Manuscript accepted on :18-08-2025
Published online on: 03-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Alaa Saadi Abbood
Second Review by: Dr. Saida Ncibi
Final Approval by: Dr. Eman Refaat Youness
Sonam Chawla* , Sanjukta Bhattacharya
, Sanjukta Bhattacharya and Aaysha Gupta
and Aaysha Gupta
Department of Biotechnology, Jaypee Institute of Information Technology (Main Campus), Noida, Uttar Pradesh, India.
Corresponding Author E-mail:contact.schawla@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3214
Abstract
Senescence is a hallmark of the natural ageing process across species. It is an irreversible arrest of the cells in a non-dividing state, restricting completion of cell cycle. Increased number of senescent cells is associated with declining health span. The switching between a normal cell to a senescent cell is governed by diverse factors, such as activation of DNA damage response, telomere attrition, raised redox imbalance etc. The senescent cells are detected through increased molecular markers of cell cycle arrest, chromatin remodeling indicators, lipofuscin, increased autophagic flux, and increase activity of senescence-associated β-galactosidase. A hyper secretory inflammatory response referred as Senescence-Associated Secretory Phenotype (SASP) is also attained. SASP contributes to low grade chronic inflammation in elderly and contributes to pathophysiology of most geriatric diseases. Cellular senescence can be managed by inclusion of dietary entities that can clear senescent cells (senolytic action), rein the SASP response (senomorphic action) or facilitate re-entry to cell cycle (senoreversal). This scoping review presents the current understanding of cellular senescence activation and detection and compiles the findings from studies wherein dietary components - bioactive polysacchrides, peptides and proteins, fatty acids, lipids and probiotic formulations, that are reported to provide nutrition as well as confer an anti-senescent advantage.
Keywords
Anti- aging diet; Senescence-associated secretory phenotype; Senescent cells; Senolytics; Senoreversal agents; Senotherapeutics
Download this article as:| Copy the following to cite this article: Chawla S, Bhattacharya S, Gupta A. Understanding Cellular Senescence and Senotherapeutics for Nutri-geriatric Solutions. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Chawla S, Bhattacharya S, Gupta A. Understanding Cellular Senescence and Senotherapeutics for Nutri-geriatric Solutions. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4ngeg5W |
Introduction
The natural process of aging is marked by a slow deterioration of body functioning over time. 2 Physiologically, senescence has essential functions in tumor suppression, embryonic differentiation and development, wound healing and tissue regeneration, etc, but its role in onset of age-related diseases cannot be overlooked.3 In fact, majority of age-related illness report a buildup of senescent cells with aberrant DNA, and a distinctive shift in the secretory composition known as – SASP, favoring inflammation associated secretory proteins, enzymes and signaling factors.4 Researchers are yet trying to decipher the dynamics between aging and senescence, with short-term buildup of senescent cells often being beneficial but long-term accumulation associated with disease incidence (Figure 1). An emerging category of pharmacological interventions targeting the pathological outcome of senescent cells are senotherapeutics. Senotherapeutics can be senomorphic (inhibiting the SASP responses), senolytics (selective elimination of senescent cells from diseases/aged tissue) or senoreversal agents that intend to switch back the “senescent” state to original state.5 In 2019 the first successful clinical trial of dastanib and quercitin combination was published, wherein subjects with idiopathic pulmonary fibrosis exhibited improvement in physical performance after 9 doses of the combination over 3 weeks.6 Since then several new senotherapeutic molecules have been tested in pre-clinical and clinical studies, the most recent being a phase one study of oral administration of dasatinib and that of quercetin in symptomatic patients with AD to evaluate the penetrance, safety and pharmacological efficiency. Notably, a decline in the senescence-related cytokines and chemokines as well as a raised level of Aβ42 in cerebrospinal fluid.7 Understanding the role in cellular senescence in human physiology and diseases can aid the discovery and development of such novel senotherapeutics which can potentially ameliorate chronic diseases and extending lifespan/healthspan. The main objective of the present narrative review is to elucidate and discuss the present knowledge underscoring the molecular/environmental triggers of senescence in cells as we age, the SASP phenotype and the current technologies available to detect senescent cells. Hereafter we have compiled the current knowledge of various types of senotherapeutics based on differences in their ability to modulate the senescent state. A larger focus of the discussion are dietary senotherapeutics – namely carbohydrates, proteins, fats and lipids and small molecules of dietary origin, that have demonstrated pre-clinical and/or clinical benefits against the senescent cells buildup and pathological outcomes.
The main objective of this review is to elucidate and discuss the present knowledge underscoring the molecular/environmental triggers of senescence in cells as we age, as well as offer nutrigeriatric solutions via highlighting the benefits of dietary senotherapeutics.
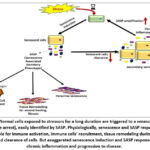 |
Figure 1: Normal cells exposed to stressors for a long duration are triggered to a senescent state (cell cycle arrest), easily identified by SASP. Physiologically, senescence and SASP response are responsible for immune activation, |
Triggers of Senescence
The phenomena of cellular senescence were initially reported in middle of 20th century by Hayflick and Moorhead as a proliferation limit in dividing somatic cells (normal human fetal fibroblasts) after multiple passaging and was called “replicative senescence” or “Hayflick limit”.8 Table 1 summarizes the differences between .
Table 1: Difference between senescent cells and quiescent cells 8
| FEATURES | SENESCENT CELLS | QUIESCENT CELLS |
| Cell cycle status | Mostly in the cell cycle’s G1 or G2 phases | During the Go stage of the cell cycle |
| Triggering factors | Aging, DNA damage mutagens | Nutrient or growth factor deprivation |
| Reversibility | Reversible | Irreversible |
| Morphology | Enlarged and flattened | Retains normal cell morphology |
| Function | Metabolically active but do not proliferate | In a state of rest but viable |
| Chromatin changes | Forms dense heterochromatin bodies | Remain same |
| Cell marker | Increased expression of p16INK4a, p21. Altered morphology and lysosomal activity | Reduced DNA synthesis and metabolic activity, low expression of proliferation markers. |
| Role | Tissue repairs immune response but aging and age-related diseases. | Cells are ready to respond to signals and lead tissue regeneration. |
The transit from normal cells into senescent phenotypic cells can be induced by one or several of the following molecular events 9:
Telomere Attrition
Is a hallmark for replicative senescence, wherein the shelterin protein complex encasing the repetitive DNA sequence, located on chromosome termini progressively shorten with cell division, correlating with increasing number of cells switching to senescent state.10 Interestingly ectopic expression of reverse transcriptase responsible for elongation of telomeres (telomerase enzyme) can counteract telomere shortening associated with progressive cell divisions and also bypass the switch to senescence. Shelterin complex consist of six proteins – TRF1, TRF2, POT1, RAP1, TIN2 and TPP1. The shelterin complex shelters the telomere-loop (t-loop)In face of “end-replication problem” faced in telomere elongation with progressive cell divisions the components of shelterin complex destabilize and expose the t-loop eventually detected as a DSB, and triggers the DDR.11
Activation of DNA Damage Response
Oxidative Stress
It is an outcome of dysregulation in free radicals generation and the defenses of inherent cellular antioxidant. Oxidative stress is in fact theorized as an active contributor to cellular ageing and is theorized in the Oxidative Damage Theory.17 ROS is primarily produced due to mitochondrial respiration, as a byproduct of ATP synthesis as oxygen can be partially reduced, and its derivatives such as superoxide anion etc. Excessive ROS, coupled with failing detox mechanisms, can damage cellular structural components (proteins, plasma membrane, carbohydrates) as well as functional multi-subunit structures – nucleic acids, enzymes etc.18 This ROS-induced damage can trigger DDR by damaging mitochondrial DNA (mtDNA) leading to mitochondrial dysfunction which in turn can increase ROS production. This mtDNA damage in concert with decreased TERT activity and Ras, p53, p21, and p16 pathways can trigger the senescence phenotype. Additionally, ROS mediated p53 activation can also inhibit autophagy in senescent cells further aggravating the turnover of proteins and damaged organelles. These senescent cells then accumulate oxidized or carbonylated proteins along with lipid peroxidation products, and glycated protein products (lipofuscin). Interestingly ROS also induces production of miR-210 and miR-494, which can in turn inhibit autophagy and aggravate senescent phenotype.19,20 Furthermore, redox imbalance in conjunction with mitochondrial functional disturbances can induced a pro-inflammation outset via activation of NF-κB signaling pathway, NLRP3 inflammasome, cGAS–STING pathway indicating at the close link between cellular senescence and inflammaging.21 Another interesting link between failing oxidative defenses and increased senescent cell population is the decreased signaling via transcription factor Nrf2 a primary gatekeeper of cellular redox balance.22 Thus, the buildup of ROS and its damage by-products, along with impaired antioxidant defenses and mitochondrial dysfunction, can initiate cellular stress responses that ultimately lead to cellular senescence.
Detection of Senescent Cells
Senescence is characterized by alterations in the cellular structural components, epigenetics and signaling pathways. A fail-safe recommendation to detect senescence in tissues is confirmation of at least three indicators giving a positive test – indicators of arrest of cell cycle progression, structural alterations such as increased vacuoles/lysosomal vesicles, multinucleation, senescence-associated heterochromatic foci, flat appearance of cell and loss of lamin B1, indicators of DNA damage, redox imbalance, upregulated senescence associated secretory phenotype markers usually associated with inflammation such as cytokines or chemokines.23,24
Markers of cell cycle arrest
Primary signaling cascades initiated by endogenous and exogenous stressors discussed above activate the p53/p21- and/or the p16/Rb pathway. The p53 pathway is activated during DDR, replicative senescence and during cellular milieu of redox imbalance, early in the cell cycle. The p16 pathway ensues during DDR response and replicative senescence, and is more often associated with maintenance of the senescent phenotype. 25,26 In either pathway, the final convergent point is Rb. Rb inactivation is critical for G1/S transition during the cell cycle via hyperphosphorylation, preventing complexing with the E2F and disrupting the Rb-E2F responsive genes’ expression via transcriptional repression. This facilitates expression of genes needed for cell cycle progression e.g.cyclin E, Proliferating Cell Nuclear Antigen. On the other hand, in senescent cells, the hypo-phosphorylated Rb binds E2F preventing the transcription of replicative genes and hence halting the cell cycle and hence arresting the cells in senescence.27,28
These signaling markers can be detected with help of robust primers, probes, and antibodies, commercially available across commercial vendors. A key technical issue to be highlighted here is the gene expression studies of signaling mediators at transcriptional level (mRNA) or protein (western blot) level, uses constitutively expressed gene as control for quantitative analysis of gene expression. However, the expression of routinely used housekeeping genes is altered which can compromise the normalization of gene expression. Notable research backed housekeeping genes for use in senescence studies are – Tubulin Alpha 1a, Vinculin, L(3)Mbt-Like protein 2, Polycomb repressive complex1 subunit etc.28
In addition to molecular markers, indicators of cell cycle progression such as cellular proliferation or DNA replication assessment can also aid detection of senescence. Temporal assessment of cell number counts spectrophotometrically, or via automated cell counters and live cell microscopy can be used for detecting the cellular proliferation. Additionally, arrest in DNA synthesis can be detected via reduced incorporation of reagents like bromodeoxyuridine during the DNA replication in synthetic phase. Proliferation marker – nuclear protein Ki67 levels can also be measured via gene expression analysis or immunostaining of cells. During the S, G2, and M phases Ki-67 accumulates in the nucleus, while during G1 and G0 phases Ki-67 levels continuously degrade.29
SAHF biogenesis
SAHF are localized stretches of facultative heterochromatin that silence the expression of “progression of cell cycle” genes and
Autophagy
Autophagy is a highly conserved and crucial process for cellular maintenance and clearance of senescent cells or compromised biomolecules and organelles, and eventually ageing. Errant autophagic pathways are a recognized hallmark of ageing.33
Macroautophagy is the principle autophagic pathway essential wherein cellular components (misfolded proteins, damaged organelles – mitochondria, in that case it is called mitophagy), and invading pathogens, are sequestered in autophagosomes. The autophagosomes merge with lysosomes, and the enclosed cellular components are degraded by the hydrolytic enzymes.34, 35 Autophagy and cellular senescence are interconnected across multiple signalling pathways and participant proteins.36 Autophagic activity declines across multiple tissues during aging, for example in macrophages and other immune cells. This decline in autophagy is considered a hallmark of cellular senescence. Compromised autophagic pathways further pushes the aggregation of cellular damage, for example; aggregosomes, non-functional mitochondria, which contribute to the senescence process.
Furthermore, autophagy is closely linked to prevention of stem cell exhaustion, oncogenic transformation etc. The decline in autophagic activity during aging can contribute to these processes, exacerbating cellular senescence.34,36 Additionally, autophagy is involved in reducing inflammatory responses, and its impairment during aging can lead to increased inflammation, another hallmark of cellular senescence. Interestingly, during aging, autophagosomes may not be able to fuse with lysosomes and build-up within the cell, which can serve as a marker of senescence. This accumulation can be monitored using various techniques, such as measuring the levels of the autophagy marker LC3-II (lipidated form of LC3) using microscopy, Western blotting, or flow cytometry. Flow cytometry with saponin permeabilization allows selective detection of membrane-bound LC3-II in immune cells.37 Autophagic flux can be assessed by blocking this fusion with inhibitors like chloroquine or bafilomycin A1. This leads to the raised LC3-II levels, which is quantified over time. Additionally, tracer dyes (Cyto-ID) can be used to detect intracellular autophagic flux using flow cytometry.37 Chaperone-mediated autophagy is the selective degradation of proteins containing a specific motif (KFERQ-like sequences) by lysosomes. CMA activity also declines with aging, impacting processes like T cell receptor (TCR) responses. CMA activity can be monitored using transgenic fluorescent protein reporters tagged with KFERQ sequences or by immunostaining for the CMA-associated proteins HSC70 and LAMP2A.38 Several anti-aging interventions, such as calorie restriction or pharmacological interventions, converge on the induction of autophagy, suggesting that maintaining autophagic activity can delay cellular senescence and aging processes.
SA-βgal staining
Detection of β-galactosidase enzyme activity at acidic pH (pH 6.0), along with p16INK4a expression, is widely used as a biomarker for cellular senescence. While usually, β-galactosidase activity is localized to lysosomes and is active up to pH 4.5, senescent cells exhibit an accumulation of this enzyme, leading to activity being detected even at suboptimal pH of 6.0, and is classified as SA β-gal activity.39
The mechanistic action of SA-β-gal activity is attributed to escalate lysosomal biogenesis and altered composition during senescence, allowing β-galactosidase detection at non-acidic pH 6.0 in senescent cells. However, SA-β-gal as a senescence marker, is not entirely specific, as high cell confluency or serum starvation can also induce this activity independent of senescence.39 Enhanced SA-β-gal is also closely linked with SASP.
Detection methods include histochemical staining on fixed tissues or flow cytometry with fluorogenic substrates like C12FDG. Detecting SA-β-gal (pH 6.0) requires lysosomal alkalinization, which may disrupt cellular physiology or necessitate fixation for sensitive cells. Due to its limitations, SA-β-gal in association with other senescence markers such as; p16INK4a, p21, SAHF, SASP, and SA-DDR assays should be used to ascertain the status of senescence in cells. In summary, SA-β-gal at pH 6.0 is the most rampantly cited senescence biomarker but should be interpreted alongside additional markers and functional assays.40
Lipofuscin detection
Lipofuscin of the ageing pigment is a yellow-brown complex accumulating in the lysosomes over the lifespan of post-mitotic cells. The composition includes oxidized cellular biomolecules and metal ions (iron, copper, zinc etc). Lipofuscin is resistant to proteolytic degradation due to its cross-linked structure and is also unaffected by exocytosis. Lipofuscin accumulation, a biological marker of replicative and γ-irradiation-induced senescence, and is highly colocalized SA-β-Gal activity. Although lipofuscin has natural fluorescence albeit with a wide emission spectrum (400-700nm), interfering with localization of the pigment. More reliable methods include use of Sudan black B dye (detecting lipids in lipofuscin), and periodic acid Schiff staining (detecting the carbohydrates)41. Additionally, chemical conjugates of Sudan Black B dye are also reported for more sensitive detection via microscopy/flowcytometry, for example GLF16 is a modified Sudan-Black-B analog with fluorescent properties recently reported by Magkouta and coworkers, enabling detection of senescence using microscopy or cytometry.42
Table 2: Biomarkers and detection methods of cellular senescence26
| Identifying Trait of senescent cells | Marker | Detection Method |
| Blocked DNA synthesis | BrdU, EdU | Staining incorporation, IF |
| No proliferation | Ki67 | Immunohistochemistry, Immunofluorosence |
| Triggered p16-pRB axis | p16INK4a | Western Blot, Immunohistochemistry, Immunofluorosence |
| Triggered p53-p21 axis | P21 | Western Blot, Immunohistochemistry, Immunofluorosence |
| DDR | γH2AX | Immunofluorosence |
| SAHF | DAPI/Hoechst 33342 | Immunofluorosence |
| Telomere end shortening | Telomere | qPCR, Fluorescence in situ hybridization |
| ROS formation | Free radicals | Chemiluminescent oxygen detection reagents, fluorometry and flow cytometry |
The Sasp Response: Secretory Signatures
The SASP response is a hypersecretory phenotype in cells that have shifted to a senescent state, with secretions having autocrine as well as long distance paracrine impact. Analysis of the senescent cells’ secretome unravels a dynamic composition of both soluble or insoluble and extracellular vesicle-related components. SASP response can exaggerate or diminish the absolute levels of these secreted molecular signals and contrasts significantly from the actively proliferating cells. Senescence messaging secretome is another name for SASP. The secretome can be triggered by stress, aging, DNA damage etc. The secretome consists of inflammatory cytokines, immune modulators (molecules that influence immune cell behavior), growth factors (proteins that regulate cell growth and repair), and proteases (enzymes that break down proteins) essentially culminating in a pro-inflammatory, pro-apoptotic, and pro-fibrotic milieu. Some common SASP factors common across cell types and different types of senescent inducing mechanisms (irradiation induced, UV-B induced, glyoxal induced) are IL-6, TNF-α, filamin B, cathepsin D, macrophage migration inhibitory factor, IL-7, IL-8.23,24,43 The autocrine action of SASP reinforces the cell cycle arrest in the cellular self and the paracrine action works on neighboring cells. Physiologically SASP has a pivotal role in signaling for immune cells (natural killer cells or macrophages) to scavenge senescent cells. Additionally, SASP also operates during mammalian development, signaling for cellular-fate reprogramming. In adult life, SASP is also recognized to act in tissue repair for e.g. skin, liver injury etc.23,44
The regulation and variability of SASP are governed by multiple mechanisms at the transcriptional, translational, mRNA stability, and secretion levels, relying on positive feedback loops for signal amplification. Interestingly, consistent stimulation of DDR signaling is crucial for maintaining the senescent milieu and driving towards a SASP outset, whereas transient DDR is insufficient. Key transcriptional regulators of SASP expression include NF-κB and C/EBPβ. The DDR activates NF-κB via post-translational modifications of NEMO by the ATM kinase, leading to the transcription of SASP-related genes. cGAS, a cytosolic DNA sensor, connects DDR to SASP initiation by activating the STING-IRF3/NF-κB pathway in response to cytoplasmic DNA, often originating from micronuclei.45 Moreover, MAPKs, especially p38, play a critical role in maintaining cell cycle arrest and activating NF-κB to promote SASP development. RBPs such as AUF1, HuR, and hnRNPA1 also significantly regulate SASP factor expression by influencing mRNA stability and translation.46 Other factors, including the scavenger receptor CD36, transcription factor GATA4, histone modifications, and various non-coding RNAs, houses the intricate regulation of multiple levels SASP.
The variability in the SASP response is influenced by the metabolic capacity of senescent cells. Notably, a dysregulation in the mitochondrial functional capacity is imperative for inducing senescence, induction of SASP outset and other senescence related cellular disturbances. For instance, depleting mitochondrial sirtuins such as SIRT3 and SIRT5 can trigger senescence and reduce the secretion of pro-inflammatory factors such as interleukin 1β, vascular endothelial growth factor etc in radiation-induced senescent human fibroblasts.47 Mitochondrial dysfunction has a unique SASP profile that overlaps with but is also distinct from conventional SASP induced triggers as discussed above, and is known as Mitochondrial Dysfunction-Associated Senescence. This offshoot of the senescence phenomena is driven by a lowered NAD+/NADH ratio, which negatively impacts NF-κB activity through the AMPK-p53 axis. Consequently, SIRT3 and SIRT5 emerge as potential targets for modulating the pro-inflammatory SASP. Furthermore, a decline in nicotinamide adenine dinucleotide levels could impair functioning of the PARP-1, an enzyme crucial for maintenance of mitochondrial genomic integrity that requires this cofactor. The PARP-1/NF-κB signaling pathway also promotes a tumor formation and metastasis associated with SASP in xenograft models, although the role of NAD+ levels in this context requires further investigation. 48 The diversity of the SASP response has been cataloged in SASP Atlas, an online comprehensive temporal and spatial proteomic database of SASP secretome, derived from different human primary cell types and triggers of senescence induction (genotoxic stress-induced, oncogene-induced, Atazanivir treatment). The platform can facilitate identification of distinct markers as well as markers elevated in all SASP responses.49
Senotherapeutics
The Sharpless group at School of Medicine, University of North Carolina pioneered the search for senotherapeutic agents in 2004.50,51 They demonstrated a higher burden of senescent cells accompanied by raised expression of p16INK4a and Arf in vivo with ageing in rats. The pathology onset, senescent cell burden and expression of p16INK4a and Arf decreased with calories restriction (established anti-aging regime for lifespan extension in pre-clinical and clinical setting). By 2005, the team was working on two strategies to address the cellular burden of senescent cells, first involved creating fusion proteins with toxins to specifically destroy senescent cells. The second strategy screened human cell in dividing state vs. senescent state, for discovering chemicals that selectively kill senescent cells bypassing non-senescent ones. Thus, senotherapeutics emerged as a class of small molecules that specifically target only the senescent cell and their pathological molecular responses. As mentioned earlier senotherapeutics can be categorized as senomorphic, senolytics or senoreversal agents.5,52 We briefly discuss the respective functional categories of senotherapeutics in the following sections, and also discuss the human dietary and natural sources of senotherapeutics:-
Senomorphic agents
Senomorphics are a class of molecules designed to modify the pathological molecular signatures of senescent cells without inducing their death. Instead of eliminating these cells, senomorphics specifically target the SASP. By blocking or inhibiting the SASP components, senomorphics help mitigate inflammation and other detrimental processes associated with aging and tissue dysfunction. The known senomorphics target pathways downstream of p38MAPK, PI3k/Akt, mTOR, and JAK/STAT pathways, or through the transcriptional circuits of factors NF-κB, STAT3 etc. Indirect antibody mediated neutralization of cytokines/chemokines interleukin 1α, interleukin 8, interleukin 6 has also been noted.23 Senomorphics hold promise for treating age-related diseases, especially with an underlying component of inflammaging such as cardiovascular diseases and neurodegenerative disorders.53 Dietary bioactive molecules known to exhibit senomorphic action are Resveratrol, Kaempferol, Apigenin, and Epigallocatechin gallate, rutin, parthenolide, phloretin and curcumin. Synthetic senomorphic molecules include metformin, loperamide, cortisol among others.54
Senolytics
Senolytics are small molecules that specifically eliminates cells in the senescent state and hence reducing their accumulation during ageing and related diseases. Known senolytics work through varied mechanisms, but a unifying across mechanisms is exploiting the vulnerabilities of senescent cells, to which a normal cell is resistant or may cope with the pharmacological perturbation. The senolytic mechanisms include targeting the senescent cell’s anti-apototic response (SCAP). Senescent cells develop resistance to apoptosis and have un/up-regulated pro-survival signals such as ephrins, PI3K etc. Senolytics can exploit this characteristic by precisely blocking the viability related signaling pathways in senescent cells, making them susceptible to apoptosis. The first line of senolytics discovered – dasatinib and quercetin, targeted the protective pro-survival signals in senescent cells.55 Additional interventional molecules can be targeted to inhibit the anti-apoptotic molecular mediators through p53 (UBX0101), Bcl-2 (ABT-263 also known as navitoclax), Akt (alvespimycin and fisetin), PI3K (fisetin), FOXO4 (FOXO4-DRI) etc. Mechanisms routed through inhibition of Hsp 90 in oxidative stress induced senescence, as well as targeted delivery (to raised β-galactosidase activity in senescent cells) of galactose derivatives of cytotoxic agents (galactose-modified duocarmycin pro-drug), also are reported.5
Senoreversal agents
It is an advance approach that reprograms the senescent cells to reinstate the original dividing state or reversing the arrest on the cell cycle. The earliest reports on senoreversal came from An and co-workers wherein in silico prediction based on molecular regulatory network of cellular senescence, PDK-1 inhibition was tested in human dermal fibroblasts using pharmacological inhibitors. The inhibition of PDK1 suppressed mTOR and nuclear factor κB, alleviating the proliferative arrest and SA-β-gal–positive cell population declined from >80% to <40%. Clinical evidence for PDK1 inhibition using kaempferol tetrasaccharide a natural senotherapeutic was put forth by Kim and co-workers.56,57 A groundbreaking study conducted by Bi and co-workers recently, reported the senoreversal capacity of human embryonic stem cell-derived exosomes, restoring the senescent cells proliferation ability, largely due microRNA-302b (enriched in human embryonic stem cell-derived exosomes) in vitro as well as in aging mice.58 Senoreversal molecules are known in limited capacity and a larger focus is on senomorphics senolytics. In fact in light of the heterogenous nature of senescent cells and triggers thereof, senotherapeutic molecules have been shown to have dual action of senolysis as well as senomorphic.55 Also, treatment regimens with senolytics are aggressive, whereas senopmorphic agents merely suppress the pathological SASP, and are deemed safer in long term studies.59
Dietary Sources of Senotherapeutics
Dasatinib and quercetin cocktail is the most clinically evaluated senotherapeutic intervention, but not without adverse effects. In a pilot trial of dasatinib and quercetin treatment (100mg/1250 mg) regime for idiopathic pulmonary fibrosis, a higher incidence of non-serious adverse events (cough, shortening of breath, nausea, general feeling of fatigue, weakness and sleep disturbance, depressive behavior etc) was reported against the placebo group.60 Additionally, the thrombocytopenia, and minor steatosis were also reported in other studies.61 The human dietary components – carbohydrates, proteins, fats and lipids, vitamins, small molecules of plant/animal origin , and probiotics, have stand alone or in concerted action have been demonstrated to modulate the burden of cellular senescence.62 We herein discuss the dietary senotherapeutics that can be easily, safely and cost-effectively included in geriatric diet regimes, offering long term benefits in preserving health span alongside lifespan [Figure 2].
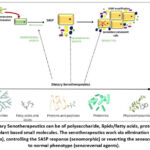 |
Figure 2: Dietary Senotherapeutics can be of polysaccharide, lipids/fatty acids, protein extracts, probiotics or plant based small molecules. |
Carbohydrates and derivatives as senotherapeutics
Carbohydrates are the foremost energy source. Excess consumption of carbohydrates and deranged metabolism/utilization of carbohydrates is positively linked with incidence of chronic disorders – obesity, insulin resistance progressing to diabetes.63 Notably, diets enriched with glucose/fructose are used to simulate animal models of accelerated ageing and at the same time restriction in calories originating from reduced carbohydrate intake improves metabolic risk markers of obesity, and metabolic syndrome.64
Nevertheless, complex carbohydrates and derivatives have demonstrated a positive impact on reigning cellular senescence at molecular level and may aid expansion of healthspan. Several research groups reported polysacchrides derived from Astragalus membranaceus, also known as Mongolian milkvetch, aids recovery of hepatocyte senescence via phosphorylation of AMPK and blockade of mTOR pathway in L02, Huh7, and LM3 cell lines as well as 15-month-old female C57BL/6 mice. Astragalus membranaceus polysacchrides when tested in rat aortic endothelial cells treated with high glucose, significant decrease was observed in the cell count positive for SA-β-Gal activity, expression of biomarkers p16, p21, and p53 and inflammasome activation.65 Angelica sinensis, a prominent herb in traditional Chinese medicine, is the source of angelica polysaccharide. The Angelica polysacchride has demonstrated anti-cellular senescence and potent antioxidant properties across cellular models – haematopoietic cells, endothelial progenitor cells, and in murine animal model of chemically induced accelerated aging. The Angelica polysacchride, administered to mice for 42 days via intraperitoneal route, alleviated redox imbalance, decreased advanced glycation end products’ titre systemically, decreased count of SA-β-Gal positive cells as well the expression of γ-H2A.X culminating via the Wnt/β-catenin signaling pathway.66,67 Polysaccharides derived from Lycium barbarum, Bletilla striata, and Rehmannia glutinous were demonstrated to control the onset of senescence in C. elegans via DAF-16 transcriptional activation downstream of insulin/insukin like growth factor signaling pathway-1.68,69 Another interesting source of anti-senesence polysacchrides are marine fucoidan, demonstrated to rescue p-cresol mediated shift to senescence in stem cells (p-cresol is a uremic toxin responsible to kidney damage) can reinstate endothelial cells from cellular senescence, and enhance their survival.70 Concurrently, in light of the emerging anti-senescent activity of poly-sacchrides and carbohydrates, an inclusion of the discussed bioactive polysaccharides alone or their source herb in geriatric dietary regime could prove beneficial, and should be pursued for innovations in nutraceuticals.
Dietary proteins and peptides as senotherapeutics
Proteins are largely associated with structural, signaling and catalytic activity across prokaryotes and eukaryotes. For a healthy human adult 2 g per kg body weight of protein consumption is recommended. Proteins in combination with carbohydrates form the major portion of human diet, and hence the recommended carbohydrate to protein ratio for heathy ageing is 10:1.71 At the cellular level, imbalance in proteostasis (homeostatic mechanisms to maintain a balanced and functional proteome) is directly linked to onset of senescence and decline in lifespan of the cell/organism. Notably, protein homeostasis is compromised in senescent cells and this aiding the re-establishment of Proteostasis, critical to offset senescence.72,73 Categorically, several peptide and protein hydrolystaes have been now reported to have senotherapeutic potential. Notably protein hydrolyzate from Angelica sinensis led to identification of A. sinensis peptides (AsiPeps) <3kDa and <20 amino acids in length, reduced the systemic reactive oxygen species levels, raised enzymatic antioxidant defences and controlled oxidative damage to structural lipids in C. elegans worms undergoing paraquat induced senescence, eventually culminating into increased lifespan.74 Another recent example is of a 13-amino acid long peptide of soybean origin – Soymetide (MITLAIPVNKPGR). Soymetide has been demonstrated to delimit the senescence associated markers (p53, p21 and p16) as well as inflammatory chemokines and cytokines in the brain tissue of doxorubicin-induced senescent mice C57BL/6 male mice via the Wnt/β-catenin signaling pathway. The senotherapeutic efficacy of soymetide was at par with that of standard senolytic combination of dasatinib and quercetin, and was effective in preserving the cognitive outcomes.75 Walnut derived protein hydrolysates and their low-molecular-weight components has shown senomorphic potential in AD mice model, notable outcomes depicted were balance pro-inflammatory factors (cytokines and chemokines), autophagic flux, and redox imbalance.76 A dipeptide identified in alkalase potato-protein hydrolysates administration in combination with moderate training in old high fat diet induced SAMP8 mouse model could decrease the burden of senescent cells and improve physiological functioning via the pAMPK/ SIRT1/ PGC-1α/ pFOXO3 pathway.77 Senomorphic action has also been observed in peptides P1 (VLVLDTDYKK) and P2 (VGINYWLAHK) derived from whey hydrolysates in a model of oxidative stress induced senescence in human dermal fibroblasts.78 However, contradictory studies also exist wherein protein-rich diets lead to declined systemic NAD+ levels, raised SASP and hence accelerated senescence in animals and in humans.79,80 Together, the conflict of high protein intake vs low protein intake must be assessed on the potential impact on senesence triggering mechanisms, and the identified bioactive senotherapeutic peptides can be carefully combined with bioactive carbohydrates to confer a multipronged benefit.
Fats and lipids as senotherapeutics
Fatty acids, especially, essential fatty acids – omega-3 and omega-6 fatty acids, are recognized to have critical role in healthy aging.81 Daily inclusion of these essential lipids in geriatric diet is proven to benefit in inflammaging, sarcopenia and osteopenia, type II diabetes, cardio-vascular health and most other age related illness.82 As modulators of senescence the role of lipids and fatty acids is only emerging now, with links indicated with triacylglycerol and diacylglycerol, phospholipids, poly- and mono-unsaturated fatty acids as well as sphingolipids.83 Investigation on sphingolipids dominate the literature interlinking lipids’ role in regulating senescence across cell types. Ceramide has been linked to induction of growth arrest and replicative senescence in cell types – bone derived mesenchymal stem cells, endothelial cells.84
S1P, the functional adversary of ceramide,85 is a pro-proliferation lipid and intracellularly decreased S1P levels are associated with accelerated senescence. However, co-treatment of cells with S1P and fumonisin B1 (decreases intracellular ceramide levels by inhibiting ceramide synthase) can reverse the signalling switch to senescent phenotype.86 Additionally, S1P binding to human telomerase reverse transcriptase can mimic the enzyme’s phosphorylation mediated stabilization and hence control the senescence-associated-telomere-damages.87 Conversely, S1P signaling via S1P receptor increases pro-senescent signaling in endothelial cells accompanied with a pro-inflammatory chemokines/cytokines and lipid mediators in ECs SASP response.68 Overall, senescent cells have a deranged lipid metabolism globally, remodeling the cellular membranes and triggering the SASP phenotype, as studied by Lizardo and co-workers in Senescent BJ fibroblasts, with a predominance of nineteen polyunsaturated fatty acids triacyl glycerols moieties. The response was governed by a CD36-mediated fatty acid uptake, being diverted to glycerolipid synthesis as a means to cope with lipotoxicity associated with replicative senescence.88
A groundbreaking observation was also reported by Fafián-Labora and co-workers during their investigations in mouse hepatic stellate cells and human primary fibroblasts, and in aged mouse hepatic tissues, the fatty acid synthase activity increases upon onset of senescence.89 And hence several clinical and preclinical studies endorse dietary inclusion of omega 3/6 fatty acids in geriatric diets leading to favorable outcomes. For instance, Chan and co-workers exhibited that consumption of n-3 polyunsaturated fatty acids, of marine origin, in subjects who had undergone renal transplant, controlled the triggering of cellular senescence, and coincident SASP response. Consumption of marine n-3 polyunsaturated fatty acids, in comparison to consumption of olive oil as placebo, for forty four weeks controlled systemic levels of IL-1α, MIP-1α, MMP-1, MMP-13 amongst other inflammatory etc, and hence aiding in successful recovery of transplant patients.90 The anti-senescence effects of marine docosahexaenoic acid and eicosapentaenoic acid have also been identified and represented in cohort of patients with coronary artery disease, wherein a decrease in replicative senescence was observed in leukocytes via preservation of telomere ends.91 This phenomena was recapitulated in vitro in endothelial cells wherein treatment with docosahexaenoic acid and eicosapentaenoic acid (from fish oil), attenuated DDR [γ-H2AX foci formation decreased by nearly 50%] linked with cellular senescence and augmented the inherent anti-oxidant defences via NRF2.92 Conclusively, lipids and fatty acids are only emerging as modulators of senescent pathways and detailed understanding of lipidomic responses of senescent cells will aid our understanding to use lipid based senolytic interventions that can be a part of geriatric diet.
Probiotics as senotherapeutics
Consumption of probiotics has beneficial effects across human health and disease pathology – probiotics can regulate immune effector responses, nutrition uptake from gut and its metabolism, brain development and cognition, progression and success of therapy in cancer, inflammatory bowel disease, Alzheimer’s, diabetes etc.93 As the ingested dietary components come in contact first with the gut microbiota and are substrates for several microbe mediated biotransformation, the composition of beneficial microbiota in the gut can very efficiently modulate the molecular and hence physiological outcomes of dietary molecules.92 The ageing population’s gut microbiome signatures indicate a loss of microbial diversity, linked to age-related decline in digestion and metabolic capacity, immunity, and general physiological functioning, potentially impacting healthspan as well as lifespan of an individual.94 Dysbiotic gut, a frequent occurrence in aged populations, is an inflammatory microenvironment that can exaggerate the SASP response, as demonstrated in a mouse model of dysbiosis. The animals had raised levels of deoxycholic acid, exacerbating the SASP response in hepatic stellate cells, further exaggerating the systemic inflammation and development of hepatocellular carcinoma.95 Thus, probiotic formulations as a part of geriatric diet can be positioned as senomorphics due to their ability to modulate the SASP and reduce systemic inflammation. For instance, the study by Jeong and co-workers exhibited the efficacy of administration of probiotic Lactobacillus strain, to aged rats for eight weeks significantly controlling the inflammaging response, systemic oxidative stress and controlled the incidence of age-related colitis.96 Fang and co-workers developed a probiotic formulation of Lactobacillus and Bifidobacterium species derived from centenarian population. The formulation when administered to the SAMP 8 mouse model exhibited positive outcomes in cognitive and behavioral performance, neuroinflammation, gut inflammation, and composition of the gut microbiota.97 All together, these finding endorse the discovery and efficacy testing of “gerobiotics” for inclusion in either geriatric dietary regimes or as nutraceutical formulations to be used in supplementation with wholesome anti-senescent diets.
Small molecules and phytocompounds as senotherapeutics
A large volume of literature exists wherein plant derived small molecules from diverse classes – polyphenols, saponins, alkaloids etc have been reported to have senomorphic or senolytic activity. The authors intended to cover the dietary constituent other than bioactive phytocompounds and hence the readership can refer to detailed reviews on the theme elsewhere.5,59 Table 1 however reports a compilation of Indian plants and herbs where the much-investigated senolytic/senomorphic compound is a part of the composition. These Indian plants can be included in a bioprospecting study to unveil novel senotherapeutic compounds.
Table 3: Phytocompounds and small molecules of plant origin with established senolytic/senomorphic activity and Indian Medicinal/Dietary Plant sources [https://neist.res.in/osadhi/index.html]
| Senolytic/senomorphic phytocompound | Indian Plants |
| Quercetin | Abelmoschus esculentus |
| Acer pictum | |
| Barleria dichotoma | |
| Daucus sativus | |
| Holigarna arnottiana | |
| Naringenin | Acacia farnesiana |
| Mazus pumilus | |
| Thespesia populinea | |
| Gallic acids | Abrus precatorius |
| Alocasia indica | |
| Eucalyptus hybrida | |
| Jurinea dolomiaea | |
| Rhodiola sacra | |
| Ferulic acids | Actaea cimicifuga |
| Apigenin | Acer laevigatum |
| Barleria cristata | |
| Colocasia esculenta | |
| Hedysarum triflorum | |
| Salvia viridis | |
| Genistein | Albizia procera |
| Canavalia gladiate | |
| Flemingia bracteate | |
| Phaseolus roxburghii | |
| Trifolium alexandrinum | |
| Pterostilbene | Anogeisus acuminata |
| Carvacrol | Achillea millefolium |
| Epigallocatechin gallate | Ceratolimon feei |
| Helianthemum glomeratum | |
| Limonium sinense | |
| Rhodiola heterodonta | |
| Thespesia populinea | |
| Lycorine | Crinum jagus |
| Hippeastrum puniceum | |
| Polianthes tuberosa | |
| Polyanthes tuberosa | |
| Conophylline | Tabernaemontana divaricata |
| Piperlongumine | Piper longum |
| Fisetin | Acacia catechu |
| Curcumin | Cucurma angustifolia |
| Berberine | Andira inermis |
| Kaempferol | Abrus precatorius |
| Resveratrol | Artocarpus chama |
Conclusion
As our understanding of molecular and cellular aspects of ageing evolves, one can expect more targeted, effective and holistic interventions to prolong the human health span. Cellular senescence and its implications on human health, especially in geriatric diseases is well established. But it is only now that we have identified the triggers responsible for switching from a normal cell type to an arrested pro-inflammatory cell (senescent cell) and the characteristics of the senescent phenotype. Senotherapeutics are an emergent class of small molecules, specifically targeted to remove senescent cells or control the SASP mediated pathological damage. The current most successful senotherapeutic combination is dasatinib and quercetin, under investigation in several clinical trials, however we reviewed the current state of the art of senotherapeutic entities that can be a part of everyday diet. Bioactive polysacchrides, peptides and proteins, fatty acids, lipids can exhibit senolytic capacity and control the buildup of senescent cells as we age. Probiotic formulation, standalone microorganisms or in combination, can confer senomorphic advantage by countering/controlling the SASP response. Conclusively, the authors endorse mechanistic investigation into dietary entities that can aid control the age-related burden of senescent cells and also provide nutrition. The dietary senotherapeutics can be included in scientifically curated geriatric diets or formulated into targeted nutraceuticals for elderly.
Acknowledgement
The authors acknowledge the infrastructure support provided by Jaypee Institute of Information Technology, NOIDA for this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Authors’ Contribution:
- Sonam Chawla conceptualized and wrote the first and the final of the present manuscript
- Sanjukta Bhattacharya performed literature survey and prepared the figures and tables
- Aaysha Gupta contributed to editing the manuscript and finalizing the present draft.
References
- Gao X, Yu X, Zhang C, et al. Telomeres and Mitochondrial Metabolism: Implications for Cellular Senescence and Age-related Diseases. Stem Cell Rev Rep. 2022;18(7):2315-2327. doi:10.1007/s12015-022-10370-8
CrossRef - López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186(2):243-278. doi:10.1016/j.cell.2022.11.001
CrossRef - Childs BG, Durik M, Baker DJ, van Deursen JM. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015;21(12):1424-1435. doi:10.1038/nm.4000
CrossRef - Ohtani N. The roles and mechanisms of senescence-associated secretory phenotype (SASP): can it be controlled by senolysis?. Inflamm Regen. 2022;42(1):11. Published 2022 Apr 2. doi:10.1186/s41232-022-00197-8
CrossRef - Rad AN, Grillari J. Current senolytics: Mode of action, efficacy and limitations, and their future. Mech Ageing Dev. 2024;217:111888. doi:10.1016/j.mad.2023.111888
CrossRef - Justice JN, Nambiar AM, Tchkonia T, et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine. 2019;40:554-563. doi:10.1016/j.ebiom.2018.12.052
CrossRef - Gonzales MM, Garbarino VR, Marques Zilli E, et al. Senolytic Therapy to Modulate the Progression of Alzheimer’s Disease (SToMP-AD): A Pilot Clinical Trial. J Prev Alzheimers Dis. 2022;9(1):22-29. doi:10.14283/jpad.2021.62
CrossRef - Terzi MY, Izmirli M, Gogebakan B. The cell fate: senescence or quiescence. Mol Biol Rep. 2016;43(11):1213-1220. doi:10.1007/s11033-016-4065-0
CrossRef - Kumari R, Jat P. Mechanisms of Cellular Senescence: Cell Cycle Arrest and Senescence Associated Secretory Phenotype. Front Cell Dev Biol. 2021;9:645593. Published 2021 Mar 29. doi:10.3389/fcell.2021.645593
CrossRef - Harley CB, Futcher AB, Greider CW. Telomeres shorten during ageing of human fibroblasts. Nature. 1990;345(6274):458-460. doi:10.1038/345458a0
CrossRef - Vessoni AT, Zhang T, Quinet A, et al. Telomere erosion in human pluripotent stem cells leads to ATR-mediated mitotic catastrophe. J Cell Biol. 2021;220(6):e202011014. doi:10.1083/jcb.202011014
CrossRef - Molinaro C, Martoriati A, Cailliau K. Proteins from the DNA Damage Response: Regulation, Dysfunction, and Anticancer Strategies. Cancers (Basel). 2021;13(15):3819. Published 2021 Jul 29. doi:10.3390/cancers13153819.
CrossRef - Shreeya T, Ansari MS, Kumar P, et al. Senescence: A DNA damage response and its role in aging and Neurodegenerative Diseases. Front Aging. 2024;4:1292053. Published 2024 Mar 21. doi:10.3389/fragi.2023.1292053
CrossRef - Su X, Huang J. The Fanconi anemia pathway and DNA interstrand cross-link repair. Protein Cell. 2011;2(9):704-711. doi:10.1007/s13238-011-1098-y.
CrossRef - Sollier J, Stork CT, García-Rubio ML, Paulsen RD, Aguilera A, Cimprich KA. Transcription-coupled nucleotide excision repair factors promote R-loop-induced genome instability. Mol Cell. 2014;56(6):777-785. doi:10.1016/j.molcel.2014.10.020.
CrossRef - Gómez-González B, García-Rubio M, Bermejo R, et al. Genome-wide function of THO/TREX in active genes prevents R-loop-dependent replication obstacles. EMBO J. 2011;30(15):3106-3119. Published 2011 Jun 24. doi:10.1038/emboj.2011.206.
CrossRef - Terman A. Garbage catastrophe theory of aging: imperfect removal of oxidative damage?. Redox Rep. 2001;6(1):15-26. doi:10.1179/135100001101535996.
CrossRef - Nousis L, Kanavaros P, Barbouti A. Oxidative Stress-Induced Cellular Senescence: Is Labile Iron the Connecting Link?. Antioxidants (Basel). 2023;12(6):1250. Published 2023 Jun 10. doi:10.3390/antiox12061250.
CrossRef - Davalli P, Mitic T, Caporali A, Lauriola A, D’Arca D. ROS, Cell Senescence, and Novel Molecular Mechanisms in Aging and Age-Related Diseases. Oxid Med Cell Longev. 2016;2016:3565127. doi:10.1155/2016/3565127
CrossRef - Hajam YA, Rani R, Ganie SY, et al. Oxidative Stress in Human Pathology and Aging: Molecular Mechanisms and Perspectives. Cells. 2022;11(3):552. doi:10.3390/cells11030552
CrossRef - Terao R, Ahmed T, Suzumura A, Terasaki H. Oxidative Stress-Induced Cellular Senescence in Aging Retina and Age-Related Macular Degeneration. Antioxidants (Basel). 2022;11(11):2189. Published 2022 Nov 5. doi:10.3390/antiox11112189.
CrossRef - Yuan H, Xu Y, Luo Y, Wang NX, Xiao JH. Role of Nrf2 in cell senescence regulation. Mol Cell Biochem. 2021;476(1):247-259. doi:10.1007/s11010-020-03901-9.
CrossRef - Giroud J, Bouriez I, Paulus H, Pourtier A, Debacq-Chainiaux F, Pluquet O. Exploring the Communication of the SASP: Dynamic, Interactive, and Adaptive Effects on the Microenvironment. Int J Mol Sci. 2023;24(13):10788. Published 2023 Jun 28. doi:10.3390/ijms241310788
CrossRef - Zhang L, Pitcher LE, Prahalad V, Niedernhofer LJ, Robbins PD. Targeting cellular senescence with senotherapeutics: senolytics and senomorphics. FEBS J. 2023;290(5):1362-1383. doi:10.1111/febs.16350
CrossRef - Beauséjour CM, Krtolica A, Galimi F, et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 2003;22(16):4212-4222. doi:10.1093/emboj/cdg417.
CrossRef - González-Gualda E, Baker AG, Fruk L, Muñoz-Espín D. A guide to assessing cellular senescence in vitro and in vivo. FEBS J. 2021;288(1):56-80. doi:10.1111/febs.15570
CrossRef - Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol. 2014;15(7):482-496. doi:10.1038/nrm3823
CrossRef - Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of Cellular Senescence. Trends Cell Biol. 2018;28(6):436-453. doi:10.1016/j.tcb.2018.02.001
CrossRef - Di Rosa F, Cossarizza A, Hayday AC. To Ki or Not to Ki: Re-Evaluating the Use and Potentials of Ki-67 for T Cell Analysis [published correction appears in Front Immunol. 2021 Sep 28;12:756641. doi: 10.3389/fimmu.2021.756641.]. Front Immunol. 2021;12:653974. Published 2021 Apr 9. doi:10.3389/fimmu.2021.653974
CrossRef - Aird KM, Zhang R. Detection of senescence-associated heterochromatin foci (SAHF). Methods Mol Biol. 2013;965:185-196. doi:10.1007/978-1-62703-239-1_12
CrossRef - Shi X, Tian B, Liu L, et al. Rb protein is essential to the senescence-associated heterochromatic foci formation induced by HMGA2 in primary WI38 cells. J Genet Genomics. 2013;40(8):391-398. doi:10.1016/j.jgg.2013.05.007
CrossRef - Chen T, Xue L, Niu J, et al. The retinoblastoma protein selectively represses E2F1 targets via a TAAC DNA element during cellular senescence. J Biol Chem. 2012;287(44):37540-37551. doi:10.1074/jbc.M111.260679
CrossRef - Zhou D, Borsa M, Simon AK. Hallmarks and detection techniques of cellular senescence and cellular ageing in immune cells. Aging Cell. 2021;20(2):e13316. doi:10.1111/acel.13316
CrossRef - Levine B, Kroemer G. SnapShot: Macroautophagy. Cell. 2008;132(1):162.e1-162.e3. doi:10.1016/j.cell.2007.12.026.
CrossRef - Yang Z, Klionsky DJ. An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol. 2009;335:1-32. doi:10.1007/978-3-642-00302-8_1
CrossRef - Kwon Y, Kim JW, Jeoung JA, Kim MS, Kang C. Autophagy Is Pro-Senescence When Seen in Close-Up, but Anti-Senescence in Long-Shot. Mol Cells. 2017;40(9):607-612. doi:10.14348/molcells.2017.0151
CrossRef - Deretic V. Autophagosome and phagosome. Methods Mol Biol. 2008;445:1-10. doi:10.1007/978-1-59745-157-4_1
CrossRef - Dice JF. Chaperone-mediated autophagy. Autophagy. 2007;3(4):295-299. doi:10.4161/auto.4144.
CrossRef - Valieva Y, Ivanova E, Fayzullin A, Kurkov A, Igrunkova A. Senescence-Associated β-Galactosidase Detection in Pathology. Diagnostics (Basel). 2022;12(10):2309. Published 2022 Sep 25. doi:10.3390/diagnostics12102309
CrossRef - Itahana K, Itahana Y, Dimri GP. Colorimetric detection of senescence-associated β galactosidase. Methods Mol Biol. 2013;965:143-156. doi:10.1007/978-1-62703-239-1_8
CrossRef - Davan-Wetton CSA, Montero-Melendez T. An optimised protocol for the detection of lipofuscin, a versatile and quantifiable marker of cellular senescence. PLoS One. 2024;19(7):e0306275. Published 2024 Jul 15. doi:10.1371/journal.pone.0306275.
CrossRef - Magkouta S, Veroutis D, Pousias A, et al. A fluorophore-conjugated reagent enabling rapid detection, isolation and live tracking of senescent cells. Mol Cell. 2023;83(19):3558-3573.e7. doi:10.1016/j.molcel.2023.09.006.
CrossRefv - Wang B, Han J, Elisseeff JH, Demaria M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol. 2024;25(12):958-978. doi:10.1038/s41580-024-00727-x.
CrossRef - Birch J, Gil J. Senescence and the SASP: many therapeutic avenues. Genes Dev. 2020;34(23-24):1565-1576. doi:10.1101/gad.343129.120.
CrossRef - Coppé JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99-118. doi:10.1146/annurev-pathol-121808-102144
CrossRef - Li W, Deng X, Chen J. RNA-binding proteins in regulating mRNA stability and translation: roles and mechanisms in cancer. Semin Cancer Biol. 2022;86(Pt 2):664-677. doi:10.1016/j.semcancer.2022.03.025.
CrossRef - Wiley CD, Velarde MC, Lecot P, et al. Mitochondrial Dysfunction Induces Senescence with a Distinct Secretory Phenotype. Cell Metab. 2016;23(2):303-314. doi:10.1016/j.cmet.2015.11.011
CrossRef - Miwa S, Kashyap S, Chini E, von Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Invest. 2022;132(13):e158447. doi:10.1172/JCI158447
CrossRef - Basisty N, Kale A, Jeon OH, et al. A proteomic atlas of senescence-associated secretomes for aging biomarker development. PLoS Biol. 2020;18(1):e3000599. Published 2020 Jan 16. doi:10.1371/journal.pbio.3000599.
CrossRef - Krishnamurthy J, Torrice C, Ramsey MR, et al. Ink4a/Arf expression is a biomarker of aging. J Clin Invest. 2004;114(9):1299-1307. doi:10.1172/JCI22475
CrossRef - Kim EC, Kim JR. Senotherapeutics: emerging strategy for healthy aging and age-related disease. BMB Rep. 2019;52(1):47-55. doi:10.5483/BMBRep.2019.52.1.293
CrossRef - Park J, Shin DW. Senotherapeutics and Their Molecular Mechanism for Improving Aging. Biomol Ther (Seoul). 2022;30(6):490-500. doi:10.4062/biomolther.2022.114.
CrossRef - Khavinson V, Linkova N, Dyatlova A, Kantemirova R, Kozlov K. Senescence-Associated Secretory Phenotype of Cardiovascular System Cells and Inflammaging: Perspectives of Peptide Regulation. Cells. 2022;12(1):106. Published 2022 Dec 27. doi:10.3390/cells12010106
CrossRef - Luís C, Maduro AT, Pereira P, Mendes JJ, Soares R, Ramalho R. Nutritional senolytics and senomorphics: Implications to immune cells metabolism and aging – from theory to practice. Front Nutr. 2022;9:958563. Published 2022 Sep 8. doi:10.3389/fnut.2022.958563.
CrossRef - Zhu Y, Doornebal EJ, Pirtskhalava T, et al. New agents that target senescent cells: the flavone, fisetin, and the BCL-XLinhibitors, A1331852 and A1155463. Aging (Albany NY). 2017;9(3):955-963. doi:10.18632/aging.101202.
CrossRef - Kim J, Kim HS, Choi DH, et al. Kaempferol tetrasaccharides restore skin atrophy via PDK1 inhibition in human skin cells and tissues: Bench and clinical studies. Biomed Pharmacother. 2022;156:113864. doi:10.1016/j.biopha.2022.113864.
CrossRef - Thompson EL, Pitcher LE, Niedernhofer LJ, Robbins PD. Targeting Cellular Senescence with Senotherapeutics: Development of New Approaches for Skin Care. Plast Reconstr Surg. 2022;150:12S-19S. doi:10.1097/PRS.0000000000009668.
CrossRef - Bi Y, Qiao X, Cai Z, et al. Exosomal miR-302b rejuvenates aging mice by reversing the proliferative arrest of senescent cells. Cell Metab. 2025;37(2):527-541.e6. doi:10.1016/j.cmet.2024.11.013.
CrossRef - Imb M, Véghelyi Z, Maurer M, Kühnel H. Exploring Senolytic and Senomorphic Properties of Medicinal Plants for Anti-Aging Therapies. Int J Mol Sci. 2024;25(19):10419. Published 2024 Sep 27. doi:10.3390/ijms251910419
CrossRef - Nambiar A, Kellogg D 3rd, Justice J, et al. Senolytics dasatinib and quercetin in idiopathic pulmonary fibrosis: results of a phase I, single-blind, single-center, randomized, placebo-controlled pilot trial on feasibility and tolerability. EBioMedicine. 2023;90:104481. doi:10.1016/j.ebiom.2023.104481
CrossRef - Raffaele M, Vinciguerra M. The costs and benefits of senotherapeutics for human health. Lancet Healthy Longev. 2022;3(1):e67-e77. doi:10.1016/S2666-7568(21)00300-7
CrossRef - James Stubbs R, Horgan G, Robinson E, Hopkins M, Dakin C, Finlayson G. Diet composition and energy intake in humans. Philos Trans R Soc Lond B Biol Sci. 2023;378(1888):20220449. doi:10.1098/rstb.2022.0449.
CrossRef - Kelly T, Unwin D, Finucane F. Low-Carbohydrate Diets in the Management of Obesity and Type 2 Diabetes: A Review from Clinicians Using the Approach in Practice. Int J Environ Res Public Health. 2020;17(7):2557. doi:10.3390/ijerph17072557
CrossRef - Sun J, Ruan Y, Xu N, et al. The effect of dietary carbohydrate and calorie restriction on weight and metabolic health in overweight/obese individuals: a multi-center randomized controlled trial. BMC Med. 2023;21(1):192. doi:10.1186/s12916-023-02869-9.
CrossRef - Yao C, Guan X, Carraro G, et al. Senescence of Alveolar Type 2 Cells Drives Progressive Pulmonary Fibrosis [published correction appears in Am J Respir Crit Care Med. 2021 Jul 1;204(1):113. doi: 10.1164/rccm.v204erratum1.]. Am J Respir Crit Care Med. 2021;203(6):707-717. doi:10.1164/rccm.202004-1274OC.
CrossRef - Shen J, Qin H, Li K, et al. The angelica Polysaccharide: a review of phytochemistry, pharmacology and beneficial effects on systemic diseases. Int Immunopharmacol. 2024;133:112025. doi:10.1016/j.intimp.2024.112025
CrossRef - Mu X, Zhang Y, Li J, et al. Angelica Sinensis Polysaccharide Prevents Hematopoietic Stem Cells Senescence in D-Galactose-Induced Aging Mouse Model. Stem Cells Int. 2017;2017:3508907. doi:10.1155/2017/3508907.
CrossRef - Zhao J, Garcia D, Gartung A, Lee MJ. Sphingosine-1-phosphate receptor subtype 2 signaling in endothelial senescence-associated functional impairments and inflammation. Curr Atheroscler Rep. 2015;17(5):504. doi:10.1007/s11883-015-0504-y
CrossRef - Guo X, Luo J, Qi J, et al. The Role and Mechanism of Polysaccharides in Anti-Aging. Nutrients. 2022;14(24):5330. Published 2022 Dec 15. doi:10.3390/nu14245330
CrossRef - Lee JH, Yun CW, Hur J, Lee SH. Fucoidan Rescues p-Cresol-Induced Cellular Senescence in Mesenchymal Stem Cells via FAK-Akt-TWIST Axis. Mar Drugs. 2018;16(4):121. Published 2018 Apr 6. doi:10.3390/md16040121
CrossRef - Le Couteur DG, Solon-Biet S, Cogger VC, et al. The impact of low-protein high-carbohydrate diets on aging and lifespan. Cell Mol Life Sci. 2016;73(6):1237-1252. doi:10.1007/s00018-015-2120-y
CrossRef - Joy J, Barrio L, Santos-Tapia C, et al. Proteostasis failure and mitochondrial dysfunction leads to aneuploidy-induced senescence. Dev Cell. 2021;56(14):2043-2058.e7. doi:10.1016/j.devcel.2021.06.009
CrossRef - Sabath N, Levy-Adam F, Younis A, et al. Cellular proteostasis decline in human senescence. Proc Natl Acad Sci U S A. 2020;117(50):31902-31913. doi:10.1073/pnas.2018138117
CrossRef - Wang Q, Huang Y, Qin C, et al. Bioactive Peptides from Angelica sinensis Protein Hydrolyzate Delay Senescence in Caenorhabditis elegans through Antioxidant Activities. Oxid Med Cell Longev. 2016;2016:8956981. doi:10.1155/2016/8956981
CrossRef - Garg A, Saroj J, Tiwari S, et al. Exploring the potential anti-senescence effects of soybean-derived peptide Soymetide in mice hippocampal neurons via the Wnt/β-catenin pathway. Front Pharmacol. 2025;16:1510337. Published 2025 Feb 25. doi:10.3389/fphar.2025.1510337.
CrossRef - Wang J, Wu Y, Chen Z, Chen Y, Lin Q, Liang Y. Exogenous Bioactive Peptides Have a Potential Therapeutic Role in Delaying Aging in Rodent Models. Int J Mol Sci. 2022;23(3):1421. Published 2022 Jan 26. doi:10.3390/ijms23031421
CrossRef - Asokan SM, Wang T, Wang MF, Lin WT. A novel dipeptide from potato protein hydrolysate augments the effects of exercise training against high-fat diet-induced damages in senescence-accelerated mouse-prone 8 by boosting pAMPK / SIRT1/ PGC-1α/ pFOXO3 pathway. Aging (Albany NY). 2020;12(8):7334-7349. doi:10.18632/aging.103081
CrossRef - Jung H, Jung D, Lee J, et al. Bioactive peptides in the pancreatin-hydrolysates of whey protein support cell proliferation and scavenge reactive oxygen species. Anim Cells Syst (Seoul). 2022;26(5):232-242. Published 2022 Oct 11. doi:10.1080/19768354.2022.2130425.
CrossRef - Nehme J, Yang D, Altulea A, et al. High dietary protein and fat contents exacerbate hepatic senescence and SASP in mice. FEBS J. 2023;290(5):1340-1347. doi:10.1111/febs.16292
CrossRef - Seyedsadjadi N, Berg J, Bilgin AA, Braidy N, Salonikas C, Grant R. High protein intake is associated with low plasma NAD+ levels in a healthy human cohort. PLoS One. 2018;13(8):e0201968. doi:10.1371/journal.pone.0201968
CrossRef - de Magalhães JP, Müller M, Rainger GE, Steegenga W. Fish oil supplements, longevity and aging. Aging (Albany NY). 2016;8(8):1578-1582. doi:10.18632/aging.101021.
CrossRef - Djuricic I, Calder PC. Beneficial Outcomes of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Human Health: An Update for 2021. Nutrients. 2021;13(7):2421. Published 2021 Jul 15. doi:10.3390/nu13072421
CrossRef - Millner A, Atilla-Gokcumen GE. Lipid Players of Cellular Senescence. Metabolites. 2020;10(9):339. Published 2020 Aug 21. doi:10.3390/metabo10090339
CrossRef - Venable ME, Lee JY, Smyth MJ, Bielawska A, Obeid LM. Role of ceramide in cellular senescence. J Biol Chem. 1995;270(51):30701-30708. doi:10.1074/jbc.270.51.30701
CrossRef - Chawla S, Sahni C, Tulsawani R, et al. Exogenous sphingosine 1-phosphate protects murine splenocytes against hypoxia-induced injury. Lipids. 2014;49(2):191-202. doi:10.1007/s11745-013-3860-9
CrossRef - Kim MK, Lee W, Yoon GH, Chang EJ, Choi SC, Kim SW. Links between accelerated replicative cellular senescence and down-regulation of SPHK1 transcription. BMB Rep. 2019;52(3):220-225. doi:10.5483/BMBRep.2019.52.3.012
CrossRef - Panneer Selvam S, De Palma RM, Oaks JJ, et al. Binding of the sphingolipid S1P to hTERT stabilizes telomerase at the nuclear periphery by allosterically mimicking protein phosphorylation. Sci Signal. 2015;8(381):ra58. Published 2015 Jun 16. doi:10.1126/scisignal.aaa4998
CrossRef - Lizardo DY, Lin YL, Gokcumen O, Atilla-Gokcumen GE. Regulation of lipids is central to replicative senescence. Mol Biosyst. 2017;13(3):498-509. doi:10.1039/c6mb00842a
CrossRef - Fafián-Labora J, Carpintero-Fernández P, Jordan SJD, et al. FASN activity is important for the initial stages of the induction of senescence. Cell Death Dis. 2019;10(4):318. Published 2019 Apr 8. doi:10.1038/s41419-019-1550-0.
CrossRef - Chan J, Eide IA, Tannæs TM, Waldum-Grevbo B, Jenssen T, Svensson M. Marine n-3 Polyunsaturated Fatty Acids and Cellular Senescence Markers in Incident Kidney Transplant Recipients: The Omega-3 Fatty Acids in Renal Transplantation (ORENTRA) Randomized Clinical Trial. Kidney Med. 2021;3(6):1041-1049. Published 2021 Oct 4. doi:10.1016/j.xkme.2021.07.010
CrossRef - Farzaneh-Far R, Lin J, Epel ES, Harris WS, Blackburn EH, Whooley MA. Association of marine omega-3 fatty acid levels with telomeric aging in patients with coronary heart disease. JAMA. 2010;303(3):250-257. doi:10.1001/jama.2009.2008
CrossRef - Sakai C, Ishida M, Ohba H, et al. Fish oil omega-3 polyunsaturated fatty acids attenuate oxidative stress-induced DNA damage in vascular endothelial cells. PLoS One. 2017;12(11):e0187934. Published 2017 Nov 9. doi:10.1371/journal.pone.0187934
CrossRef - Li HY, Zhou DD, Gan RY, et al. Effects and Mechanisms of Probiotics, Prebiotics, Synbiotics, and Postbiotics on Metabolic Diseases Targeting Gut Microbiota: A Narrative Review. Nutrients. 2021;13(9):3211. Published 2021 Sep 15. doi:10.3390/nu13093211.
CrossRef - Tuikhar N, Keisam S, Labala RK, et al. Comparative analysis of the gut microbiota in centenarians and young adults shows a common signature across genotypically non-related populations. Mech Ageing Dev. 2019;179:23-35. doi:10.1016/j.mad.2019.02.001
CrossRef - Yoshimoto S, Loo TM, Atarashi K, et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome [published correction appears in Nature. 2014 Feb 20;506(7488):396. Hattori, Masahisa [corrected to Hattori, Masahira]]. Nature. 2013;499(7456):97-101. doi:10.1038/nature12347
CrossRef - Jeong JJ, Kim KA, Jang SE, Woo JY, Han MJ, Kim DH. Orally administrated Lactobacillus pentosus var. plantarum C29 ameliorates age-dependent colitis by inhibiting the nuclear factor-kappa B signaling pathway via the regulation of lipopolysaccharide production by gut microbiota [published correction appears in PLoS One. 2015 Nov 12;10(11):e0142521. doi: 10.1371/journal.pone.0142521.]. PLoS One. 2015;10(2):e0116533. Published 2015 Feb 17. doi:10.1371/journal.pone.0116533.
CrossRef - Fang X, Yue M, Wei J, et al. Evaluation of the Anti-Aging Effects of a Probiotic Combination Isolated From Centenarians in a SAMP8 Mouse Model. Front Immunol. 2021;12:792746. Published 2021 Dec 2. doi:10.3389/fimmu.2021.792746
CrossRef
Abbreviation List
cGAS – cyclic GMP–AMP synthase ; CIP1 – CDK interacting protein-1 ; CMA – Chaperone-mediated autophagy ; DDR – DNA Damage Response; DSB – Double stranded break; HP1 – heterochromatin protein 1 ; MAPKs – mitogen-associated protein kinases ; MiDAS – Mitochondrial Dysfunction-Associated Senescence ; mtDNA – mitochondrial DNA ; mTOR – Mammalian target of Rapamycin; NEMO – NF-κB essential modulator ; NER – Nucleotide excision repair ; NF-κB -Nuclear Factor kappa-light-chain-enhancer of activated B cells; NLRP3 – (NOD)-like receptor protein 3; p21 – Cyclin-dependent kinase inhibitor 1 (CDKN1A); PDK1 – 3-Phosphoinositide-dependent protein kinase 1; PI3K – Phosphoinositide 3-Kinase; POT1 – protection of the telomere 1; QoL : Quality of Life; RAP1 – repressor/activator protein 1; Rb – Retinoblastoma protein; RBPs – RNA-binding proteins ; ROS – Reactive Oxygen Species; S1P – Sphingosine-1-phosphate; SAHF – Senescence-Associated Heterochromatin Foci; SAMP8 – Senescence accelerated mouse-prone 8; SASP – Senescence-Associated Secretory Phenotype; SA-β-Gal – Senescence-Associated β-Galactosidase; SCAPs – Senescent cell anti‐apoptotic pathways; STAT – Signal Transducer and Activator of Transcription; STING – Stimulator of interferon genes; TCR – T cell receptor ; TRF1 – telomere repeat factors 1







