excluding respiratory motions, with mice considered immobile when hanging passively and exhibiting complete motionlessness. Both the total duration of immobility and the latency to the first immobility were quantified. Animal behavior was video recorded using a camera positioned anterior to the test apparatus, and locomotor activity was analyzed offline using the OptiMouse MATLAB toolbox,23 a validated automated tracking system.
LFP Data Acquisition and Processing
Neural signals and concurrent behavioral data from freely moving mice during the TST were acquired via a synchronized video-LFP recording setup, implemented as reported previously.24 Raw LFP signals were digitized at a sampling rate of 2 kHz using a Power Lab (16/35) system equipped with a 16-bit analog-to-digital converter (AD Instruments, Australia). The signals were conditioned by using a series of filters: a high-pass filter (0.3 Hz cutoff), a low-pass filter (1 kHz cutoff), and a 50 Hz notch filter to eliminate mains interference. Data was stored for offline analysis using LabChart 7 Pro software (AD Instruments). Simultaneous video footage was recorded at a resolution of 640 × 480 pixels and a rate of 30 frames per second. Prior to analysis, all recorded LFP traces were carefully screened for movement artifacts or electrical noise; only segments with stable, high-quality signals were selected for further processing. Spectral analysis of the LFP data was conducted within LabChart 7 Pro using a Fast Fourier Transform (FFT) algorithm. For PAC assessment, the curated LFP data were exported in EDF format and analyzed with the Brainstorm toolbox running in the MATLAB environment.25
Power spectral density (PSD) analysis
The spectral analysis feature in LabChart 7 Pro was used to determine LFP power spectra. A bandpass filter with a frequency range of 1 to 100 Hz was used to process the LFP signals. Starting with the FFT function (2.56 s FFT size, Hanning window function, 50% overlap), the spectral power analysis shows that the power spectral density (PSD) is μV²/Hz. PSD values acquired during the TST were adjusted to the pre-treatment baseline for intergroup comparisons. This was done by using the following formula to determine the percentage change for each frequency bin between 1 and 100 Hz: (PSD during TST / Baseline PSD) × 100. After that, the normalized spectral power was averaged throughout the predefined frequency bands of interest, which were delta (0.5–4 Hz), theta (5–8 Hz), alpha (9–12 Hz), beta (13–30 Hz), low-gamma (31–45 Hz), and high-gamma (60–100 Hz). All investigations excluded the 45–55 Hz frequency band in order to reduce contamination from electrical line noise.
PAC analysis
PAC analysis was performed to investigate the interaction between the phase of slow oscillations and the amplitude of fast oscillations in the NAc and dHIP. The raw LFP signals from both the NAc and dHIP were first preprocessed using a bandpass filter with a frequency range of 1–100 Hz. PAC was computed using the modulation index (MI) method described by.26 We selected a 2–6-minute segment of LFP recordings during the TST for groups treated with distilled water and fluoxetine. All PAC computations, including the generation of comodulograms to graphically represent delta-gamma and theta-gamma coupling dynamics, were executed within the Brainstorm toolbox integrated with MATLAB.
Statistical analysis
All data are presented as the mean ± standard error of the mean (SEM). Statistical analyses were conducted using GraphPad Prism (Version 9.0.0), with a p-value of p ≤ 0.05 considered statistically significant. Behavioral data from TST were analyzed using an unpaired two-tailed t-test to compare the total immobility time between the control (DW) and fluoxetine (Flu20) groups. This test was deemed appropriate as it involved a single, planned comparison between two independent experimental groups.
The PSD data were reanalyzed to account for multiple comparisons across frequency bands. A two-way ANOVA was performed for each brain region (NAc and dHIP), with Treatment (DW vs. Flu20) and frequency band (delta, theta, alpha, beta, L-gamma, H-gamma) as independent factors. Following significant interaction, post-hoc tests with a Holm-Šídák correction for multiple comparisons were applied to identify significant differences between the treatment groups within each specific frequency band.
For the PAC analysis, the four pre-defined modulation index (MI) pairs in each brain region were compared using unpaired two-tailed t-tests. To control the family-wise error rate from these multiple comparisons, the Holm-Šídák correction was subsequently applied to the results.
Results
Behavioral Assessments
Behavioral analysis focused on immobility duration during the TST, specifically within the 2–6-minute interval. Fluoxetine-treated mice showed a significant reduction in the immobility time in comparison to controls (t = 15.6, df = 14, p ≤ 0.001). The mean immobility time was 87± 4 and 22 ± 3 seconds for DW and FLU20 groups, respectively. This robust and consistent decrease, as illustrated in Figure 2, supports the antidepressant-like effect of fluoxetine and indicates its sustained efficacy in attenuating depressive-like behavior over the observation period.
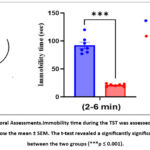 |
Figure 2: Behavioral Assessments.Immobility time during the TST was assessed across 2-6 min. The values show the mean ± SEM. The t-test revealed a significantly significant difference between the two groups (***p ≤ 0.001). |
PSD analysis in the NAc
The overall LFP power spectrum for the NAc during the TST is displayed in Figure 3A. To analyze the effect of fluoxetine across the different frequency bands, we conducted a two-way ANOVA.
The analysis revealed a significant main effect of treatment (F(1,72) = 81.2, p < 0.001), a significant main effect of frequency band (F(5,72) = 21.7, p < 0.001), and a significant interaction between treatment and frequency band (F(5,72) = 2.61, p = 0.032). This significant interaction indicates that the effect of fluoxetine on spectral power varied depending on the specific frequency band.
Post-hoc comparisons were used to pinpoint these specific differences. As illustrated in Figures 3B–G, acute fluoxetine administration resulted in significantly elevated power in the delta, alpha, beta, L-gamma, and H-gamma bands relative to the control (DW) group. However, theta power was not significantly altered by the treatment. These findings suggest that fluoxetine enhances oscillatory activity in the NAc across a broad but specific range of frequencies during the TST.
 |
Figure 3: Effects of acute fluoxetine on LFP spectral power in the NAc. (A) Average PSD across the 1–100 Hz frequency range during the TST for mice treated with distilled water (DW) or fluoxetine (Flu20). |
PSD analysis in the dHIP
The LFP power spectrum for the dorsal hippocampus (dHIP) during the TST is shown in Figure 4A. We performed a two-way ANOVA to analyze the effects of fluoxetine treatment across the different frequency bands.
The analysis revealed a highly significant interaction between treatment and frequency band (F(5,72) = 29.8, p < 0.001). This indicates that the effect of fluoxetine on LFP power was strongly dependent on the specific frequency. Because the drug suppressed some frequencies while enhancing others, the overall main effect of treatment was not significant (F(1,72) = 1.18, p = 0.281).
Post-hoc analysis clarified this complex, frequency-dependent effect, as shown in Figures 4B–G. Specifically, fluoxetine administration led to a significant suppression of power in the slower rhythms, including the delta, theta, and alpha bands, when compared to the control group. In sharp contrast, fluoxetine significantly enhanced power in higher frequencies, including both the low-gamma and high-gamma bands. Beta power was not significantly altered by the treatment. These findings demonstrate that acute fluoxetine reorganizes oscillatory dynamics in the dHIP, selectively dampening slow-wave activity while amplifying fast gamma oscillations.
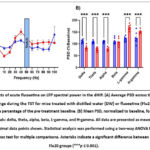 |
Figure 4: Effects of acute fluoxetine on LFP spectral power in the dHIP. (A) Average PSD across the 1–100 Hz frequency range during the TST for mice treated with distilled water (DW) or fluoxetine (Flu20). |
PAC analysis
The PAC analysis was performed on raw LFP signals recorded from the NAc and dHIP during the 2–6-minute interval of the TST. Data was extracted from the DW and Flu20 groups. Comodulogram plots revealed prominent coupling between slow-frequency phases (delta and theta) and high-frequency amplitudes (low and high gamma) in both brain regions (Figures 5A–B and 6A–B). To quantify coupling strength, modulation index (MI) values were computed for four PAC pairs: delta/L-gamma, delta/H-gamma, theta/L-gamma, and theta/H-gamma. MI values were averaged across animals within each group for statistical comparison.
In NAc figure 5C–F, fluoxetine significantly enhanced PAC across all four frequency pairs relative to the DW group: delta/L-gamma (t = 2.44, df = 12, p = 0.031), delta/H-gamma (t = 3.80, df = 12, p = 0.003), theta/L-gamma (t = 3.00, df = 12, p = 0.011), and theta/H-gamma (t = 3.20, df = 12, p = 0.008).
 |
Figure 5: The NAc PAC. (A–B) Representative group-averaged comodulograms for mice treated with distilled water (DW) or fluoxetine (20 mg/kg; FLU20) during the 2–6-minute interval of the TST. |
In the dHIP (Figure 6C–F), fluoxetine significantly increased delta/H-gamma (t = 3.55, df = 12, p = 0.004) and theta/H-gamma (t = 2.84, df = 12, p = 0.015) coupling but significantly decreased theta/L-gamma coupling (t = 2.31, df = 12, p = 0.039). There were no discernible intergroup variations in delta–low gamma PAC.
 |
Figure 6: The dHIP PAC. (A–B) Representative group-averaged comodulograms for mice treated with distilled water (DW) or fluoxetine (20 mg/kg; FLU20) during the 2–6-minute interval of the TST. |
Discussion
This study investigated the effects of acute fluoxetine on the NAc-dHIP circuit neural oscillatory dynamics during the TST, a validated behavioral paradigm for assessing depressive-like states in rodents.27 While behavioral assays confirmed a robust antidepressant-like effect of fluoxetine, evidenced by a significant reduction in immobility, our electrophysiological findings offer novel insights into the rapid circuit-level reorganization underlying this effect. PSD analysis revealed that fluoxetine profoundly modulated LFP activity in both the NAc and dHIP, two core components of the corticolimbic network critically implicated in emotional regulation and affective disorders.2,28
In the NAc, fluoxetine induced a broad enhancement of oscillatory power, particularly within the gamma range, consistent with previous studies linking gamma-band activity to cognitive engagement, synaptic plasticity, and affective stability.29,30 Gamma suppression has been widely observed in depression models and correlates with behavioral despair,31,32 thus, the observed gamma elevation may reflect a core mechanism by which fluoxetine exerts its antidepressant effects. Fluoxetine also significantly increased beta-band power in the NAc, which may reflect enhanced behavioral activation and motor readiness. Beta oscillations within the ventral striatum have been shown to respond to psychostimulants and are tightly regulated by dopaminergic tone 33. Given the NAc’s dense innervation by dopaminergic neurons from the VTA and its (D1- & D2)-expressing medium spiny neurons, 34,35 it is likely that fluoxetine’s serotonergic action indirectly boosts dopamine signaling, enhancing beta activity through D1-dominant pathways.
Additionally, delta-band power was elevated in the NAc following fluoxetine, which, although often associated with rest states, has also been linked to dopamine-related signaling in the basal ganglia during stress and reward.36,37 This increase may reflect a neuromodulatory adjustment within mesolimbic dopaminergic circuits, 38 particularly during active coping. Together, these frequency-specific changes in the NAc indicate that fluoxetine reorganizes the oscillatory landscape in a manner aligned with behavioral recovery, enhancing gamma- and beta-band activity associated with affective improvement and motor drive, and modulating delta rhythms potentially linked to dopaminergic tone.
In the dHIP, fluoxetine selectively increased low and high gamma power while suppressing slower rhythms, including delta, theta, and alpha bands. These results were identical to previous studies.18,39 Gamma oscillations in the hippocampus are essential for local circuit synchrony and are modulated by monoamines such as dopamine and Gamma oscillations in the hippocampus are essential for local circuit synchrony and are modulated by monoamines such as dopamine and serotonin.40–42 Dopaminergic stimulation has been shown to restore gamma activity in depression models, supporting the interpretation that fluoxetine may normalize hippocampal function via monoaminergic modulation. These gamma enhancements may reflect not only local circuit excitation but also a restoration of information processing and cognitive flexibility under stress. Studies have shown that reduced gamma dynamics in the hippocampus are associated with despair-like behaviors, while increased gamma burst activity is linked to adaptive, survival-related responses.43 Our findings underscore the importance of gamma oscillations in the dHIP as both a functional correlation and a potential biomarker of antidepressant efficacy.
To further explore circuit-level coordination, we examined PAC, a cross-frequency interaction that reflects hierarchical communication between neural oscillations. Fluoxetine significantly enhanced PAC across limbic structures. In the NAc, delta- and theta-phase coupling with low- and high-gamma amplitudes was markedly elevated, supporting a model in which fluoxetine may reflect dopaminergic-glutamatergic integration in stress-modulated circuits.26 The DW group, by contrast, exhibited diminished PAC strength, potentially reflecting monoaminergic hypofunction.
In the dHIP, fluoxetine increased delta- and theta-high gamma PAC while suppressing theta-low gamma interactions, indicating a temporal reorganization of oscillatory synchrony. The enhancement of high-frequency PAC may support increased precision in information encoding. At the same time, the suppression of theta–low gamma coupling could reflect a shift away from maladaptive network states induced by stress. This is consistent with evidence linking theta–gamma coupling to associative memory encoding and retrieval.15 Moreover, deep brain stimulation research has demonstrated that cognitive tasks improve theta–gamma coupling, and such coupling may be disrupted in neuropsychiatric conditions.44 The ability of fluoxetine to normalize the theta–high gamma interactions suggests that PAC alterations are not merely correlative, but potentially mechanistic in the expression and reversal of depressive-like states.
Collectively, our findings demonstrate that acute fluoxetine is sufficient to rapidly modulate both spectral power and cross-frequency coupling in the NAc and dHIP, reflecting circuit-wide reorganization. These changes parallel to behavioral recovery during the TST, supporting a framework in which antidepressant efficacy is mediated by dynamic, frequency-specific reconfiguration of limbic network activity. This supports emerging views that rapid antidepressant action—traditionally associated with agents like ketamine—may also be achievable with SSRIs under specific neurophysiological conditions, particularly via modulation of gamma oscillations and PAC.45,46 These results offer critical insights into the electrophysiological correlations of therapeutic engagement and highlight PAC and gamma activity as candidate biomarkers for antidepressant response.
Limitations
It’s important to consider several limitations when interpreting the findings of this study. First, our experiments were conducted exclusively in male mice, which may limit the generalizability of the results, especially given the known sex differences in the prevalence and treatment of major depressive disorder. Second, we administered a single, high acute dose of fluoxetine (20 mg/kg) designed to elicit a robust, measurable neurophysiological signal for mechanistic investigation. This regimen does not model the chronic, lower-dose administration typical of clinical SSRI treatment, and the long-term neuroadaptive effects may differ significantly. Third, the nature of our data is correlational; while we observed a strong association between changes in limbic oscillations and antidepressant-like behavior, this study cannot establish a causal link. Techniques such as optogenetics would be required to determine if these oscillatory changes directly drive the behavioral outcomes. Finally, while we recorded simultaneously from the NAc and dHIP, we did not directly measure the functional connectivity or information flow between these two regions. Future studies employing analyses like coherence or Granger causality are needed to understand how fluoxetine modulates the broader NAc-dHIP circuit dynamics. These limitations highlight important avenues for future research.
A critical limitation of this study is the inherent confound of motor activity within the TST paradigm. The observed electrophysiological changes, particularly the enhanced beta and gamma power in the NAc, occurred alongside a significant increase in physical struggling in the fluoxetine-treated mice. Consequently, it is challenging to definitively disentangle the neural correlates of a shift in motivational state from signals that are purely representative of increased motor output. However, our previous work confirms that the observed increase in gamma activity is a specific pharmacological action of fluoxetine and not merely an artifact of motor behavior. 18 Nevertheless, future experiments are necessary to fully isolate these distinct components of antidepressant action. For example, utilizing behavioral paradigms that assess motivation and reward with less demanding motor requirements, such as the sucrose preference test for anhedonia or an effort-based progressive ratio task, would be highly informative. Recording limbic oscillations during these tasks could clarify whether the observed changes in gamma activity and phase-amplitude coupling are fundamental biomarkers of an improved affective state, independent of the vigorous motor engagement seen in the TST.
Conclusion
In summary, our results demonstrate that a single, acute dose of fluoxetine significantly reduces despair-like behavior in the TST. This behavioral improvement is tightly linked with a rapid and profound reorganization of neural oscillations within the NAc and dHIP. Specifically, the antidepressant-like effect was associated with increased gamma-band power and modulated phase-amplitude coupling in these key limbic structures. These findings highlight that specific oscillatory dynamics, particularly in the gamma range, serve as potential circuit-level correlates of fluoxetine’s acute therapeutic action. While promising, these electrophysiological signatures should be interpreted with caution. Further studies in pathological models of depression are essential to validate whether these network changes are truly indicative of a sustained antidepressant response. Ultimately, this work provides a foundation for exploring the rapid neuromodulatory effects of SSRIs and points toward specific circuit dynamics that warrant deeper investigation in the search for better treatments for depression.
Acknowledgment
The authors thank members of the Biosignal Research Center for Health, Prince of Songkla University, Thailand, for their support and suggestions. The authors also thank the anonymous reviewers and the editor for their useful suggestions, which helped to improve this manuscript.
Funding Sources
This research was funded by the Prince of Songkla University, Graduate School, Grant Number PHD2565-003 (2022).
Conflicts of Interest
The authors have no conflicts of interest to declare in relation to this study.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author upon request.
Ethics Statement
All experimental procedures involving animals were conducted in strict accordance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines and the European Science Foundation criteria for the ethical use of animals in research. The protocol was reviewed and approved by the Prince of Songkla University Animal Care and Use Committee (Project License Number: MHESI 68014/2048). Every effort was made to minimize animal suffering and to reduce the number of animals used in the study.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to produce material from other sources
Not Applicable
Authors’ Contributions
Ibrahim Abbas Atiyah: Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing. Seree Niyomdecha: Methodology, Visualization, Writing—original draft, Writing—review & editing. Dania Cheaha: Supervision, Conceptualization, Funding Acquisition, Project Administration, Validation, Writing—Review & Editing.
References
- Nestler EJ. Role of the Brain’s Reward Circuitry in Depression: Transcriptional Mechanisms. In: International Review of Neurobiology. Academic Press Inc. 2015;124:151-170.
CrossRef - Nestler EJ, Carlezon WA. The Mesolimbic Dopamine Reward Circuit in Depression. Biol Psychiatry. 2006;59(12):1151-1159.
CrossRef - Hanuka S, Olson EA, Admon R, et al. Reduced anhedonia following internet-based cognitive-behavioral therapy for depression is mediated by enhanced reward circuit activation. Psychol Med. 2023;53(10):4345-4354.
CrossRef - Ma Y, Guo C, Luo Y, et al. Altered neural activity in the reward-related circuit associated with anhedonia in mild to moderate Major Depressive Disorder. J Affect Disord. 2024;345:216-225.
CrossRef - Halahakoon DC, Kieslich K, O’Driscoll C, Nair A, Lewis G, Roiser JP. Reward-Processing Behavior in Depressed Participants Relative to Healthy Volunteers. JAMA Psychiatry. 2020;77(12):1286.
CrossRef - De Groote A, de Kerchove d’Exaerde A. Thalamo-Nucleus Accumbens Projections in Motivated Behaviors and Addiction. Front Syst Neurosci. 2021;15:711350..
CrossRef - Xu Y, Lin Y, Yu M, Zhou K. The nucleus accumbens in reward and aversion processing: insights and implications. Front Behav Neurosci. 2024; 18:1420028.
CrossRef - Fanselow MS, Dong HW. Are the Dorsal and Ventral Hippocampus Functionally Distinct Structures? Neuron. 2010;65(1):7-19.
CrossRef - Yang X, Su Y, Yang F, et al. Neurofunctional mapping of reward anticipation and outcome for major depressive disorder: a voxel-based meta-analysis. Psychol Med. 2022;52(15):3309-3322.
CrossRef - Wang S, Leri F, Rizvi SJ. Anhedonia as a central factor in depression: Neural mechanisms revealed from preclinical to clinical evidence. Prog Neuropsychopharmacol Biol Psychiatry. 2021;110:110289.
CrossRef - D. Rosenblat J, McIntyre RS. Pharmacological Treatment of Major Depressive Disorder. Major Depressive Disorder. 2020:103-119.
CrossRef - Voytek B, Canolty RT, Shestyuk A, Crone NE, Parvizi J, Knight RT. Shifts in gamma phase-amplitude coupling frequency from theta to alpha over posterior cortex during visual tasks. Front Hum Neurosci. 2010;4:191.
CrossRef - Zheng C, Zhang T. Alteration of phase-phase coupling between theta and gamma rhythms in a depression-model of rats. Cogn Neurodyn. 2013;7(2):167-172.
CrossRef - Samiee S, Lévesque M, Avoli M, Baillet S. Phase-amplitude coupling and epileptogenesis in an animal model of mesial temporal lobe epilepsy. Neurobiol Dis. 2018;114:111-119.
CrossRef - Tort ABL, Komorowski RW, Manns JR, Kopell NJ, Eichenbaum H. Theta-Gamma Coupling Increases during the Learning of Item-Context Associations. 2009;8 106(49):20942-7.
CrossRef - Luque‐García A, Teruel‐Martí V, Martínez‐Bellver S, Adell A, Cervera‐Ferri A, Martínez‐Ricós J. Neural oscillations in the infralimbic cortex after electrical stimulation of the amygdala. Relevance to acute stress processing. Journal of Comparative Neurology. 2018;526(8):1403-1416.
CrossRef - Atiyah I, Kumarnsit E, Cheaha D, Atiyah IA, Kongnual R. The Effects of Acute Fluoxetine treatment on Hippocampal Spectral Power Density. In: RSU International Research Conference 2020. 2020:441-448.
- Atiyah IA, Niyomdecha S, Cheaha D. Neural oscillations in the nucleus accumbens–dorsal hippocampal circuits and behavioral effects of acute fluoxetine administration during the Tail suspension test in mice. Exp Brain Res. 2025;243(7):174.
CrossRef - Clark RN, Ashby CR, Dewey SL, Ramachandran PV, Strecker RE. Effect of acute and chronic fluoxetine on extracellular dopamine levels in the caudate-putamen and nucleus accumbens of rat. Synapse. 1996;23(3):125-131.
CrossRef - Shen HW, Hagino Y, Kobayashi K, et al. Regional differences in extracellular dopamine and serotonin assessed by in vivo microdialysis in mice lacking dopamine and/or serotonin transporters. Neuropsychopharmacology. 2004;29(10):1790-1799.
CrossRef - Paxinos G, Franklin KBJ. The mouse brain in stereotaxic coordinates: hard cover edition. Academic Press. 2001;2:360.
- Can A, Dao DT, Terrillion CE, Piantadosi SC, Bhat S, Gould TD. The Tail Suspension Test. JoVE (Journal of Visualized Experiments). 2012;(59):e3769.
CrossRef - Ben-Shaul Y. OptiMouse: A comprehensive open source program for reliable detection and analysis of mouse body and nose positions. BMC Biol. 2017;15(1):1-22.
CrossRef - Khunphet P, Kumarnsit E, Issuriya A, Cheaha D. Estrogen deficiency affects synchronized neural connectivity in the olfactory bulb-nucleus accumbens circuit: A local field potential study in ovariectomized mouse model. Horm Behav. 2024;164:105587.
CrossRef - Tadel F, Baillet S, Mosher JC, Pantazis D, Leahy RM. Brainstorm: A user-friendly application for MEG/EEG analysis. Comput Intell Neurosci. 2011;2011:879716.
CrossRef - Andino-Pavlovsky V, Souza AC, Scheffer-Teixeira R, Tort ABL, Etchenique R, Ribeiro S. Dopamine modulates delta-gamma phase-amplitude coupling in the prefrontal cortex of behaving rats. Front Neural Circuits. 2017;11:29.
CrossRef - Steru L, Chermat R, Thierry B, Simon P. The Tail Suspension Test: A New Method for Screening Antidepressants in Mice. Vol 85.; 1985.
CrossRef - Russo SJ, Nestler EJ. The brain reward circuitry in mood disorders. Nat Rev Neurosci. 2013;14(9):609-625.
CrossRef - Guan A, Wang S, Huang A, et al. The role of gamma oscillations in central nervous system diseases: Mechanism and treatment. Front Cell Neurosci. 2022;16:962957.
CrossRef - Mcewen BS. Central Effects of Stress Hormones in Health and Disease: Understanding the Protective and Damaging Effects of Stress and Stress Mediators. Eur J Pharmacol. 2028;583(2-3):174-185.
CrossRef - Yin YY, Yan JZ, Lai SX, et al. Gamma oscillations in the mPFC: A potential predictive biomarker of depression and antidepressant effects. Prog Neuropsychopharmacol Biol Psychiatry. 2024;129: 110893.
CrossRef - Iturra-Mena AM, Aguilar-Rivera M, Arriagada-Solimano M, Pérez-Valenzuela C, Fuentealba P, Dagnino-Subiabre A. Impact of stress on gamma oscillations in the rat nucleus accumbens during spontaneous social interaction. Front Behav Neurosci. 2019;13:151.
CrossRef - Bymaster FP, Zhang W, Carter PA, et al. Fluoxetine, but not other selective serotonin uptake inhibitors, increases norepinephrine and dopamine extracellular levels in prefrontal cortex. Psychopharmacology (Berl). 2002;160(4):353-361.
CrossRef - Attachaipanich S, Ozawa T, Macpherson T, Hikida T. Dual Roles for Nucleus Accumbens Core Dopamine D1-Expressing Neurons Projecting to the Substantia Nigra Pars Reticulata in Limbic and Motor Control in Male Mice. eNeuro. 2023;10(6):ENEURO.0082-23.2023.
CrossRef - Cole SL, Robinson MJF, Berridge KC. Optogenetic self-stimulation in the nucleus accumbens: D1 reward versus D2 ambivalence. PLoS One. 2018;13(11):e0207694.
CrossRef - Whalen TC, Willard AM, Rubin JE, Gittis AH. Delta Oscillations Are a Robust Biomarker of Dopamine Depletion Severity and Motor Dysfunction in Awake Mice. J Neurophysiol. 2020;124(2)312-329.
CrossRef - Lemaire N, Hernandez LF, Hu D, Kubota Y, Howe MW, Graybiel AM. Effects of dopamine depletion on LFP oscillations in striatum are task- and learning-dependent and selectively reversed by L-DOPA. Proc Natl Acad Sci U S A. 2012;109(44):18126-18131.
CrossRef - Mohebi A, Pettibone JR, Hamid AA, et al. Dissociable dopamine dynamics for learning and motivation. Nature. 2019;570(7759):65-70.
CrossRef - Atiyah I, Atiyah IA, Kongnual R, Kumarnsit E, Cheaha D. The Effects of Acute Fluoxetine Treatment on Hippocampal Spectral Power Density. RSU International Research Conference. 2020;441-448.
- Wang L, Zhao D, Wang M, et al. Modulation of Hippocampal Gamma Oscillations by Dopamine in Heterozygous Reeler Mice in vitro. Front Cell Neurosci. 2020;13:586.
CrossRef - Reakkamnuan C, Cheaha D, Kumarnsit E. Nucleus Accumbens Local Field Potential Power Spectrums, Phase-Amplitude Couplings and Coherences Following Morphine Treatment. Acta Neurobiol Exp (Wars). 2017;77(3):214-224.
CrossRef - Reakkamnuan C, Cheaha D, Samerphob N, Sa-ih N, Kumarnsit E. Adaptive changes in local field potential oscillation associated with morphine conditioned place preference in mice. Physiol Behav. 2021;235.
CrossRef - Pollali E, Hollnagel JO, Çalışkan G. Hippocampal gamma-band oscillopathy in a mouse model of Fragile X Syndrome Title: Hippocampal gamma oscillopathy in FMR1 knockout mouse. bioRxiv. 2021
CrossRef - Sun Y, Giacobbe P, Tang CW, et al. Deep brain stimulation modulates gamma oscillations and theta-gamma coupling in treatment resistant depression. Brain Stimul. 2015;8(6):1033-1042.
CrossRef - Fuchikami M, Thomas A, Liu R, et al. Optogenetic stimulation of infralimbic PFC reproduces ketamine’s rapid and sustained antidepressant actions. Proc Natl Acad Sci U S A. 2015;112(26):8106-8111.
CrossRef - Zhang HL, Sun Y, Wu ZJ, et al. Hippocampal PACAP signaling activation triggers a rapid antidepressant response. Mil Med Res. 2024;11(1):49.
CrossRef
Abbreviations list
DW Distilled water
dHIP Dorsal hippocampus
FLU20 Fluoxetine 20 mg/kg
LFP Local field potential
NAc Nucleus accumbens
PAC Phase amplitude coupling
PSD Power spectral density
SEM Standard error of the mean
TST Tail suspension test







