Manuscript accepted on :22-08-2025
Published online on: 08-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Arif Ansori
Second Review by: Dr. Hassan Shora
Final Approval by: Dr. Prabhishek Singh
Ngakan Ketut Wira Suastika* and Ketut Suega
and Ketut Suega
Department of Internal Medicine Faculty of Medicine, Udayana University, Denpasar, Indonesia
Corresponding Author E-mail: wira.suastika@unud.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3255
Abstract
Non-Hodgkin lymphoma (NHL) comprises a diverse array of lymphoid malignancies with different clinical presentations and prognostic outcomes. Ki-67, a nuclear protein, is a potential biomarker for assessing disease aggressiveness and predicting therapeutic responses in NHL. This retrospective cohort study aimed to evaluate the association between Ki-67 expression, aggressiveness, and therapeutic response in 87 patients with NHL who received first-line chemotherapy. Ki-67 expression was assessed using immunohistochemistry, and the therapeutic response was evaluated based on the International Working Group Consensus Response Evaluation Criteria in Lymphoma (RECIL 2017). Significant differences in Ki-67 expression levels were found between aggressive and indolent NHL (p < 0.001) and between the stable-progressive disease and partial-complete response groups (p = 0.006). A Ki-67 expression cut-off of >55% distinguished aggressive from indolent NHL, with an area under the curve (AUC) of 0.938 (p < 0.001). In aggressive NHL, a Ki-67 expression cut-off of >75% predicted stable-progressive disease (AUC = 0.719, p = 0.005) with a relative risk (RR) of 2.07 (p = 0.042). In indolent NHL, a cut-off of >35% was not statistically significant for predicting therapeutic response. These findings suggest an association between Ki-67 expression, aggressiveness, and therapeutic response in patients with NHL. However, further studies with larger cohorts are required to validate Ki-67 as a reliable and independent predictor in clinical practice.
Keywords
Aggressiveness; Biomarker; Ki-67; Non-Hodgkin's lymphoma; Therapeutic response
Download this article as:| Copy the following to cite this article: Suastika N. K. W, Suega K. Ki-67 Expression as a Biomarker for Aggressiveness and Therapeutic Response in Non-Hodgkin's Lymphoma. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Suastika N. K. W, Suega K. Ki-67 Expression as a Biomarker for Aggressiveness and Therapeutic Response in Non-Hodgkin's Lymphoma. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3JS90XB |
Introduction
Non-Hodgkin lymphoma (NHL) is the most commonly diagnosed hematological neoplasm worldwide. More than 500,000 new cases are diagnosed.1,2 In recent decades, the incidence of NHL has increased significantly.3 Non-Hodgkin lymphoma is a complex group of lymphoid malignancies that exhibit clinical heterogeneity, ranging from indolent to highly aggressive.4 This heterogeneity can complicate treatment strategy planning and prognosis assessment. Indolent NHL may only require non-intensive therapy or even no immediate therapy, whereas aggressive NHL requires rapid and intensive treatment to increase survival.5,6 A comprehensive understanding of these heterogeneous clinical manifestations is essential for developing effective management strategies. Reliable biomarkers are crucial for determining disease aggressiveness and predicting therapeutic responses, enabling clinicians to develop effective therapeutic strategies that enhance patient prognosis.
Ki-67, a nuclear protein, is frequently used as a biomarker for cell growth and replication.7 Its expression is evident across each stage of the cell cycle, excluding the resting phase.8,9 The expression of Ki-67 protein is crucial for the diagnosis and prognosis of several malignancies.10,11,12 A high Ki-67 index in non-Hodgkin’s lymphoma has been recognized as a potential indicator of a more aggressive course of the disease and poorer prognosis.13 However, the application of Ki-67 in NHL remains inconsistent, necessitating further studies. Some studies have found a strong relationship between Ki-67 expression and aggressiveness and therapeutic response, whereas others have found a weak or non-existent relationship.10,14 Moreover, studies investigating the predictive significance of Ki-67 across different histological types of NHL is lacking.
This study aimed to investigate the association between Ki-67 expression, disease aggressiveness, and therapeutic response in patients with NHL. By identifying and clarifying this association, Ki-67 may help clinicians determine appropriate and effective therapeutic strategies for patients with NHL.
Materials and Methods
Study design and participants
This study employed an observational design with a retrospective cohort approach. Study participants were consecutively sampled from patients with NHL aged 18 years or older who received complete first-line chemotherapy at Professor Ngoerah Hospital, Bali, between January 2023 and October 2024. Subjects who met the inclusion criteria were recorded for therapeutic response, and epidemiological, clinical, laboratory, histopathological, and immunohistochemical data were collected retrospectively. Patients who did not complete all cycles of first-line chemotherapy and those with incomplete data were excluded.
Non-Hodgkin’s lymphoma is classified into two categories, aggressive and indolent, based on its clinical and histopathological features. Aggressive types include diffuse large B-cell lymphoma (DLBCL), cutaneous lymphoma, plasmablastic lymphoma, anaplastic lymphoma, and natural killer (NK)/T-cell lymphomas. Indolent types include follicular lymphoma, mucosa-associated lymphoid tissue (MALT) lymphoma, marginal zone lymphoma (MZL), and mantle cell lymphoma. The clinical stage was assessed according to the Ann Arbor criteria.15 Stages I to IIA were classified as early stages, whereas stages IIB to IV were classified as advanced stages.16
Ki-67 expression was assessed by immunohistochemistry (IHC). This method involves the staining of tissue samples using antibodies that are specific to Ki-67, followed by the calculation of the percent of Ki-67 positive cells. Patients received systemic therapy with cyclophosphamide, doxorubicin, vincristine, and prednisone with rituximab (R-CHOP) as first-line therapy according to the protocol. Therapeutic response was classified into two categories: partial-complete response and stable-progressive disease. The assessment was based on the International Working Group Consensus Response Evaluation Criteria in Lymphoma (RECIL 2017).17 Response assessment is performed using clinical examination and imaging techniques, such as ultrasonography, magnetic resonance imaging (MRI), or computed tomography (CT).
Data Analysis
The Mann-Whitney U test was applied to determine the differences in Ki-67 expression levels based on aggressiveness and therapeutic response. The optimal cut-off value of Ki-67 expression for assessing aggressiveness and predicting the therapeutic response has been determined using the receiver operating characteristic (ROC) curve. To identify the Ki-67 cut-off for predicting therapeutic response, ROC analysis was performed based on the type of aggressive or indolent LNH. The chi-square test was employed to determine the relative risk of high Ki-67 expression levels for the development of stable-progressive disease in patients with NHL after completing a cycle of chemotherapy.
Data analysis was performed using SPSS software (version 25.0). The significance threshold was set at p < 0.05.
Results
A total of 87 patients were enrolled in this study. The majority of patients (60%) had histological-type DLBCL, and 33% were in stage IIB. A total of 31% of patients achieved a complete response (Table 1).
Table 1: Characteristics of Patients
| Variable | Value(n = 87) |
| Age, years | 57 (23 – 82) |
| Sex, n (%) | |
| Female | 33 (37.9) |
| Male | 54 (62.1) |
| Histologic type, n (%) | |
| DLBCL | 53 (60.9) |
| Follicular lymphoma | 18 (20.7) |
| Marginal zone lymphoma | 3 (3.4) |
| NK/T cell lymphoma | 3 (3.4) |
| Mantle cell lymphoma | 5 (5.7) |
| MALT lymphoma | 2 (2.3) |
| Plasmablastic lymphoma | 1 (1.1) |
| Cutaneous lymphoma | 1 (1.1) |
| Anaplastic lymphoma | 1 (1.1) |
| Aggressiveness, n (%) | |
| Aggressive | 61 (70.1) |
| Indolent | 26 (29.9) |
| Stage of disease, n (%) | |
| IB | 5 (5.7) |
| IIA | 16 (18.4) |
| IIB | 29 (33.3) |
| IIIA | 4 (4.6) |
| IIIB | 28 (32.2) |
| IV | 5 (5.7) |
| Chemotherapy response, n (%) | |
| Complete response | 27 (31.0) |
| Partial response | 26 (29.9) |
| Stable disease | 26 (29.9) |
| Progressive disease | 8 (9.2) |
| Ki-67 expression, % | 70 (10 – 90) |
Numerical data are expressed as median (minimum-maximum), while categorical variables are represented as counts (percentages).
Table 2 shows the significant differences in disease stage and Ki-67 expression between patients with aggressive and indolent types of LNH. A significant difference in Ki-67 expression was observed between the stable-progressive and partial-complete response groups.
Table 2: The difference in variable values based on aggressiveness and response to first-line therapy in NHL
| Variable | Value | p-value | Value | p-value | ||
| Aggressive (n = 61) | Indolent (n = 26) | Stabile – Progressive disease(n = 34) | Partial – Complete (n = 53) | |||
| Age category | ||||||
| >60 years old | 21 (63.6) | 12 (36.4) | 0.429 | 15 (45.5) | 18 (54.5) | 0.468 |
| £60 years | 40 (74.1) | 14 (25.9) | 19 (35.2) | 35 (64.8) | ||
| Sex, n (%) | ||||||
| Female | 22 (66.7) | 11 (33.3) | 0.758 | 11 (33.3) | 22 (66.7) | 0.527 |
| Male | 39 (72.2) | 15 (27.8) | 23 (42.6) | 31 (57.4) | ||
| Comorbidities, n (%) | ||||||
| With comorbidities | 21 (72.4) | 8 (27.6) | 1.000 | 14 (48.3) | 15 (51.7) | 0.313 |
| Without comorbidities | 40 (69.0) | 18 (31.0) | 20 (34.5) | 38 (65.5) | ||
| Stage of disease | ||||||
| Advanced stage | 53 (79.1) | 14 (20.9) | 0.002* | 26 (38.8) | 41 (61.2) | 0.923 |
| Early stage | 8 (40.0) | 12 (60.0) | 8 (40.0) | 12 (60.0) | ||
| Hemoglobin level, gr/dL | 11.7 (4.2 – 15.8) | 12.3 (6.2 – 15.9) | 0.654 | 11.70 (4.17 – 15.90) | 12.20 (6.50 – 15.11) | 0.423 |
| White blood cell counts, x103/mL | 8.16 (2.49 – 16.76) | 7.59 (2.84 – 33.55) | 0.461 | 7.70 (2.84 – 13.88) | 8.29 (2.49 – 33.55) | 0.176 |
| Platelet count, x103/mL | 295.0 (65 – 709) | 289.95 (3 – 542) | 0.922 | 288.40 (65.17 – 675.00) | 300.00 (3.00 – 709.00) | 0.509 |
| Albumin level, gr/dL | 3.40 (1.70 – 4.60) | 3.50 (1.98 – 5.20) | 0.586 | 3.40 (1.90 – 4.60) | 3.50 (1.70 – 5.20) | 0.149 |
| BUN, mg/dL | 9.80 (4.90 – 28.5) | 11.00 (6.00 – 73.70) | 0.339 | 10.15 (6.60 – 73.70) | 10.30 (4.90 – 28.50) | 0.784 |
| Serum creatinine, mg/dL | 0.91 (0.41 – 4.24) | 1.46 (0.49 – 8.50) | 0.406 | 0.85 (0.65 – 2.26) | 0.82 (0.56 – 2.44) | 0.273 |
| SGOT, U/L | 21.10 (12.90 – 118.00) | 25.40 (15.00 – 98.00) | 0.224 | 21.95 (14.30 – 118.00) | 22.40 (12.90 – 98.00) | 0.821 |
| SGPT, U/L | 18.50 (4.00 – 91.00) | 17.05 (4.00 – 88.00) | 0.731 | 16.05 (5.00 – 52.00) | 19.00 (4.00 – 91.00) | 0.053 |
| Ki-67 expression, % | 80 (30 – 90) | 30 (10 – 60) | <0.001* | 70 (20 – 90) | 60 (10 – 90) | 0.006* |
Numerical data are expressed as median (minimum-maximum), while categorical variables are represented as counts (percentages).
*: statistically significant
For the determination of NHL aggressiveness, we found a cut-off value of Ki-67 expression of >55% to distinguish between aggressive and indolent NHL, with an AUC of 0.938 (95% CI, 0.892–0.984), p < 0.001 (Graph 1A and Table 3). For predicting therapeutic response, we identified a Ki-67 expression cut-off value of >75% as being associated with the occurrence of stable-progressive disease in aggressive NHL, with an AUC of 0.719 (95% CI, 0.588-0.849), p = 0.005 (Graph 1B and Table 3). In indolent NHL, we identified a cut-off value of >35% with an AUC of 0.614; however, this finding was not statistically significant (Graph 1C and Table 3).
Table 3: The optimal cut-off value of Ki-67 expression levels for determining aggressiveness and predicting therapy response in non-Hodgkin lymphoma patients
| Cut-off | Sensitivity(%) | Specificity(%) | Area under curve | 95% Confidence interval | p-value | |
| The cut-off value for aggressiveness | ||||||
| Ki-67 expression, % | >55 | 83.6 | 96.2 | 0.938 | 0.892 – 0.984 | <0.001* |
| Cut-off value for predicting therapeutic response | ||||||
| Aggressive NHL | ||||||
| Ki-67 expression, % | >75 | 68.2 | 60.0 | 0.719 | 0.588 – 0.849 | 0.005* |
| Indolent NHL | ||||||
| Ki-67 expression, % | >35 | 55.6 | 58.8 | 0.614 | 0.391 – 0.838 | 0.346 |
*: statistically significant; AUC: area under the curve
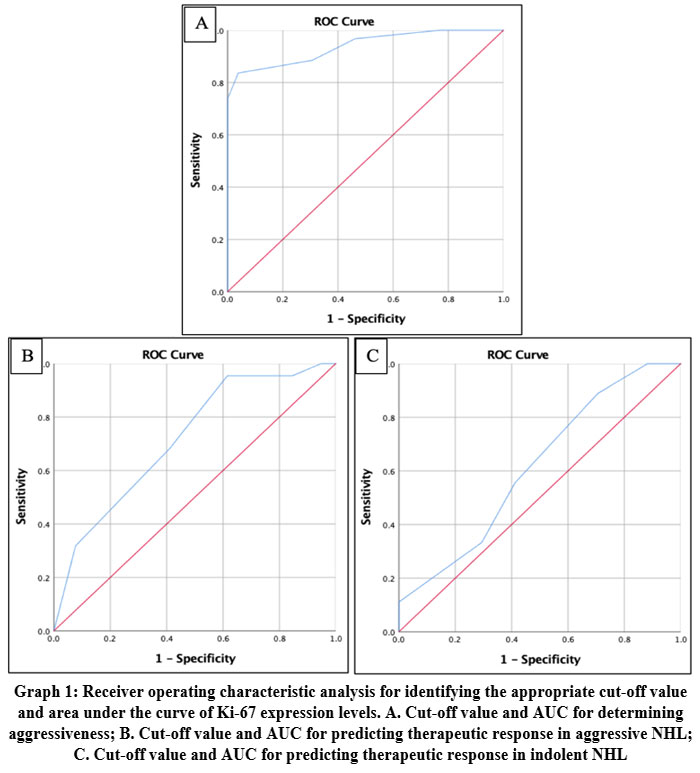 |
Graph 1: Receiver operating characteristic analysis for identifying the appropriate cut-off value and area under the curve of Ki-67 expression levels. Click here to view Graph |
Using Chi-square analysis, we found that high Ki-67 expression levels (greater than 75%) had a relative risk (RR) of 2.07 (95% CI, 1.01–4.36; p = 0.042) for the development of stable-progressive disease after first-line therapy in aggressive NHL.
Discussion
Our study found significant differences in Ki-67 expression levels between patients with aggressive and indolent NHL. Several studies have supported these findings. Studies have reported a mean Ki-67 index of 97.6% for aggressive lymphomas.13 Diffuse large B-cell lymphoma, the predominant form of aggressive NHL, exhibits a mean Ki-67 index of 66.4%.18 In indolent NHL, Ki-67 expression levels were generally low, with a mean of 26.6%. Follicular lymphoma, the predominant form of indolent non-Hodgkin lymphoma, exhibits an average Ki-67 expression level of 34.6%.18 The study also showed that high Ki-67 expression in patients with NHL could predict a more aggressive disease course, necessitating more intensive treatment regimens.18 Higher Ki-67 expression levels are associated with advanced disease stages and higher lactate dehydrogenase (LDH) levels, indicating a more aggressive disease.19 Beyond percentage differences, Ki-67 expression may also exhibit variability in staining intensity and nuclear distribution, with certain tumors displaying focal or heterogeneous staining patterns. This variability can affect scoring accuracy and inter-observer agreement, highlighting the need for standardized evaluation methods.20,21
Our study found that a Ki-67 expression level >55% was the optimal cut-off point for differentiating aggressive lymphoma from indolent lymphoma. This cut-off value was supported by a high AUC of 0.938, indicating a highly accurate performance. Other studies have found that Ki-67 expression levels greater than 45% can help discriminate aggressive lymphoma from indolent lymphoma.13,22 In NHL, high Ki-67 expression is associated with high tumor growth rates and poor prognoses. These findings emphasize the role of Ki-67 in risk stratification and guide clinicians in therapeutic decision-making.
We found significant differences in Ki-67 expression levels between patients with stable-progressive disease and those with partial-complete responses. The present findings are consistent with the results of other studies that found higher levels of Ki-67 expression to be correlated with higher International Prognostic Index (IPI) scores, indicating a poorer treatment response and prognosis.23,24 We found that the cut-off level for Ki-67 expression was greater than 75% for the occurrence of stable-progressive disease in aggressive NHL. The risk of stable-progressive disease is two-fold higher in patients with high Ki-67 expression. These results align with studies on DLBCL, which have found that Ki-67 expression levels greater than 70% are associated with lower overall survival (OS).13,25 However, Hubner et al. found that the IPI score was a more significant risk factor for OS than Ki-67 in their multivariate analysis.25 The modified Prognostic Index for T-cell lymphoma (mPIT) integrates Ki-67 into its scoring system, thereby reinforcing its prognostic significance in T-cell lymphomas.26,27
The prognostic significance of Ki-67 varies among the different NHL subtypes. In DLBCL, elevated Ki-67 levels are consistently correlated with adverse outcomes.13,25,28 Conversely, the results for follicular lymphoma are more heterogeneous.13,22 In indolent NHL, we identified a cut-off value of >35% for the occurrence of stable-progressive disease; however, this finding was not statistically significant. Another study on follicular lymphoma (FL) found that Ki-67 expression greater than 30% was associated with longer progression-free survival (PFS) in patients treated with R-CHOP but not in those treated with bendamustine-rituximab (BR).29 Other studies have shown that high Ki-67 expression is associated with disease progression within 24 months in patients with FL and correlates with poorer outcomes.30 These findings contradict those of previous studies, highlighting the complexity and potential variability of Ki-67 prognostic value in different patient subsets and treatment regimens.
This study had several limitations that should be acknowledged. First, the sample size was relatively small, which may affect the generalizability of the findings, especially when analyzing Ki-67 expression across diverse NHL subtypes. Second, the assessment of Ki-67 expression relied on visual scoring by pathologists, which may be subject to inter-observer variability, particularly given the heterogeneity in staining intensity and nuclear distribution. Third, the study was conducted at a single institution.
Conclusion
There is an association between Ki-67 expression, aggressiveness, and therapeutic response in patients with non-Hodgkin lymphoma. Future studies should focus on validating these findings in larger, multicenter cohorts to improve the generalizability of Ki-67 as a prognostic and predictive marker for various non-Hodgkin lymphoma subtypes.
Acknowledgment
The authors wish to acknowledge the Institute for Research and Community Service and Dean of the Faculty of Medicine, Udayana University, for their financial support of this study.
Funding Source
This study received financial support from the Udayana University Research Grant (grant number: B/229.62/UN14.4.A/PT.01.03/2025).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The present study was approved by the Institutional Review Board of the Faculty of Medicine, Udayana University (0256/UN14.2.2.VII.14/LT/2024).
Informed Consent Statement
Participants have signed informed consent forms. No personal data has been written in the manuscript.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Ngakan Ketut Wira Suastika: Conceptualization, Data Collection, Analysis, Writing – Original Draft.
- Ketut Suega: Methodology, Writing – Review & Editing, Funding Acquisition Resources, Supervision.
References
- Chu Y, Liu Y, Fang X, et al. The epidemiological patterns of non-Hodgkin lymphoma: global estimates of disease burden, risk factors, and temporal trends. Front Oncol. 2023;13:1059914.
CrossRef - Mafra A, Laversanne M, Gospodarowicz M, et al. Global patterns of non-Hodgkin lymphoma in 2020. Int J Cancer. 2022;151(9):1474–1481
CrossRef - Farmanfarma K.K, Kiasara S.H, Hassanipour S, et al. Non-Hodgkin’s lymphoma in the world: an epidemiological review. WCRJ. 2020;7(6):e1520.
CrossRef - Zelenetz A.D, Abramson J.S, Advani R.H, et al. Non-Hodgkin’s lymphomas clinical practice guidelines in oncology. JNCCN. 2010;8(3):288-334.
- Armitage J.O, Gascoyne R.D, Lunning M.A, et al. Non-hodgkin lymphoma. The lancet. 2017;390(10091):298-310.
CrossRef - Pratap S, Scordino T.S. Molecular and cellular genetics of non-Hodgkin lymphoma: Diagnostic and prognostic implications. Exp Mol Pathol. 2019;106:44–51.
CrossRef - Juríková M, Danihel Ľ, Polák Š, et al. Ki67, PCNA, and MCM proteins: Markers of proliferation in the diagnosis of breast cancer. Acta Histochem. 2016;118(5):544–552.
CrossRef - Røge R, Nielsen Sø, Riber-Hansen R, et al. Impact of Primary Antibody Clone, Format, and Stainer Platform on Ki67 Proliferation Indices in Breast Carcinomas. Appl Immunohistochem Mol Morphol. 2019;27(10):732–739.
CrossRef - Miller I, Min M, Yang C, et al. Ki67 is a Graded Rather than a Binary Marker of Proliferation versus Quiescence. Cell Rep. 2018;24(5):1105-1112.
CrossRef - Li L.T, Jiang G, Chen Q, et al. Ki67 is a promising molecular target in the diagnosis of cancer. Mol Med Rep. 2015;11(3):1566–1572.
CrossRef - Tong G, Zhang G, Liu J, et al. Cutoff of 25% for Ki67 expression is a good classification tool for prognosis in colorectal cancer in the AJCC‑8 stratification. Oncol Rep. 2020;43(4):1187–1198.
CrossRef - Yang D, Zhou Y, Lv K. Analysis of Ki67 Protein Expression and Clinicopathological Features in Patients with Peritoneal Mesothelioma. Altern Ther Health Med. 2024;30(9):202–209
- Broyde A, Boycov O, Strenov Y, et al. Role and prognostic significance of the Ki-67 index in non-Hodgkin’s lymphoma. Am J Hematol. 2009;84(6):338–343.
CrossRef - Kreipe H. Ki67: biological intertumor variance versus variance of assay. Pathologe. 2018;39:272–727.
CrossRef - Armitage J.O. Staging Non-Hodgkin Lymphoma. CA Cancer J Clin. 2005;55(6):368–376.
CrossRef - Advani R.H, Hoppe R.T, Maeda L.S, et al. Stage I-IIA non-bulky Hodgkin’s lymphoma. Is further distinction based on prognostic factors useful? the Stanford experience. Int J Radiat Oncol Biol Phys. 2011;81(5):1374-1379.
CrossRef - Younes A, Hilden P, Coiffier B, et al. International Working Group consensus response evaluation criteria in lymphoma (RECIL 2017). Ann Oncol. 2017;28(7):1436-1447.
CrossRef - Hashmi A.A, Hussain Z.F, Faridi N, et al. Distribution of Ki67 proliferative indices among who subtypes of non-hodgkin’s lymphoma: Association with other clinical parameters. Asian Pac J Cancer Prev. 2014;15(20):8759–8763.
CrossRef - Rahimi H, Borojerdi Z.R, Azimi S.A, et al. Prognostic significance evaluation of B-cell lymphoma 2 (BCL2) and Ki-67 expression in diffuse large B-cell lymphoma patients. Immunopathol Persa. 2019;6(1):e07.
CrossRef - do Nascimento Santos T.A, da Silva A.K.F, Cunha K.S.G, et al. Current advances in cancer immunohistochemistry: a new perspective for the Ki-67 biomarker. Ecancermedicalscience. 2025;19:1863.
CrossRef - Dias E.P, Oliveira N.S.C, Serra-Campos A.O, et al. A novel evaluation method for Ki-67 immunostaining in paraffin-embedded tissues. Virchows Arch. 2021;479(1):121-131.
CrossRef - Bairey O. Role and prognostic significance of the Ki-67 index in non-Hodgkin’s lymphoma. European J Clin Med Oncol. 2010; 2(1): 1-7.
- Jakovic L.R, Mihaljevic B.S, Jovanovic M.D.P, et al. The expression of Ki-67 and Bcl-2 in Hodgkin’s lymphoma: Correlation with the international prognostic score and bulky disease: A study by the Serbian lymphoma study group (SLG). Med Oncol. 2007;24(1):45-53.
CrossRef - Li S.D, Ba Y.P, Zhang P.W. Prognosis factors analysis of 56 cases of non-Hodgkin’s lymphoma originating from the nasopharynx. J Clin Otolaryngol Head Neck Surg. 2019;33(7):651-653.
- Huber F, Zwickl-Traxler E, Pecherstorfer M, et al. Evaluation of ki-67 as a prognostic marker in diffuse large b-cell lymphoma—a single-center retrospective cohort study. Curr Oncol. 2021;28(6):4521–4529.
CrossRef - Went P, Agostinelli C, Gallamini A, et al. Marker expression in peripheral T-cell lymphoma: a proposed clinical-pathologic prognostic score. J Clin Oncol. 2006;24(16):2472-2479.
CrossRef - Skrypets T, Novosad O, Pastushenko Y, et al. Ukraine data on prognostic factors and treatment outcomes in patients with peripheral t-cell lymphomas. Klin Onkol. 2019;32(6):436-444.
CrossRef - Li Z.M, Huang J.J, Xia Y, et al. High Ki‐67 expression in diffuse large B‐cell lymphoma patients with non‐germinal center subtype indicates limited survival benefit from R‐CHOP therapy. Eur J Haematol. 2012;88(6):510-517.
CrossRef - Nichols M.M, Ondrejka S.L, Patil S, et al. Ki67 proliferation index in follicular lymphoma is associated with favorable outcome in patients treated with R-CHOP. Leuk Lymphoma. 2023;64(8):1433–1441.
CrossRef - Narita H, Kuroiwa K, Kawaguchi Y, et al. High Ki-67 Expression Predicting a Risk Factor for the Progression of Disease within 24 Months and Microenvironment in Follicular Lymphoma. Int J Mol Sci. 2024;25(20):11057.
CrossRef
Abbreviations List
AUC: under the curve
BR: bendamustine-rituximab
DLBCL: diffuse large B-cell lymphoma
FL: follicular lymphoma
IHC: immunohistochemistry
IPI: International Prognostic Index
LDH: lactate dehydrogenase
MALT: mucosa-associated lymphoid tissue
MZL: marginal zone lymphoma
NHL: Non-Hodgkin lymphoma
PFS: progression-free survival
R-CHOP: cyclophosphamide, doxorubicin, vincristine, and prednisone with rituximab
ROC: receiver operating characteristic
SGOT: Serum Glutamic Oxaloacetic Transaminase
SGPT: Serum Glutamic Pyruvic Transaminase







