Manuscript accepted on :14-08-2025
Published online on: 25-08-2025
Plagiarism Check: Yes
Reviewed by: Dr. Feng Li and Dr. Elina Margarida Ribeiro Marinho
Second Review by: Dr. Patel Maulik
Final Approval by: Dr. Patorn Piromchai
Madhanagopal Manoharan1,2* , Bhavadharani Babu3
, Bhavadharani Babu3 , Sindu Divakaran3
, Sindu Divakaran3 , Gobi Saravanan Kaliaraj2
, Gobi Saravanan Kaliaraj2 , Perumal Sudalai4
, Perumal Sudalai4 ,and Rajendra Thilahar Chelladurai5
,and Rajendra Thilahar Chelladurai5
1Department of Mechanical Engineering, Indian Institute of Information Technology Design and Manufacturing Kancheepuram, Chennai, India.
2Center of excellence for Additive Manufacturing, Sathyabama Institute of Science and Technology, Chennai, India.
3Department of Biomedical Engineering, Sathyabama Institute of Science and Technology, Chennai, India.
4Department of Mechanical Engineering, Karpaga Vinayaga College of Engineering and Technology, Chengalpet, India.
5Department of Artificial Intelligence and Data Science, Saveetha Engineering College, Chennai, India.
Corresponding Author E-mail: madhanmsm10@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3239
Abstract
The usage of Additive Manufacturing (AM) by the healthcare industry to manufacture biomedical implants results in mass customized product at an affordable price. Prior to the advent of AM, these biomedical implants are fabricated using conventional manufacturing techniques such as casting, machining etc. These patient specific implants may aid the medical workers in the placement, inventory with increased customer satisfaction. There is a clear distinction between make or buy decision for these medical implants and systematic investigation is needed to procure these AM implants. In addition, these implants are designed and manufactured from the Digital Imaging and Communication in Medicine (DICOM) file from the respective patients. Polymer based Material Extrusion and Resin based Vat Photo polymerization are the commonly used AM techniques, which assist the health workers in validating the implant design. While the metal AM technique such as Powder Bed Fusion are used the widely employed method to fabricate the actual medical implants, which can be directly fit on the patient body. Most of the implants are used for dental applications using maxillofacial surgery and orthopaedic applications for bone joints. This paper briefs the impact of AM in the healthcare industry for mass customisation, strategies, methodologies and applications especially for implants. And some of the software tools used for the design, analysis and manufacturing of these implants are also discussed. Mass customisation and cost effectiveness are the major driving force for the use of medical implants. These patient specific medical implants with AM are useful for developing industries to overcome the expense associated with patient specific medical implants, which is usually fabricated using conventional manufacturing processes.
Keywords
Additive Manufacturing; Affordability; Biomedical implants; Customized implants; Three-dimensional printing
Download this article as:| Copy the following to cite this article: Manoharan M, Babu B, Divakaran S, Kaliaraj G. S, Sudalai P, Chelladurai R. T. Recent Advancements in the Bio-medical Implant Production using Additive Manufacturing – Comparison, Challenges and Future Scope. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Manoharan M, Babu B, Divakaran S, Kaliaraj G. S, Sudalai P, Chelladurai R. T. Recent Advancements in the Bio-medical Implant Production using Additive Manufacturing – Comparison, Challenges and Future Scope. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3HQKWUi |
Introduction
Additive Manufacturing (AM) or 3D printing plays a major role in healthcare sectors. This article provides a comprehensive survey on the use of AM techniques for manufacturing customised medical and dental implants at an affordable price. The types of implants, manufacturing methodologies, applications, advantages and drawbacks of the medical implants are discussed in this work. Implant devices can be placed using surgery in the jaw portion for rejuvenating the broken anatomical structures especially bone regions.1 This type of implants is majorly used for osteosynthesis for correcting the existing bone and enhancing the growth of bones. Figure 1. shows the sequence of steps involved in fabricating implants using AM.
 |
Figure 1: Steps involved in fabricating implants using AM. |
There are several types of AM techniques used for manufacturing implants.2 Powder Bed Fusion technique involves the use of a laser source, electron beam source to melt a thin layer of metal powder, which is then solidified to create the implant. This technique can be further divided into two subtypes: selective laser melting (SLM) and electron beam melting (EBM). In Directed Energy Deposition technique, metal powder or wire is fed into a melt pool created by a laser or an electron beam, and the material is added layer by layer to create the implant. Material extrusion based AM technique is the use of the deposition of a thermoplastic or metal filament, which is melted and extruded through a nozzle to create the implant. In the Binder jetting technique, a powder bed is selectively sprayed with a liquid binder to bind the particles together and form the implant. Sheet lamination involves the layering of thin sheets of metal or polymer to create the implant. AM techniques have lot of advantages and disadvantages, while the selection of a particular technique for manufacturing implants depends on factors such as the type of implant, the required precision and resolution, the desired mechanical properties, and the materials.
Materials and Methods
Implants are manufactured from a variety of materials, each with its own unique properties and benefits.3 Metals such as titanium and its alloys, cobalt-chromium, and stainless steel are used in the implant production because of their strength and biocompatibility. They can be shaped and sized to fit specific anatomical locations. Ceramics such as zirconia and alumina are used in implants. They are also highly resistant to wear and corrosion, making them a popular choice for joint replacement implants. Polymers such as polyethylene, polypropylene, and silicone are used in a variety of implants, including breast implants, vascular grafts, and temporary sutures. They are lightweight, flexible, and biocompatible. Composites are materials made from a combination of many materials to create a new material with specific properties. For instance, polymers reinforced with carbon fibers are used in orthopaedic implants to provide strength and rigidity. Biological materials such as human or animal tissue are also used in implants, such as in heart valve replacements. These materials are often processed and treated to reduce the risk of infection or rejection by the body. Factors affecting implant selection include specific application, patient’s anatomy and medical history, and the expected longevity of the implant.4 The choice of material also affects the cost and availability of the implant,5 as well as the complexity of the surgical procedure.
Abutment-implant connections
The abutment-implant connection is a critical interface in dental implant systems, as it directly affects the stability and performance of the implant.6 External hexagon type of connection features an external hexagon on the implant and a matching hexagonal slot on the abutment. Sometimes, there will be a difficulty to position accurately and can result in implant stability. Internal hexagon connection features an internal hexagonal shape on the implant and a matching hexagonal end on the abutment. It provides a strong connection and is easier to position accurately than the external hexagon connection. Morse taper features can result in improved stability and longevity, with the implant tapering down to a narrower diameter than the abutment. This creates a tight, frictional fit between the implant and abutment and provides excellent stability. One-piece implant features an abutment that is an integral part of the implant. The one-piece design eliminates the abutment-implant interface, which can result in improving the stability and mechanical performance. Figure 2 shows the overall classification of medical implants, which can be manufactured using AM.
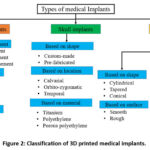 |
Figure 2: Classification of 3D printed medical implants. |
The choice of abutment-implant connection depends on several factors, including the specific implant system being used, the anatomy of the patient, and the desired outcome. The design and quality of the abutment-implant connection can directly affect the stability and longevity of the implant, as well as its ability to withstand functional loads7. Proper selection and placement of the abutment-implant connection is critical to ensuring optimal outcomes for dental implant patients. The comparison of knee implants, skull implants and dental implants by considering the factors such as dissimilarities, advantages, limitations and applications are provided in the following table 1.
Table 1: Comparison of implants based on their properties.
| Similarities | Feature | Knee | Skull | Dental |
| Material types | Titanium, Co-Cr alloys, UHMWPE | Titanium, PEEK, PMMA | Titanium, Zirconia | |
| Biocompatibility | Yes | Yes | Yes | |
| Surgical procedure required | Yes | Yes | Yes | |
| Customization using CAD/CAM & 3D printing | Increasingly used | Widely used for patient-specific implants | Common in crowns, bridges, and root implants | |
| Longevity | Long-term | Long-term | Long-term | |
| Integration with bone | Osseointegration or mechanical fixation | Osseointegration is possible | Osseointegration is critical. | |
| Advantages | · Restores joint mobility.· Improves quality of life.
· Durable for minimum of 15 to 20 years. |
· Protects brain.· Aesthetic reconstruction.
· Can be patient-specific and tailored for congenital issues. |
·High success rate.·Maintains jaw bone density.
·Improves chewing and speech. |
|
| Limitations | · Wear and tear of polyethylene insert.· Risk of infection or loosening.
· Not ideal for very young patients. |
· Risk of infection or implant rejection.· Limited bone integration in some materials.
· Complex surgery with neurological risks. |
· Bone resorption risk.· Requires adequate bone volume.
· Requires lot of expense treatment if oral hygiene is not maintained properly. |
|
| Applications | · Osteoarthritis.· Rheumatoid arthritis.
· Post-traumatic arthritis. |
· Trauma reconstruction.· Tumor resection.
· Congenital deformities. |
·Tooth replacement.·Full/partial dentures.
·Orthodontic anchorage |
|
Dental implants
Dentistry involves the detection and control of teeth related bones by proper control with the available medicinal techniques and tools. The implants manufactured using AM are highly customizable, patient-specific and reduce the effort of the dental doctors for placing implants in the appropriate locations. AM allows for the creation of implants that are customized to the anatomy of the patient.7 This means that the implant will fit perfectly and function more effectively. Additionally, the patient will experience less discomfort and a faster recovery time. With AM, it is possible to use a wider range of materials, including metals, polymers, and ceramics, each with their own unique properties. This means that biomedical implants can be designed with materials that are biocompatible, durable, and have the required mechanical properties for the intended applications such as fatigue resistance, osseointegration, etc.8 AM allows for the creation of complex geometries that cannot be produced using traditional manufacturing methods. This is particularly useful in the creation of biomedical implants, which often require intricate designs to mimic the shape and function of natural structures in the body.9 AM can produce biomedical implants more quickly and at a lower cost than traditional manufacturing methods. This is due to the fact that there is less waste, and the manufacturing process can be automated to a greater degree. AM allows for rapid prototyping and design iteration, which is particularly useful for testing new designs and refine more quickly and efficiently, leading to better products and improved patient outcomes. In summary, AM offers several benefits for the production of biomedical implants, including customization, the ability to use a wider range of materials, the ability to create complex geometries, time and cost savings, and improved research and development capabilities. As a result, it is becoming an increasingly popular option in the biomedical industry. The medical implants need careful consideration of design, manufacturing and development along with analyzing the cost associated with producing these biomedical implants. Figure 3. shows the classification of bone implants based on the impact of damage, particularly dental implants.
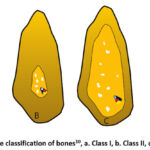 |
Figure 3: Qualitative classification of bones10, a. Class I, b. Class II, c. Class III, d. Class IV. |
Some of the stochastic cost modelling techniques are used to quantify the supply-chain associated costs, which affects the cost of production of these implants using AM techniques. An investigation is required to understand the economic feasibility to produce these customised biomedical implants at the hospital locations.
Classification of dental implants
There are four types of dental implants such as endosteal implants, sub-periosteal implants, all-on-4 dental implants, and implant over dentures. Dental implants can be classified based on several criteria, including shape, size, surface characteristics, and the materials used to manufacture them. These implants are classified based on shape such as cylindrical, tapered and conical. Cylindrical implants have a uniform diameter along the length of the implant. Tapered implants are wider at the top than at the bottom, mimicking the natural shape of the tooth root. Conical implants have a wider base and a narrower tip. Based on size of implant, the implants are classified into narrow diameter, standard diameter and wide diameter implants. Narrow diameter implants have a diameter of less than 3.5 mm and are often used in areas where there is limited space or bone density. Standard diameter implants have a diameter of between 3.5 mm and 4.2 mm and are suitable for most patients. Wide diameter implants have a diameter of greater than 4.2 mm and are used when more stability is required. Implants are classified based on the nature of surface characteristics. Smooth surface implants have a polished surface and are used when there is sufficient bone density. Rough surface implants have a textured surface to encourage bone growth and osseointegration. These implants are then classified into the nature of materials used for making implants. Titanium implants are the most commonly used implants and are known for their biocompatibility and durability. Zirconia implants are newer implants made of a white ceramic material that is more aesthetically pleasing than titanium.
Additive Manufacturing of dental implants
AM is a promising technology for the production of dental implants. AM enables the creation of highly precise and customized dental implants. With reverse engineering and design software, the dental anatomy of the patient can be captured, and a custom implant can be designed and printed based on their specific needs. Figure 4. shows the Basal Osseo-integrated Implant (BOI) optimized using customized 3D printing.
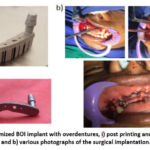 |
Figure 4: a) Optimized BOI implant with overdentures, i) post printing and ii) post processed and b) various photographs of the surgical implantation.11 |
Traditional methods of producing dental implants can be time-consuming and labor-intensive. AM can reduce production time and costs by allowing for the creation of dental implants in a single process without the need for molds or multiple manufacturing steps. AM enables the use of a wide range of materials, including biocompatible metals, ceramics, and polymers, which can be selected based on their specific mechanical and biological properties for each individual patient. Traditional manufacturing methods may result in significant waste of material due to the need for molds and other tooling.12 AM can significantly reduce waste by only using the necessary material to produce the implant. AM can improve patient outcomes by creating more accurate and precise dental implants, reducing the failure of the implant, and also improves the implant quality. Overall, AM is a promising technology for manufacturing dental implants, with potential benefits for both patients and manufacturers. As research continues, AM is likely to become an increasingly popular and effective method for creating customized dental implants.
Treatment with dental implants
The first step is to have a dentist consultancy as well as dental implant specialist for identifying yourself as a potential candidate for dental implants. The dentist examination includes your mouth and take X-rays or CT scans to evaluate the condition of your teeth, gums, and jawbone. Once the dentist determines that you are a suitable candidate for dental implants, they will create a personalized treatment plan that outlines the number of implants required, the location of the implants, and any additional procedures that may be necessary. Figure 5. shows the treatment options for replacing missing teeth with the implants.
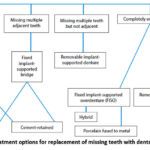 |
Figure 5: Treatment options for replacement of missing teeth with dental implants.10 |
The dentist will surgically place the dental implant into the jawbone. The implant will act as a replacement for the tooth root and will be left to heal and fuse with the bone through a process called osseointegration. After the implant has fused with the bone, the dentist will attach an abutment to the top of the implant. The abutment is a small connector that protrudes from the gum line and will support the dental crown. Once the abutment is securely attached to the implant, the dentist will place a dental crown on top of the abutment. The dental crown will be custom-made to match the shape, size, and colour of your existing teeth, and will be permanently attached to the abutment. After the implant treatment is complete, you will need to visit the dentist for regular check-ups and cleanings. It is important to maintain good oral hygiene habits to prevent infection or damage to the implant. The entire dental implant treatment process can take several months to complete, depending on the individual case. It is important to follow the dentist’s instructions and attend all follow-up visits to ensure the success of the treatment. With proper care, dental implants can last for many years and improve your oral health, speech, and chewing ability.
Current research in dental implants
Current research in dental implants is focused on improving the design and materials of dental implants, as well as developing new techniques to enhance the success of implant surgery. Researchers are exploring various surface modifications to improve the osseointegration (the process of bone growing around and attaching to the implant) of dental implants. These modifications include coatings of bioactive materials such as hydroxyapatite or modifying the implant’s surface topography. 3D printing technology is being used to create custom-designed dental implants with greater precision and accuracy13, which can improve the fit and function of the implant and reduce the risk of complications. Researchers are investigating the use of smart implants that can monitor and report on the implant’s performance, including osseointegration and the presence of bacteria. These implants could help detect early signs of implant failure or infection and improve the long-term success of the implant. Researchers are exploring the use of tissue engineering techniques to create biological replacements for missing or damaged teeth. This could eliminate the need for traditional dental implants and provide more natural-looking and functioning replacement teeth. In bone regeneration, researchers are investigating various methods to stimulate bone regeneration in the jaw to improve the success of dental implant surgery. These methods include the use of growth factors, stem cells, and other regenerative medicine techniques. Overall, current research in dental implants is focused on improving the safety, efficacy, and long-term outcomes of dental implant surgery. These advances can improve the health of patients with missing or damaged teeth and reduce the need for traditional dentures or bridges.
Orthopaedic implants
Orthopaedic implants are commonly used in orthopaedic surgery to treat a variety of conditions such as fractures, joint degeneration, and osteoporosis.14 Orthopaedic implants are usually made from metals, ceramics, polymers, and composites, depending on their intended use and the specific requirements of the implant. Joint replacements are used to replace damaged or diseased joints, such as the hip, knee, or shoulder. Joint replacements are typically made of a combination of metal and plastic. Bone plates and screws are used to hold fractured bones together while they heal. They are typically made of titanium or stainless steel. Spinal implants are used to stabilize the spine and treat conditions such as degenerative disc disease and spinal stenosis. Spinal implants can be made of a variety of materials, including titanium, cobalt-chromium alloys, and polymers. Orthopaedic trauma implants are used to treat fractures and other types of traumatic injuries. They can include bone screws, plates, rods, and wires. The manufacturing process for orthopaedic implants involves several steps, including material selection, design and prototyping, manufacturing, and testing. Implant testing can include mechanical and fatigue testing to evaluate the strength and durability of the implant, biocompatibility testing to assess the implant’s interaction with biological tissue, and microbiological testing to evaluate the implant’s ability to resist infection. Overall, orthopaedic implants have revolutionized the field of orthopaedic surgery and have greatly improved the quality of life for millions of people. By using advanced materials, manufacturing techniques, and testing protocols, orthopaedic implant manufacturers can ensure the safety, reliability, and effectiveness of their products.
Types of orthopaedic implants
There are several types of orthopaedic implants used in modern medicine, each designed for a specific purpose. Joint replacements are used to replace damaged or diseased joints in the body, such as the hip, knee, or shoulder. They are typically made of a combination of metal and plastic. Bone plates and screws are used to hold fractured bones together while they heal. They are usually made of titanium or stainless steel. Spinal implants are used to stabilize the spine and treat conditions such as degenerative disc disease and spinal stenosis. They can be made of a variety of materials, including titanium, cobalt-chromium alloys, and polymers. Orthopaedic trauma implants are used to treat fractures and other types of traumatic injuries. They can include bone screws, plates, rods, and wires. Orthopaedic joint fixation are used to provide support and stability to joints during the healing process after surgery. They can include external fixation devices, such as braces and casts, or internal fixation devices, such as screws and plates. Orthopaedic prosthetics are used to replace missing or damaged body parts, such as limbs or joints. They can include prosthetic limbs, hip or knee replacements, and spinal cord stimulators. The selection of a specific type of orthopaedic implant depends on the patient’s individual needs, the type of injury or condition being treated, and the surgeon’s preferences. Each type of implant has its own unique properties, advantages, and disadvantages, and careful consideration is given to the selection of the most appropriate implant for each patient.
Knee implants
Knee implants, also known as knee arthroplasty or knee replacement surgery, are medical devices designed to replace damaged or diseased knee joints with artificial components. These implants are typically used to treat patients who have severe knee pain and stiffness caused by osteoarthritis, rheumatoid arthritis, or other joint diseases or injuries.15 The knee implant surgery involves partial knee replacement, total knee replacement, and revision knee replacement. The choice of implant depends on the extent of knee damage and the patient’s specific needs and goals. Knee implant surgery is a major surgical procedure that requires careful planning, preparation, and aftercare. Patients undergoing knee replacement surgery will typically need to participate in physical therapy and rehabilitation programs to regain strength and mobility in the affected knee.
Classification of knee implants
Knee implants can be classified in several ways based on various criteria. Total knee replacement (TKR) involves replacing the entire knee joint with an implant. Partial knee replacement (PKR) involves replacing only the damaged part of the knee joint with an implant. Revision knee replacement involves replacing a previously implanted knee implant with a new one due to wear, loosening, or other complications. Based on the design of the implant, they are classified into fixed-bearing, mobile-bearing, medial-pivot and gender-specific implants. In fixed-bearing, the femoral and tibial components of the implant are fixed in place and move together on a fixed plastic spacer. In case of mobile-bearing, the plastic spacer is designed to move independently of the femoral and tibial components, allowing for more natural knee movement. Medial-pivot are designed to mimic the natural movement of the knee joint, with a medial pivot that allows for rotation and stability. In gender-specific implants, they are designed with anatomical differences between men and women in mind, such as differences in the shape and angle of the knee joint. Based on the materials usage, they are classified into metal-on-plastic, ceramic-on-plastic and metal-on-metal implants. In metal-on-plastic implants, the femoral component is made of metal, and the tibial component is made of plastic. In ceramic-on-plastic implants, the femoral component is made of ceramic, and the tibial component is made of plastic. In metal-on-metal implant, both the femoral and tibial components are made of metal. Implant selection depends on the age of the patient, level of activity, degree of knee damage, and medical history, as well as the preference of surgeon with their experience.
Additive manufacturing for knee implants
AM is also being explored for the production of knee implants.16 AM enables the creation of highly customized knee implants, which can be designed based on a patient’s specific anatomy. This can improve the fit and function of the implant and reduce the risk of implant failure. Traditional methods of producing knee implants can be time-consuming and labor-intensive. AM can reduce production time and costs by allowing for the creation of knee implants in a single process without the need for molds or multiple manufacturing steps. With AM, it is possible to create complex structures with precise geometry that can improve the mechanical properties of the implant. This can lead to improved implant performance and longevity. AM enables the use of a wide range of materials, including metals, ceramics, and polymers, which can be selected based on their specific mechanical and biological properties for each individual patient. AM can significantly reduce waste by only using the necessary material to produce the implant, which can improve the sustainability of the manufacturing process. Overall, AM is a promising technology for the production of knee implants, with potential benefits for both patients and manufacturers. As research continues, AM is likely to become an increasingly popular and effective method for creating customized knee implants. The long-term performance of these implants need to be studied considering the potential risks associated with 3D printing technology.
Recent trends in knee implants
Recent research in knee implants has focused on improving the design and materials used in knee replacement surgery, as well as developing new techniques to improve patient outcomes. Researchers are exploring the use of advanced imaging techniques, such as MRI and CT scans, to create personalized knee implants that fit the patient’s anatomy more closely. This can improve the accuracy of the surgery and reduce the risk of implant failure. 3D printing technology is being used to create knee implants with customized shapes and designs, which can improve the fit and function of the implant. Researchers are also investigating the use of 3D printing to create patient-specific surgical guides, which can improve the accuracy and precision of the surgery. In wear-resistant materials, researchers are developing new materials for knee implants that are more wear-resistant than traditional materials, such as metal and plastic. Robotics-assisted surgery is being used in knee replacement surgery to improve the accuracy and precision of the surgery. Robotic systems can create a 3D model of the patient’s knee, which can guide the placement of the implant with greater accuracy than traditional techniques. Overall, recent research in knee implants is focused on improving the safety, efficacy, and long-term outcomes of knee replacement surgery. These advances may enhance the life for patients with knee osteoarthritis or other knee conditions that require surgery.
Cranioplasty implants
Skull implants, also known as cranial implants, are medical devices used to reconstruct or replace missing or damaged parts of the skull. These implants are typically used to treat patients who have undergone surgery to remove a part of the skull due to injury or disease, or who have congenital abnormalities of the skull.17 Skull implants are usually made of biocompatible materials such as titanium, PMMA polyethylene, or porous polyethylene. The implant is custom-designed to fit the patient’s skull and is placed over the missing or damaged area of the skull.18 The process of creating a skull implant involves taking a CT scan of the patient’s head to create a 3D model of the skull. This model is used to design the implant, which is then manufactured using computer-aided design and manufacturing (CAD/CAM) techniques. Skull implants are typically placed under the scalp and secured with screws or plates.19 The procedure is performed under general anesthesia and may require a hospital stay of a few days. Patients may experience some discomfort and swelling after the procedure, but this typically resolves within a few weeks. Skull implants can improve the appearance of the skull and help protect the brain from injury.20 They can improve the patient’s quality of life by reducing pain and restoring normal skull function.21 However, like any surgical procedure, skull implant surgery carries some risks, including infection, bleeding, and implant failure.
Classification of skull implants
Skull implants can be classified based on several criteria, including the material used, shape, and location.22 Some of the commonly used materials for cranial implants are titanium, polyethylene and porous polyethylene. Titanium is a strong, lightweight, biocompatible metal that is commonly used in skull implant surgery. Polyethylene is a type of plastic that is also biocompatible and commonly used in skull implants. Porous polyethylene is a type of polyethylene that has a porous structure, which allows for better integration with the surrounding bone tissue. Based on the shape of implants, custom-made and pre-fabricated implants are used for fabricating implants. Custom-made skull implants are designed specifically for the individual patient and are made to fit the exact shape of the skull defect. Pre-fabricated skull implants are available in a variety of standard sizes and shapes and are selected by the surgeon to best fit the patient’s skull defect. Calvarial implants are used to reconstruct the upper part of the skull (calvaria), which includes the frontal, parietal, and occipital bones. Orbito-zygomatic implants are used to reconstruct the bones around the eye socket (orbit) and cheekbone (zygoma). Temporal implants are used to reconstruct the temple region of the skull. Custom-made implants are often preferred when the defect is large or complex, while pre-fabricated implants may be sufficient for smaller defects.
Additive manufacturing for skull implants
AM is also being used for the production of skull implants.23 3D printing enables the creation of highly customized skull implants, which can be designed based on a patient’s specific anatomy. This can improve the fit and function of the implant and reduce the risk of implant failure. With 3D printing, it is possible to create skull implants with a high level of accuracy, which can improve the aesthetic appearance of the implant and its integration with surrounding tissues. Traditional methods of producing skull implants can be time-consuming and labor-intensive. AM can reduce production time and costs by allowing for the creation of skull implants in a single process without the need for molds or multiple manufacturing steps. AM enables the use of a wide range of materials24, including metals, ceramics, and polymers, which can be selected based on their specific mechanical and biological properties for each individual patient. AM can significantly reduce waste by only using the necessary material to produce the implant, which can improve the sustainability of the manufacturing process. Overall, AM is a promising technology for the production of skull implants,18 with potential benefits for both patients and manufacturers. However, more research is needed to fully understand the long-term performance of these implants and the potential risks associated with 3D printing technology.
Recent trends in skull implants
Recent research in skull implants has focused on improving the materials and techniques used in cranial reconstruction. 3D printing technology has revolutionized the field of cranial reconstruction by enabling the creation of patient-specific implants with high precision and accuracy. Researchers are exploring various 3D printing techniques and materials, including titanium and biocompatible polymers, to create custom-made skull implants. Bio-active materials are substances that stimulate bone growth and integration with the implant. Researchers are investigating the use of bioactive materials, such as hydroxyapatite, to improve the long-term outcomes of skull implant surgery by promoting bone regeneration and reducing the risk of implant failure. Nano-technology involves the manipulation of materials at the nanoscale level. Researchers are exploring the use of nanotechnology to create skull implants with enhanced mechanical properties, such as greater strength and durability. Virtual reality technology can be used to create a 3D visualization of the patient’s skull defect and plan the surgery in a virtual environment. This can improve the accuracy and safety of the procedure and reduce the risk of complications. Researchers are conducting studies to evaluate the long-term outcomes of skull implant surgery, including the rate of implant failure, infection, and other complications. This research can help identify factors that contribute to successful outcomes and inform best practices for skull implant surgery. Overall, recent research in skull implants is focused on improving the safety, efficacy, and long-term outcomes of cranial reconstruction surgery. These advances have the potential to improve the quality of life for patients who require skull implants due to injury or disease.
Factors to be considered for placing implants
The placement of dental implants needs to consider several factors for sufficient bone density and quality, which are essential for the success of dental implants. Before placing an implant, the dentist will evaluate the bone density and quality of the patient’s jaw using x-rays, CT scans, or other imaging techniques. Healthy gums are critical to the success of dental implants. If the gums are inflamed or infected, the implant may not heal properly or may even fail. The dentist will assess the patient’s gum health and treat any issues before proceeding with implant placement. The force with which a patient bites down can affect the success of dental implants. Patients with a stronger bite force may require stronger or more durable implants to withstand the extra force. The position and angle of adjacent teeth can affect the placement and stability of the implant. The dentist will take into account the position of nearby teeth and their roots to determine the optimal location for the implant. The patient’s overall health and medical history will be evaluated to ensure that they are healthy enough to undergo implant surgery and that there are no contraindications that could affect the success of the implant. The type of implant and material used will depend on the patient’s needs and preferences, as well as the dentist’s recommendations. Parameters such as the shape, size and material of the implant will be considered to ensure the best possible outcome. By considering these and other factors, dentists can ensure that dental implants are placed in the most optimal location and manner for each patient, resulting in the best possible outcomes.
Implant placement methods
There are different implant placement methods used by dentists and oral surgeons. One-stage implant placement method involves placing the implant into the jawbone and attaching the abutment, which connects the implant to the replacement tooth or bridge, at the same time.25 The implant is then left to heal, and the replacement tooth or bridge is attached once the implant has fused to the bone. Two-stage implant placement method involves placing the implant into the jawbone and then covering it with gum tissue, allowing it to heal and integrate with the bone. After a few months, a second surgery is performed to expose the implant and attach the abutment. Immediate implant placement method involves placing the implant immediately after a tooth extraction.26 It can be a one-stage or two-stage procedure, depending on the patient’s needs and the dentist’s recommendation. Guided implant placement method involves using computer-guided technology to plan and place the implant with high accuracy and precision.27 It allows the dentist to create a 3D model of the patient’s mouth and simulate the implant placement, ensuring optimal positioning and avoiding critical structures such as nerves and sinuses. Implant stability is a critical factor for the success of dental implants.28 Implants that are not stable can lead to implant failure, which can cause discomfort, pain, and require additional treatments.29 Implant stability can be affected by various factors,30 including the bone density, implant design, surgical technique, and implant material.
Stability of implants
The primary method of measuring implant stability is by using a tool called a resonance frequency analysis (RFA) device, which measures the implant stability quotient (ISQ). The ISQ scale ranges from 1 to 100, with higher values indicating greater stability.31 An ISQ value of 60 or higher is generally considered to be ideal for implant stability. Implant stability can be assessed by the surgeon during implant placement surgery.32 The surgeon will test the stability of the implant by applying a gentle force to the implant with a torque wrench or other device. The amount of force required to move the implant is an indicator of the implant’s stability.33 Several factors can affect implant stability. These include the patient’s bone density and quality, the surgical technique used, the implant design, and the material used to make the implant.32 Implants made of high-quality materials such as titanium have been shown to provide better stability than implants made of other materials. To ensure optimal implant stability, it’s important to work with an experienced and skilled dentist or oral surgeon.34 They will use the appropriate techniques and materials to provide the best possible outcome for your implant placement. In additional, post-operative procedures need to be followed for promoting implant stability and increase the likelihood of long-term implant success.
Durability of implants
Dental implants are used for long-lasting and durable solution for the absence of teeth. While the durability of dental implants depends on several factors, studies have shown that dental implants have a high success rate, with many lasting for 25 years or more. The durability of dental implants is affected by several factors, including the type of implant, the material used, the patient’s oral hygiene, and any underlying health conditions. Implants made from high-quality materials such as titanium or zirconia have been shown to provide better durability than those made from other materials. The surgical technique used to place the implant can also affect its durability. The implant must be placed in the correct position and angle to ensure that it can withstand the pressure of biting and chewing. The quality and quantity of the patient’s bone also play a critical role in the implant’s durability. If there is insufficient bone or the bone is of poor quality, the implant may be less stable and less durable. With proper care and maintenance, they can provide a natural-looking, functional, and comfortable replacement for missing teeth that can last for many years. It’s important to work with a skilled and experienced dentist or oral surgeon who can assess your individual needs and recommend the best treatment options for your specific situation.
Morphology of implants
Dental implants have a unique morphology designed to replicate the root structure of a natural tooth. The morphology of an implant is designed to maximize osseointegration, or the process by which the implant fuses with the surrounding bone, and to provide stability and support for the replacement tooth or teeth. The morphology of a dental implant can vary depending on the type of implant and the manufacturer. However, most dental implants consist of three parts: the implant fixture, the abutment, and the prosthetic crown or bridge. The implant fixture is the part of the implant that is surgically placed into the jawbone. It is typically made of biocompatible materials such as titanium or zirconia and has a threaded or roughened surface to facilitate Osseo integration. The abutment is the connecting piece that attaches to the implant fixture and provides a platform for the prosthetic crown or bridge. It is typically made of the same material as the implant fixture and can be angled or straight depending on the location of the implant and the position of the replacement tooth. The prosthetic crown or bridge is the visible part of the implant, which almost looks like a natural tooth. It is typically made of materials such as porcelain, ceramic, or composite resin and is custom-made for meeting the patient specification. In addition to the three main parts, some implants may also have additional features such as platform switching or bone level design, which can affect the morphology of the implant and its ability to integrate with the surrounding bone. Overall, the morphology of a dental implant is designed to provide a stable and durable solution for missing teeth that closely replicates the structure and function of a natural tooth35. Your dentist or oral surgeon can help you determine which type of implant is best suited to your individual needs and can provide more information about the specific morphology of the implant.
Results
Medical implants are designed to replace, support, or augment a missing bio-structure.36 Implants can be made from a variety of materials, including metals, ceramics, polymers, and composites, depending on their intended use and the specific requirements of the implant.37 The manufacturing process for medical implants involves several steps, including material selection, design and prototyping, manufacturing, and testing. The materials used to make medical implants must be biocompatible, meaning they do not cause an adverse reaction in the human body. Some commonly used materials include titanium and its alloys, stainless steel, cobalt-chromium alloys, ceramics such as zirconia and alumina, and polymers such as poly ethylene and Poly Ether Ether Ketone (PEEK). These material have their own unique properties, and the material selection depends on its specific applications. Figure 6 shows various types of surgical guides manufactured with different surface finish requirements using material jetting based AM process.
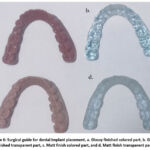 |
Figure 6: Surgical guide for dental implant placement, a. Glossy finished colored part, b. Glossy finished transparent part, c. Matt finish colored part, and d. Matt finish transparent part. |
The manufacturing process for medical implants38 can vary depending on the type of implant and the materials used. Some common methods include computer-aided design (CAD) and computer-aided manufacturing (CAM), 3D printing, and injection moulding. In general, the process involves creating a prototype of the implant, refining the design based on testing and feedback, and then manufacturing the implant using specialized techniques and equipment. Medical implants must undergo rigorous testing to ensure their safety and efficacy. Implant testing can include mechanical and fatigue testing to evaluate the strength and durability of the implant, biocompatibility testing to assess the implant’s interaction with biological tissue, and microbiological testing to evaluate the implant’s ability to resist infection. In addition, the implant must be tested for its ability to integrate with the surrounding tissue and to ensure that it does not cause any adverse reactions or side effects. Overall, the development of medical implants requires a thorough understanding of materials science, engineering, and biology. The selection of appropriate materials, careful design and manufacturing, and thorough testing are all critical components of the implant development process. By using high-quality materials, advanced manufacturing techniques, and rigorous testing protocols, medical implant manufacturers can ensure the safety, reliability, and effectiveness of their products.
Future technologies of medical implants
Medical implants are a rapidly evolving field of technology, and there are several emerging technologies that show promise for the future of implants. 3D printing technology is being used to create customized implants that fit a patient’s unique anatomy.39 This technology enables the creation of complex, patient-specific designs that can improve implant performance and reduce the risk of complications. Nanotechnology is being used to create implants with enhanced properties, such as increased strength, durability, and resistance to infection. Nanoparticles can be added to implant materials to create new properties, such as improved bone growth or enhanced drug delivery. Bioactive coatings are being developed to promote tissue growth and healing around implants. These coatings can improve the integration of the implant with surrounding tissues and reduce the risk of infection and rejection. Implants with wireless communication capabilities can transmit data about implant performance, patient activity, and other health metrics to healthcare providers. This technology can enable more personalized and effective patient care. Self-powered implants that generate their own energy are being developed to reduce the need for battery replacement or external power sources. This technology can improve the longevity and reliability of implants. Overall, these emerging technologies have the potential to improve the performance, durability, and safety of medical implants, and to enhance patient outcomes.40 As these technologies continue to develop, we can expect to see further advancements in the field of medical implants that will improve the lives of patients around the world.
Implant-bone interface and osseointegration
The implant-bone interface is the area where the implant comes into contact with the bone. A stable and long-lasting implant-bone interface is crucial for the success of an implant, as it allows the implant to function properly and integrate with the surrounding bone tissue.41 Osseointegration is a complex biological process that involves the formation of new bone tissue around the implant surface. The implant is typically made of a biocompatible material, such as titanium or a titanium alloy that allows it to integrate with the surrounding bone tissue. When the implant is placed in the bone, it initially forms a fibrous tissue layer around it.42 This layer eventually gives way to new bone tissue that grows around and into the implant surface, creating a strong and stable bond between the implant and the bone. Several factors influence the success of osseointegration, including implant design, surface properties, and surgical technique.43 The surface of the implant is an important factor, as a rough surface can promote bone growth and improve implant stability. Implant surface treatments, such as acid etching or sandblasting, can increase surface roughness and promote osseointegration. Figure 7. shows the open porous cellular structures produced using AM.
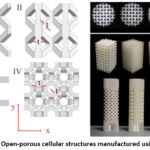 |
Figure 7: Open-porous cellular structures manufactured using AM.44 |
The surgical technique used to place the implant is also critical for the success of osseointegration. The implant must be placed in the bone in such a way that it is surrounded by healthy bone tissue and is not subjected to any sort of excessive mechanical stress. Adequate blood supply is also critical for the success of osseointegration, as it provides the nutrients and oxygen necessary for bone growth. Overall, a stable and long-lasting implant-bone interface is essential for the success of an implant, and osseointegration is a key factor in achieving this stability. Advances in implant design, surface treatments, and surgical techniques are continually improving the success rates of osseointegration, leading to better patient outcomes and increased quality of life for those who receive implants.
Discussion
Choosing the right implant system is an important decision that depends on several factors, including the patient’s anatomy, the surgical technique, and the expected outcome. The material used to manufacture the implant is an important factor, as it affects the implant’s strength, durability, and biocompatibility. Implants are typically made from materials such as titanium or a titanium alloy, cobalt-chromium, or polyethylene. The design of the implant can affect its stability, durability, and performance. The implant design should be tailored to the patient’s specific needs and anatomy to ensure proper fit and function. The surface of the implant can affect osseointegration and long-term stability. Implants with roughened or porous surfaces can promote bone growth and improve osseointegration. The implant system should be compatible with the surgical technique being used. Some implant systems are designed for minimally invasive surgery, while others may require more extensive surgical approaches. The cost of the implant system is an important factor, as it can vary widely depending on the materials used, the design, and the manufacturer. The cost of the implant system should be balanced with the potential benefits to the patient, such as improved function, reduced pain, and increased quality of life. The manufacturer of the implant system should provide adequate support, including training, technical support, and post-operative follow-up. This can help ensure that the implant system is used correctly and that any issues or complications are addressed in a timely manner. Overall, choosing the right implant system depends on a range of factors that must be considered on a case-by-case basis. By carefully considering these factors, surgeons can select the implant system that meets the needs of their patients and improves their outcomes.
Software associated with implant design
There are various software programs that are associated with implant design for dental, knee, and skull implants. Computer-Aided Design (CAD) software is used to design the implant based on the specific anatomy of the patient. The software allows the designer to create a digital model of the implant,45 which can be used to visualize the implant before it is produced. Computer-Aided Manufacturing (CAM) software is used to program the 3D printer or other manufacturing equipment to produce the implant.46 The software translates the digital model of the implant into machine-readable code, which is used to control the manufacturing process. Finite Element Analysis (FEA) software is used to simulate the behavior of the implant under various loads and stresses. FEA can be used to optimize the design of the implant to ensure that it can withstand the forces it will be subjected to in the patient’s body. Imaging software is used to capture images of the patient’s anatomy, such as CT scans or MRIs. The images can be used to create a 3D model of the patient’s anatomy, which can be used to design the implant. Planning and simulation software is used to plan the surgical procedure and simulate the placement of the implant in the patient’s body. The software can help to ensure that the implant is placed in the optimal position and orientation for the best possible outcome. Overall, software plays a critical role in the design and production of implants. The use of advanced software programs can help to ensure that the implant is customized for the patient’s specific needs and can improve the long-term success of the implant.
Advantages of AM over traditional manufacturing
This layered manufacturing process can make customized implants and thereby meeting the specific needs of the patient. In addition, the traditional manufacturing may require machining or fabricating the parts with dies and molds. All these work require longer time and larger amount of cost for part fabrication. Further, these implants promote osseointegration and it will be available in the form of metal implants commercially. Polymer implants with PLA material is on the research and development phase for potential application in human trials. Thus, these layer based AM process may cut down cost and fabrication time for customized implants. In addition, these AM providing design freedom, which overcomes the complexity associated with fabricating parts based on the designer idea and there is less or minimum modifications required as per Design for Additive Manufacturing guidelines.
Commercial industries for implants manufacturing
There are several commercial industries involved in the manufacturing of implants for dental, knee, and skull implants. In dental implant industry, it involves the design and production of dental implants, which are used to replace missing teeth.47 Companies such as Straumann, Nobel Biocare, and Dentsply Sirona are some of the leading players in this industry. Knee implant industry involves the design and production of knee implants, which are used to replace damaged knee joints.48 Companies such as Zimmer Biomet, Stryker, and Smith & Nephew are some of the leading players in this industry. Skull implant industry involves the design and production of skull implants, which are used to repair or replace damaged portions of the skull.49 Companies such as KLS Martin, Medtronic, and Johnson & Johnson are some of the leading players in this industry. Implant manufacturing equipment industry involves the production of equipment and machinery used to manufacture implants.50 Companies such as Renishaw, EOS, and 3D Systems are some of the leading players in this industry. Material supplier industry involves the production and supply of materials used to manufacture implants, such as metals,51 ceramics, and polymers. Companies such as Carpenter Technology, Heraeus, and DSM are some of the leading players in this industry. Overall, there are several commercial industries used for the design and production of implants,52 which contribute to the development and availability of these life-changing medical devices.
Conclusion
The future of implants is very promising, with ongoing research and development aimed at improving the design, production, and performance of implants for dental, knee, and skull applications. Here are some potential future developments in the field of implants:
With advancements in technology such as additive manufacturing and 3D printing, it may become possible to produce implants that are even more customized to the patient’s specific anatomy, resulting in better fit and function.
The use of new biomaterials, smart materials, and mimicking the natural properties of materials such as bone and tissue, may lead to better integration of the implant with the patient’s body and improved long-term performance.
The development of minimally invasive surgical techniques, such as robotic surgery and navigation systems, may result in reduced surgical trauma and faster recovery times for patients.
Understanding the implant materials and designing for customer needs, may lead to implants that have better wear resistance, reducing the need for revision surgeries.
Sensors and wireless communication technologies may enable the use of technologies such as remote monitoring and early detection of problems in the implants.
Overall, the future of implants looks very promising, with the potential for significant improvements in customization, biomaterials, surgical techniques, wear resistance, and remote monitoring.
Acknowledgement
The authors would like to thank the management of Sathyabama Institute of Science and Technology for providing all the essential infrastructure to carry out this research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable.
Author contributions
- Madhanagopal Manoharan – Conceptualization, research, writing and drafting.
- Bhavadharani Babu – Data collection, analysis and writing,
- Sindu Divakaran – Visualization, writing,
- Gobi saravanan Kaliaraj – supervision, Funding Acquisition, Resources,
- Perumal Sudalai – Methodology, analysis,
- Rajendra Thilahar Chelladurai – Supervision, Resources.
References
- Honigmann P, Sharma N, Okolo B, Popp U, Msallem B, Thieringer F. M, Patient-specific surgical implants made of 3D printed PEEK: Material, technology, and scope of surgical application, Biomed Res. Int., 2018; 4520636.
CrossRef - Sheoran A. J, Kumar H, Arora P. K, Moona G, Bio-medical applications of additive manufacturing: A review, Procedia Manuf. 51, 2020; 663–670.
CrossRef - Saha M, Additive Manufacturing and Characterisation of Biomedical Materials, SSRN Electron. J., 2022; 9781003286806.
CrossRef - Hoang D, Perrault D, Stevanovic M, Ghiassi A, Today surgical applications of three-dimensional printing: A review of the current literature & how to get started, Ann. Transl. Med. 4; 2016.
CrossRef - Calvo-Haro J A, Pascau J, Mediavilla-Santos L, et al. Conceptual evolution of 3D printing in orthopedic surgery and traumatology: from “do it yourself” to “point of care manufacturing,” BMC Musculoskelet. Disord. 22, 2021; 1–10.
CrossRef - Garner E, Kolken H.M.A, Wang C.C.L, Zadpoor A.A., Wu J, Compatibility in microstructural optimization for additive manufacturing, Addit. Manuf. 26, 2019; 65–75.
CrossRef - Emelogu A, Marufuzzaman M, Thompson S. M, Shamsaei N, Bian L, Additive manufacturing of biomedical implants: A feasibility assessment via supply-chain cost analysis, Addit. Manuf. 11, 2016; 97–113.
CrossRef - Kumar R, Kumar M, Chohan J. S, The role of additive manufacturing for biomedical applications: A critical review, J. Manuf. Process. 64, 2021; 828–850.
CrossRef - Mohseni M, Bas O, Castro N.J, Schmutz B., Hutmacher D.W, Additive bio-manufacturing of scaffolds for breast reconstruction, Addit. Manuf. 30; 2019.
CrossRef - Warreth A, Ibieyou N, O’Leary R.B, Cremonese M, Abdulrahim M, Dental implants: An overview, Dent. Update. 44, 2017; 596–620.
CrossRef - Santosh kumar M, Ravikumar Y, Alwala A. M, A 3D Printed Osseointegrated Combined Jaw and Dental Implant Prosthesis – A Case Study, Rapid Proto. J, 2016; 23(6), 1164-1169.
CrossRef - Culmone C, Smit G, Breedveld P, Additive manufacturing of medical instruments: A state-of-the-art review, Addit. Manuf. 27, 2019; 461–473.
CrossRef - Manmadhachary A, Ravi Kumar Y, Krishnanand L, Improve the accuracy, surface smoothing and material adaption in STL file for RP medical models, J. Manuf. Process. 21, 2016; 46–55.
CrossRef - Morales-Gómez J A, Garcia-Estrada E, Leos-Bortoni J. E, et al., Cranioplasty with a low-cost customized poly methyl methacrylate implant using a desktop 3D printer, J. Neurosurg. 130, 2019; 1721–1727.
CrossRef - Kumar L, Haleem A, Javaid M, Impact of three dimensional printing in orthopedics, Glob. Heal. J. 5, 2021; 178–182.
CrossRef - Eldesouky I, Harrysson O, West H, Elhofy H, Electron beam melted scaffolds for orthopedic applications, Addit. Manuf. 17, 2017; 169–175.
CrossRef - Jindal S, Manzoor F, Haslam N, Mancuso E, 3D printed composite materials for craniofacial implants: current concepts, challenges and future directions, Int. J. Adv. Manuf. Technol. 112, 2021; 635–653.
CrossRef - da Silva Júnior E.B, de Aragão A.H, de Paula Loureiro M, et al., Cranioplasty with three-dimensional customised mould for poly methyl methacrylate implant: a series of 16 consecutive patients with cost-effectiveness consideration, 3D Print. Med. 7, 2021; 1–11.
CrossRef - Schön S.N, Skalicky N, Sharma N, Zumofen D.W, Thieringer F.M, 3D-Printer-Assisted Patient-Specific Polymethyl Methacrylate Cranioplasty: A Case Series of 16 Consecutive Patients, World Neurosurg. 148, 2021; e356–e362.
CrossRef - Yerragunta T, Kanala R.R, Yerramneni V.K, Kolpakawar S, Rangan V, Designer Cranioplasty at Budget Prices: A Novel Use of 3D Printing Technology, Indian J. Neurosurg. 10, 2021; 194–198.
CrossRef - Mohsen A.M.R, Mohamed Gabr M.D, The Use of Prefabricated 3D Printed PEEK Implants for Repair of Skull Defects, Med. J. Cairo Univ. 90, 2022; 345–350.
CrossRef - Pöppe J.P, Spendel M, Schwartz C., Winkler P.A, Wittig J, The “springform” technique in cranioplasty: custom made 3D-printed templates for intraoperative modelling of polymethylmethacrylate cranial implants, Acta Neurochir. (Wien). 164, 2022; 679–688.
CrossRef - Park H, Choi J.W, Jeong W.S, Clinical Application of Three-Dimensional Printing of Polycaprolactone/Beta-Tricalcium Phosphate Implants for Cranial Reconstruction, J. Craniofac. Surg. 33, 2022; 1394–1399.
CrossRef - Czyżewski W, Jachimczyk J, Hoffman Z, et al., Low-Cost Cranioplasty—A Systematic Review of 3D Printing in Medicine, Materials (Basel). 15, 2022.
CrossRef - Wang X, Shaheen E, Shujaat S, et al., Influence of experience on dental implant placement: an in vitro comparison of freehand, static guided and dynamic navigation approaches, Int. J. Implant Dent. 8, 2022.
CrossRef - Ebenezer V, Balakrishnan K, Asir R.V.D, Sragunar B, Immediate placement of endosseous implants into the extraction sockets, J. Pharm. Bioallied Sci. 7, 2015; S234–S237.
CrossRef - John A.V, Abraham G, Alias A, Two-visit CAD/CAM milled dentures in the rehabilitation of edentulous arches: A case series, J. Indian Prosthodont. Soc. 19, 2019; 88–92.
CrossRef - Al Sawai A.A, Rajmohan C.S, Labib H, Tabiuk S, Advances in Dental Implant Positioning Techniques and Their Clinical Implications, Open J. Stomatol. 07, 2017; 121–135.
CrossRef - Mistry A, Ucer C, Thompson J.D, Khan R.S, Karahmet E, Sher F, 3D guided dental implant placement: Impact on surgical accuracy and collateral damage to the inferior alveolar nerve, Dent. J. 9(9), 2021; 99.
CrossRef - Afshari A, Shahmohammadi R, Mosaddad S.A, et al., Free-Hand versus Surgical Guide Implant Placement, Adv. Mater. Sci. Eng. 2022; 2022.
CrossRef - Vollmer A, Saravi B, Lang G, et al., Factors influencing primary and secondary implant stability—a retrospective cohort study with 582 implants in 272 patients, Appl. Sci. 10, 2020; 1–14.
CrossRef - Al-Zubaidi S.M, Madfa A.A, Mufadhal A.A, Aldawla M.A, Hameed O.S, Yue X.G, Improvements in Clinical Durability from Functional Biomimetic Metallic Dental Implants, Front. Mater. 7, 2020.
CrossRef - Ivanova V, Chenchev I, Zlatev S, Mijiritsky E, Correlation between primary, secondary stability, bone density, percentage of vital bone formation and implant size, Int. J. Environ. Res. Public Health, 18, 2021.
CrossRef - Mistry G, Shetty O, Shetty S, Singh R, Measuring implant stability: A review of different methods, J. Dent. Implant. 4, 2014; 165.
CrossRef - Bandela V, Munagapati B, Komala J, Basany R.B, Patil S.R, Kanaparthi S, Evaluating the primary stability of implants by two different insertion methodsin compromised bone – A pilot study, Biomed. Pharmacol. J., 13, 2020; 1833–1838.
CrossRef - Zita Gomes R, De Vasconcelos M.R, Lopes Guerra I.M, De Almeida R.A.B, De Campos Felino A.C, Implant Stability in the Posterior Maxilla: A Controlled Clinical Trial, Biomed Res. Int. 2017, 2017; 6825213.
CrossRef - Kittur N, Oak R, Dekate D, Jadhav S, Dhatrak P, Dental implant stability and its measurements to improve osseointegration at the bone-implant interface: A review, Mater. Today Proc. 43, 2020; 1064–1070.
CrossRef - Juneja M, Thakur N, Kumar D, Gupta A, Bajwa B, Jindal P, Accuracy in dental surgical guide fabrication using different 3-D printing techniques, Addit. Manuf. 22, 2018; 243–255.
CrossRef - Wixted C.M, Peterson J.R, Kadakia R.J, Adams S.B, Three-dimensional Printing in Orthopaedic Surgery: Current Applications and Future Developments, J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 5, 2021; 00230–00211.
CrossRef - Li B, Zhang M, Lu Q, et al., Application and Development of Modern 3D Printing Technology in the Field of Orthopedics, Biomed Res. Int. 2022; 8759060.
CrossRef - Swami V, Vijayaraghavan V, Swami V, Current trends to measure implant stability, J. Indian Prosthodont. Soc. 16, 2016; 124–130.
CrossRef - Huang H, Wu G, Hunziker E, The clinical significance of implant stability quotient (ISQ) measurements: A literature review, J. Oral Biol. Craniofacial Res. 10, 2020; 629–638.
CrossRef - Staedt H, Kämmerer P.W, Goetze E, Thiem D.G.E, Al-Nawas B, Heimes D, Implant primary stability depending on protocol and insertion mode — an ex vivo study, Int. J. Implant Dent. 6, 2020; 6(1):49.
CrossRef - Limmahakhun S, Oloyede A, Sitthiseripratip K, Xiao Y, Yan C, 3D-printed cellular structures for bone biomimetic implants, Addit. Manuf. 15, 2017; 93–101.
CrossRef - Kernen F, Kramer J, Wanner L, Wismeijer D, Nelson K, Flügge T, A review of virtual planning software for guided implant surgery – Data import and visualization, drill guide design and manufacturing, BMC Oral Health. 20, 2020; 1–10.
CrossRef - Abdudeen A, Abu Qudeiri J.E, Kareem A, Valappil A.K, Latest Developments and Insights of Orthopedic Implants in Biomaterials Using Additive Manufacturing Technologies, J. Manuf. Mater. Process. 6, 2022; 162.
CrossRef - Stricker A, Bergfeldt T, Fretwurst T, et al., Impurities in commercial titanium dental implants – A mass and optical emission spectrometry elemental analysis, Dent. Mater. 38, 2022; 1395–1403.
CrossRef - Petrovic V, Vicente J, Ramn J, Portols L, Additive Manufacturing Solutions for Improved Medical Implants, Biomedicine. 2012.
CrossRef - Akmal J.S, Salmi M, Mäkitie A, Björkstrand R, Partanen J, Implementation of industrial additive manufacturing: Intelligent implants and drug delivery systems, J. Funct. Biomater. 9, 2018.
CrossRef - Prakash D, Davis R, Kumar Sharma A, Design and fabrication of dental implant prototypes using additive manufacturing, IOP Conf. Ser. Mater. Sci. Eng., 561, 2019.
CrossRef - Bai S.Q, Long G, Cheng C, et al., Additive Manufacturing of Customized Metallic, Metals (Basel). 9, 2019; 1004.
CrossRef - Huang S, Wei H, Li D, Additive manufacturing technologies in the oral implant clinic: A review of current applications and progress, Front. Bioeng. Biotechnol. 11, 2023; 1–16.
CrossRef







