Manuscript accepted on :23-07-2025
Published online on: 25-08-2025
Plagiarism Check: Yes
Reviewed by: Dr. Joyeeta Bhattacharya
Second Review by: Dr. Manas Das
Final Approval by: Dr. Eman Refaat Youness
Monisha Prasad1 , Sulthan Al Rashid2
, Sulthan Al Rashid2 , Vinjmur Ragavan Yashvanthan2*
, Vinjmur Ragavan Yashvanthan2* , Bruhan Math Prakash3
, Bruhan Math Prakash3 and Malarveni Damodaran Lakshmi Priya4
and Malarveni Damodaran Lakshmi Priya4
1Center for Global Health Research, Saveetha Medical College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Saveetha University, Chennai, India
2Department of Pharmacology, Saveetha Medical College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Saveetha University, Chennai, India
3Department of Pharmacology, Andaman and Nicobar Islands, Institute of Medical Sciences, India
4Department of Biochemistry, Auxilium College (Autonomous),Vellore, Tamil Nadu, India
Corresponding Author Email: ysvanth12@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3217
Abstract
This review explores the mechanistic role of finerenone, a selective nonsteroidal mineralocorticoid receptor (MR) antagonist, in the management of hypertension. Finerenone exerts its antihypertensive effects by selectively targeting MR pathways, thereby modulating blood pressure and offering substantial cardiorenal protection. By inhibiting aldosterone activity within the renin–angiotensin–aldosterone system, it reduces sodium and water retention, oxidative stress, fibrosis, and inflammation in renal and cardiovascular tissues. This narrative review is based on a comprehensive analysis of peer-reviewed preclinical and clinical studies retrieved from databases such as PubMed and Scopus, using terms including “finerenone,” “hypertension,” “mineralocorticoid receptor antagonists,” and “cardiorenal protection.” The review highlights finerenone’s superior efficacy and safety profile compared to traditional MR antagonists such as spironolactone. These findings underscore its potential to reduce blood pressure and prevent hypertension-induced organ damage with fewer adverse effects. The dual action of finerenone in blood pressure regulation and organ protection positions it as a promising therapeutic candidate. Ongoing research is warranted to further optimize its clinical applications in hypertension and related complications.
Keywords
Antihypertensive; Blood Pressure regulation; Cardiovascular; Finerenone; Hypertension; Mineralocorticoid Receptor; Renal Protection; Renin-angiotensin-aldosterone system
Download this article as:| Copy the following to cite this article: Prasad M, Al-Rashid S, Yashvanthan V. R, Prakash B. M, Priya M. D. L. Mechanistic Understanding of Finerenone in Hypertension Management: A Narrative Review. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Prasad M, Al-Rashid S, Yashvanthan V. R, Prakash B. M, Priya M. D. L. Mechanistic Understanding of Finerenone in Hypertension Management: A Narrative Review. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/465dym1 |
Introduction
Hypertension is recognized as a major global health concern due to its strong association with cardiovascular morbidity and mortality, posing a significant challenge to public health systems worldwide.1 Finerenone, a nonsteroidal mineralocorticoid receptor (MR) antagonist with distinct mechanistic properties, has emerged from recent advances in therapeutic strategies aimed at more effective hypertension management. A more comprehensive understanding of finerenone’s role in blood pressure regulation is developing as researchers explore its molecular and cellular mechanisms.2,3
The mineralocorticoid receptor plays a crucial role in maintaining salt and water balance, making it an important pharmacological target for antihypertensive therapy. Unlike traditional MR antagonists such as spironolactone, which exhibit broader receptor interactions and are associated with undesirable side effects, finerenone demonstrates high receptor selectivity. This enables more targeted MR modulation, thereby enhancing therapeutic efficacy while minimizing adverse effects.3-5
Finerenone exerts its antihypertensive action by inhibiting aldosterone, a key hormone in the renin–angiotensin–aldosterone system (RAAS). This inhibition reduces sodium retention, oxidative stress, fibrosis, and inflammation in renal and cardiovascular tissues.2,4 These effects extend beyond simple blood pressure control, offering meaningful protection against cardiac and renal damage. Through its anti-inflammatory and antifibrotic actions, finerenone addresses both the hemodynamic and structural consequences of hypertension.5,6
Clinical trials have played a pivotal role in demonstrating the efficacy and safety of finerenone. By elucidating the molecular pathways influenced by the drug, these studies highlight its potential for use in tailored therapeutic regimens. Given its unique mechanism of action and favourable safety profile, finerenone shows promise in combination therapy approaches aimed at optimizing hypertension control.4-6
This review aims to synthesize the current body of literature by examining the mechanistic underpinnings of finerenone’s effects in hypertension. It seeks to provide a clear and comprehensive overview of how finerenone modulates blood pressure and protects the cardiorenal system, thereby contributing to the ongoing discourse on its potential to reshape hypertension management and improve patient outcomes.
Literature search strategy
A comprehensive literature search was conducted to identify relevant preclinical and clinical studies on finerenone in hypertension, focusing on mechanistic insights, efficacy, and safety. The primary sources were obtained from databases like PubMed and Scopus using the following search terms: “finerenone,” “hypertension,” “mineralocorticoid receptor antagonists,” and “cardiorenal protection.” Relevant studies were selected based on their focus on molecular insights, therapeutic efficacy, and safety profiles of finerenone in comparison with traditional antihypertensive treatments
Hypertension
Hypertension, or high blood pressure, is a chronic condition characterized by persistently elevated blood pressure levels. It remains one of the leading global health concerns and is a major risk factor for cardiovascular events such as myocardial infarction and stroke.7 The condition typically develops gradually and often remains asymptomatic until significant complications arise. Common contributing factors include genetic predisposition, unhealthy dietary habits, physical inactivity, obesity, smoking, and excessive alcohol consumption. Early diagnosis and appropriate management are essential to reduce the risk of associated complications and preserve long-term cardiovascular health.7,8
Blood pressure is measured in millimeters of mercury (mmHg) and includes two components: systolic pressure (during heart contraction) and diastolic pressure (during heart relaxation). Hypertension is generally defined as a blood pressure reading equal to or greater than 130/80 mmHg.9 If left untreated, chronic hypertension can cause damage to target organs such as the heart, kidneys, brain, and vasculature, increasing the likelihood of developing coronary artery disease, heart failure, stroke, and chronic kidney disease.10
Hypertension is broadly categorized as either primary (essential), with no identifiable underlying cause, or secondary, which results from conditions such as renal disease or endocrine disorders. Because blood pressure fluctuates throughout the day, consistent and accurate monitoring is crucial for diagnosis and treatment.7-9
Pharmacological interventions form the cornerstone of hypertension management. Treatment regimens are often tailored to the patient’s age, comorbid conditions, and overall cardiovascular risk. Combination therapies are commonly employed to achieve better blood pressure control and minimize adverse effects. Continued advancements in pharmacological research have led to the development of newer antihypertensive agents and therapeutic strategies aimed at improving clinical outcomes.9
Current Pharmacological interventions in Hypertension
Pharmacological therapy plays a pivotal role in the management of hypertension by effectively reducing blood pressure and lowering the risk of cardiovascular events. A wide range of antihypertensive drug classes is available, each acting through distinct mechanisms to regulate blood pressure.11 The choice of medication is typically individualized, considering patient-specific factors such as age, comorbid conditions, and potential side effects.
The following table 1 provides an overview of the major classes of antihypertensive drugs, their mechanisms of action, and commonly associated adverse effects:
| S.no | Drug Class | Mechanism of Action | Side Effects |
| 1 | ACE Inhibitors | Inhibit the angiotensin-converting enzyme (ACE), leading to vasodilation and reduced fluid retention.12,13 | Hyperkalemia, dry cough |
| 2 | Angiotensin II Receptor Blockers (ARBs) | Block angiotensin II receptors, promoting vasodilation and reduced sodium and water retention.13,14 | Hyperkalemia |
| 3 | Calcium Channel Blockers (CCBs) | Inhibit calcium entry into vascular and cardiac smooth muscle cells, leading to vasodilation and reduced myocardial contractility.15,16 | Peripheral edema, dizziness, headache |
| 4 | Beta-Blockers | Inhibit catecholamine binding to β-adrenergic receptors, reducing heart rate and myocardial contractility.17,18 | Bradycardia, bronchospasm (notably with non-selective agents), fatigue, hypotension |
| 5 | Diuretics | Promote renal excretion of sodium and water, thereby reducing blood volume and systemic vascular resistance.19,20 | Electrolyte disturbances (e.g., hypokalaemia, hyponatremia), dehydration, polyuria, hypotension |
| 6 | Alpha Blockers | lock α-adrenergic receptors, resulting in vasodilation and reduced peripheral resistance.21 | Orthostatic hypotension, dizziness, fatigue |
| 7 | Renin Inhibitors | Inhibit renin activity, thereby reducing the conversion of angiotensinogen to angiotensin I and subsequent production of angiotensin II.22 | Diarrhoea, Hypotension, and Hyperkalaemia |
Finerenone
Finerenone is a selective, nonsteroidal mineralocorticoid receptor (MR) antagonist currently under evaluation for its potential to reduce cardiovascular and renal complications, particularly in individuals with diabetes and chronic kidney disease (CKD). It has the chemical formula C₂₁H₂₂N₄O₃ and is characterized by low water solubility. It is available in 10 mg and 20 mg oral tablet formulations.23 Compared to traditional steroidal MR antagonists such as spironolactone and eplerenone, finerenone offers a more favourable safety profile, with a lower risk of hyperkalemia and acute reductions in estimated glomerular filtration rate (eGFR).6,24
Multiple large-scale clinical trials—including FIDELIO-DKD, FIGARO-DKD, ARTS-HF, and ARTS-DN—have demonstrated the clinical efficacy of finerenone in reducing albuminuria, improving cardiovascular outcomes, and slowing the progression of diabetic kidney disease.25 Finerenone acts by antagonizing the effects of mineralocorticoids such as aldosterone and cortisol, thereby reducing inflammation and fibrosis in renal and cardiac tissues. Its high receptor selectivity and minimal hemodynamic effects translate into a lower incidence of hyperkalemia and reduced treatment discontinuation rates when compared to spironolactone. Moreover, it avoids common adverse effects such as gynecomastia and excessive eGFR decline, making it particularly suitable for patients with diabetes and renal impairment.26
Preclinical studies further highlight finerenone’s advantages, including optimal tissue distribution in the heart and kidneys, a short half-life, and the absence of active metabolites. It has been shown to reduce profibrotic cytokine expression and suppress tenascin gene activity, leading to reductions in biomarkers such as cardiac troponin T and B-type natriuretic peptide (BNP) in models of pressure overload. Unlike steroidal MRAs, finerenone functions as an inverse agonist, inhibiting co-regulator recruitment even in the absence of aldosterone, thereby enhancing its cardioprotective and anti-inflammatory potential.25,26
In patients with heart failure with reduced ejection fraction (HFrEF), finerenone has demonstrated superior tolerability compared to eplerenone, with lower rates of hyperkalemia, reduced cardiovascular mortality, and fewer adverse cardiac events. These findings underscore its promise as a novel therapeutic agent for managing renal and cardiovascular complications in patients with diabetes and CKD.26,27
Finerenone role in cardiovascular complications
Finerenone is a novel, nonsteroidal, and selective mineralocorticoid receptor antagonist (MRA) developed to address a range of cardiovascular and renal disorders, particularly in patients with chronic kidney disease (CKD) associated with type 2 diabetes mellitus (T2DM). It specifically inhibits the mineralocorticoid receptor (MR), which mediates the effects of aldosterone—a hormone that promotes sodium and water retention and contributes to inflammation and fibrosis in multiple organs, including the heart and kidneys. By antagonizing MR activation, finerenone helps to mitigate these deleterious effects, thereby potentially improving cardiovascular and renal outcomes.27,28
Finerenone has been extensively studied for its therapeutic potential in cardiovascular disease, with a particular focus on its renoprotective and cardioprotective effects in patients with CKD and T2DM. Major clinical trials, such as FIDELIO-DKD and FIGARO-DKD, have assessed its efficacy in this population. These studies demonstrated that finerenone significantly reduced the risk of kidney failure, delayed the progression of eGFR decline, and lowered the incidence of renal and cardiovascular events.29,30
Like all pharmacological agents, finerenone is associated with potential side effects. The most commonly reported adverse effect in clinical trials is hyperkalemia, particularly in patients with pre-existing renal dysfunction or those concurrently taking other potassium-elevating medications. As such, the use of finerenone may be contraindicated in individuals with severe renal impairment, baseline hyperkalemia, or those on potassium-sparing diuretics. Clinicians are advised to evaluate patient-specific risk factors and comorbidities before initiating therapy.
Despite these considerations, finerenone represents a promising advancement in the treatment of patients with CKD and T2DM. Its novel mechanism of selective MR antagonism provides an alternative strategy to mitigate both cardiovascular and renal risk factors, enhancing the therapeutic landscape for these high-risk populations.27-31
Mechanistic insights of finerenone in hypertension
The antihypertensive effects of finerenone are primarily attributed to its selective inhibition of the mineralocorticoid receptor (MR) and its subsequent modulation of the renin–angiotensin–aldosterone system (RAAS) (Figure 1). A detailed understanding of finerenone’s mechanistic actions provides critical insights into its role in blood pressure regulation, particularly in patients with hypertension.
Mineralocorticoid Receptor Blockade
Aldosterone is a key mineralocorticoid hormone responsible for regulating the balance of potassium (K⁺) and sodium (Na⁺) across epithelial tissues, including those in the gastrointestinal tract, salivary glands, and the distal convoluted and connecting tubules of the kidneys. Beyond its role in epithelial tissues, aldosterone also exerts pathophysiological effects in non-epithelial tissues, including the heart, kidneys, and vascular system.
Excessive aldosterone activity promotes sodium and water retention, leading to increased intravascular volume and elevated blood pressure. It also contributes to structural and functional alterations in cardiovascular and renal tissues. These changes are largely mediated through the MR, which, when activated, exacerbates inflammation, fibrosis, and vascular remodeling.
Finerenone, as a selective MR antagonist, binds to and inhibits the activation of MR by aldosterone. This inhibition prevents the downstream effects of aldosterone, including salt and water retention, thereby reducing blood volume and lowering blood pressure.32 By attenuating MR activation, finerenone not only improves hemodynamic parameters but also confers protective effects on target organs affected by hypertension, particularly the heart and kidneys.33
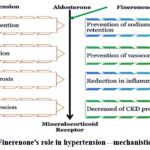 |
Figure 1: Finerenone’s role in hypertension—mechanistic overviewClick here to view Figure |
Finerenone enhances blood pressure regulation, vascular flow, and tissue integrity by reducing sodium retention, vasoconstriction, inflammation, and fibrosis. Additionally, it decreases albuminuria and slows the progression of chronic kidney disease (CKD).
Aldosterone-Mediated Vasoconstriction
Aldosterone influences the vascular endothelium through both short-term, non-genomic effects and long-term, genomic mechanisms. These actions play a critical role in regulating vascular tone and peripheral resistance. One of the key features of aldosterone-induced vasculopathy is the reduction in endothelial nitric oxide (NO) production and bioavailability, accompanied by an increase in superoxide radical formation, which degrades endogenous NO. This imbalance impairs endothelial function and promotes vasoconstriction, contributing to elevated blood pressure.34
Finerenone mitigates these effects by selectively blocking MR activation, thereby counteracting aldosterone-induced endothelial dysfunction. Through MR inhibition, finerenone enhances NO-mediated vasodilation and improves vascular compliance. These vasodilatory effects offer therapeutic benefits in conditions characterized by vascular dysfunction, including hypertension and related cardiovascular disorders.35
Anti-Inflammatory and Anti-Fibrotic Effects
In addition to its primary role in regulating fluid and electrolyte balance via aldosterone signaling in the kidneys, the mineralocorticoid receptor (MR) also plays important roles in vascular homeostasis and tissue repair. However, MR overactivation—whether through elevated aldosterone levels, excessive MR expression, or ligand-independent mechanisms such as cortisol signaling—can lead to pathological outcomes. This overactivation triggers the upregulation of pro-inflammatory and pro-fibrotic gene expression, resulting in progressive organ dysfunction, particularly within the renal and cardiovascular systems.36
In the kidneys, sustained MR activation contributes to tubulointerstitial injury, glomerulosclerosis, renal fibrosis, reduced glomerular filtration rate (GFR), and proteinuria. In the heart, it promotes oxidative stress, inflammatory infiltration, myocardial fibrosis, and hypertrophic remodeling. Vascular consequences of MR overactivation include aberrant vascular remodeling, endothelial dysfunction, and increased arterial stiffness.
Finerenone, due to its high MR selectivity, has been shown to effectively counter these fibrotic and inflammatory pathways. It inhibits profibrotic signaling in cardiac fibroblasts and downregulates MR overexpression in experimental models. Notably, in murine models with constitutively active Rac1—a condition that mimics pathological MR activation—finerenone reduced ventricular dilatation and myocardial fibrosis, thereby attenuating structural cardiac remodeling.37
These findings underscore finerenone’s potential as a disease-modifying agent. By dampening MR-mediated inflammation and fibrosis, finerenone may protect both the heart and vasculature from chronic hypertensive injury. Its anti-fibrotic and anti-inflammatory effects offer a promising therapeutic strategy to prevent maladaptive remodeling associated with long-term hypertension.38
Renal effects
The kidneys play a central role in regulating fluid balance and blood pressure, and impaired renal function is both a cause and consequence of hypertension. Elevated blood pressure accelerates renal dysfunction, while compromised renal function exacerbates hypertension, creating a self-perpetuating cycle that characterizes hypertensive kidney disease. Beyond its hemodynamic influence, the mineralocorticoid receptor (MR) has been increasingly recognized as a key mediator in the pathogenesis of end-organ damage, including chronic kidney disease (CKD) and heart failure.39
In individuals with metabolic syndrome, adipocyte-derived factors can aberrantly stimulate adrenal aldosterone production—particularly in the context of high salt intake. This inappropriate aldosterone secretion contributes to the development of salt-sensitive hypertension, a condition marked by elevated blood pressure secondary to sodium retention. Over time, chronic hypertension leads to progressive damage to both cardiac and renal tissues. There is growing evidence that the aldosterone/MR signaling axis plays a critical role in mediating these effects, especially under conditions of excessive dietary sodium.40
Finerenone, through its selective suppression of MR activity, has demonstrated significant potential in improving renal outcomes. By antagonizing MR-mediated pathways, finerenone reduces the deleterious effects of aldosterone on renal structures. Clinically, it has been shown to decrease urinary albumin excretion (albuminuria) and slow the progression of CKD.41 These effects represent an important therapeutic advancement in the treatment of hypertension-related renal damage, offering improved renal protection and long-term disease modification for high-risk patient populations.
Discussion
Finerenone, a nonsteroidal mineralocorticoid receptor antagonist (MRA), represents an important advancement in antihypertensive therapy due to its high receptor selectivity and distinct pharmacological profile. Unlike traditional MRAs such as spironolactone and eplerenone, finerenone minimizes off-target hormonal interactions and adverse endocrine effects by selectively antagonizing the mineralocorticoid receptor (MR), which is implicated in aldosterone-mediated sodium retention, vascular inflammation, and fibrosis.
Finerenone distinguishes itself by its ability to simultaneously regulate blood pressure and safeguard target organs, especially the heart and kidneys. This dual action addresses the fundamental pathophysiological mechanisms of hypertension, which include oxidative stress, endothelial dysfunction, and tissue remodeling. Preclinical studies have confirmed its ability to suppress MR-driven expression of pro-inflammatory and profibrotic genes, and these findings have translated into measurable clinical benefits.
Major trials such as FIDELIO-DKD and FIGARO-DKD demonstrated that finerenone significantly reduces systolic blood pressure (SBP), albuminuria, and cardiovascular event rates in patients with chronic kidney disease (CKD) and type 2 diabetes mellitus (T2DM). Interestingly, even with a short half-life, its 24-hour BP-lowering effect persisted with once-daily dosing—suggesting durable receptor modulation beyond mere pharmacokinetic duration.42 In patients with resistant hypertension (RH) and moderate-to-severe chronic kidney disease (CKD), finerenone has shown a more favourable safety and efficacy profile compared to spironolactone combined with patiromer or placebo. Specifically, it was associated with a lower incidence of hyperkalemia, greater reduction in urinary albumin-to-creatinine ratio (UACR), and slower progression of renal dysfunction.43 Importantly, finerenone is approved for use in patients with type 2 diabetes mellitus (T2DM) and CKD at estimated glomerular filtration rates (eGFR) as low as 25 mL/min/1.73 m², extending its applicability to those at highest risk of cardiorenal complications.44
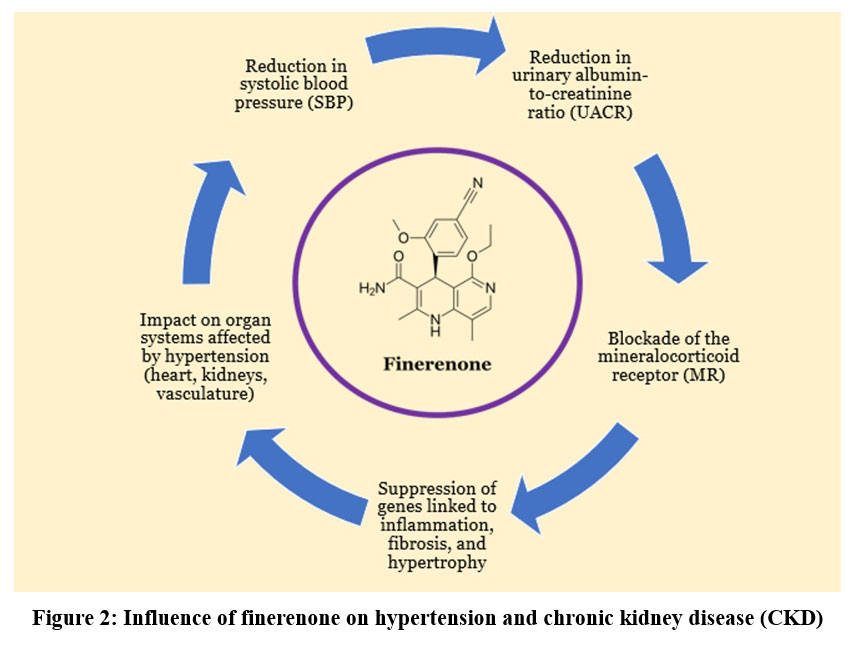 |
Figure 2 Influence of finerenone on hypertension and chronic kidney disease (CKD)Click here to view Figure |
As illustrated in Figure 2, finerenone exerts beneficial effects across multiple domains, including improvements in organ function, reductions in UACR, systolic blood pressure (SBP), and a lower risk of hyperkalemia, supported by both clinical and preclinical studies.
From a comparative standpoint, traditional steroidal MRAs such as spironolactone and eplerenone have demonstrated long-established benefits, particularly in patients with heart failure and reduced ejection fraction (HFrEF).45 However, spironolactone’s known associations with gynecomastia, hyperkalemia, and metabolic disturbances (such as increased HbA1c and cortisol levels) limit its acceptability in patients with T2DM and CKD.46 Finerenone, in contrast, has demonstrated comparable or superior efficacy in clinical endpoints without worsening glycemic control or eliciting steroid-like side effects.47
The potential clinical scope of finerenone continues to expand. Evidence supports its evaluation across diverse indications—including hypertension in CKD, essential hypertension, primary hyperaldosteronism, diabetic nephropathy, nephrotic syndrome, and liver cirrhosis—where its favourable safety and efficacy profile may offer therapeutic advantage over conventional options.48 However, these extensions of indication require careful patient selection and further real-world validation.
Mechanistically, finerenone’s benefit in hypertension lies in its ability to disrupt maladaptive MR signaling responsible for vascular stiffening, cardiac hypertrophy, and renal fibrosis. In contrast to spironolactone, which may incompletely suppress MR in non-epithelial tissues, finerenone demonstrates better distribution to cardiovascular tissues and acts as an inverse agonist, enhancing its anti-inflammatory and anti-fibrotic effects.49
In T2DM patients with proteinuria CKD, finerenone’s impact on albuminuria has emerged as a surrogate marker for long-term renal and cardiovascular outcomes.50 By reducing proteinuria and delaying the decline in estimated glomerular filtration rate (eGFR), finerenone has been shown to significantly attenuate disease progression, making it particularly valuable in managing diabetic nephropathy.50-52
From a treatment strategy perspective, finerenone also offers potential synergy when combined with other antihypertensive classes, such as SGLT2 inhibitors or renin–angiotensin system (RAS) blockers. These combinations may enable more effective blood pressure control with lower doses, reduced adverse event profiles, and better long-term adherence.
In conclusion, finerenone stands out as a next-generation MRA with a strong safety profile, validated efficacy across key clinical trials, and mechanistic rationale for use in high-risk hypertensive populations. Comparative analyses with steroidal MRAs reinforce its role in personalized, organ-protective hypertension management, especially for patients with CKD and T2DM. Future research should explore its long-term outcomes in broader populations and its role in multidrug regimens aimed at comprehensive cardiorenal protection.
Conclusion
Finerenone represents a significant advancement in the management of hypertension, offering dual benefits in blood pressure control and organ protection, particularly in patients with coexisting chronic kidney disease (CKD) and type 2 diabetes mellitus (T2DM). Its selective antagonism of the mineralocorticoid receptor (MR) effectively mitigates aldosterone-driven mechanisms such as sodium retention, vascular inflammation, fibrosis, and endothelial dysfunction, which are central to hypertension-induced end-organ damage.
Evidence from major clinical trials—including FIDELIO-DKD and FIGARO-DKD—demonstrates that finerenone reduces systolic blood pressure, urinary albumin-to-creatinine ratio (UACR), and the incidence of cardiovascular and renal events in high-risk populations. Notably, it maintains these benefits even at eGFR levels as low as 25 mL/min/1.73 m², making it accessible to patients with advanced CKD, where traditional MRAs are often contraindicated due to hyperkalemia risk.
Furthermore, finerenone has shown favorable safety outcomes, avoiding endocrine-related side effects such as gynecomastia and glucose intolerance, which are more frequently associated with spironolactone. Its neutral effect on glycemic markers and lower hyperkalemia incidence supports its long-term use in diabetics with kidney dysfunction.
As clinical research continues to explore its role in resistant hypertension, heart failure, and nephrotic syndromes, finerenone is well-positioned to become an integral component of personalized and combination therapy regimens. Future real-world studies and head-to-head comparisons will help refine its optimal placement in treatment algorithms.
In conclusion, supported by robust clinical evidence and a favourable mechanistic profile, finerenone offers a novel, safe, and effective approach for hypertension management in complex patient populations. Its integration into guideline-based therapy has the potential to transform outcomes in those at highest cardiovascular and renal risk.
Acknowledgment
The authors express their gratitude to Saveetha Institute of Medical and Technical Sciences for supporting this research and extended thanks to the Department of Pharmacology, Saveetha Medical College and Hospitals, for their invaluable guidance, resources, and facilities that significantly contributed to the completion of this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Monisha Prasad: Methodology, Data Collection, Writing – Original Draft.
- Sulthan Al Rashid: Conceptualization, Data Analysis, Writing – Review & Editing.
- Vinjmur Ragavan Yashvanthan: Supervision, Validation, Final Approval.
- Bruhan Math Prakash: Critical Review, Visualization, Editing.
- Malarveni Damodaran Lakshmi Priya: Resources, Literature Review, Data Interpretation.
References
- Judd E, Calhoun DA. Apparent and true resistant hypertension: definition, prevalence and outcomes. J Hum Hypertens. 2014;28(8):463-468.
CrossRef - Tu L, Thuillet R, Perrot J. Mineralocorticoid receptor antagonism by finerenone attenuates established pulmonary hypertension in rats. Hypertension. 2022;79(10):2262-2273.
CrossRef - Przezak A, Bielka W, Pawlik A. Hypertension and type 2 diabetes—the novel treatment possibilities. Int J Mol Sci. 2022;23(12):6500.
CrossRef - Sawami K, Tanaka A, Node K. Recent understandings about hypertension management in type 2 diabetes: what are the roles of SGLT2 inhibitor, GLP-1 receptor agonist, and finerenone? Hypertens Res. 2023;46(8):1892-1899.
CrossRef - Kintscher U, Bakris GL, Kolkhof P. Novel non-steroidal mineralocorticoid receptor antagonists in cardiorenal disease. Br J Pharmacol. 2022;179(13):3220-3234.
CrossRef - Rico-Mesa JS, White A, Ahmadian-Tehrani A, Anderson AS. Mineralocorticoid receptor antagonists: a comprehensive review of finerenone. Curr Cardiol Rep. 2020;22(11):140.
CrossRef - Oliveras A, de la Sierra A. Resistant hypertension: patient characteristics, risk factors, co-morbidities and outcomes. J Hum Hypertens. 2014;28(4):213-217.
CrossRef - Buonacera A, Stancanelli B, Malatino L. Stroke and hypertension: an appraisal from pathophysiology to clinical practice. Curr Vasc Pharmacol. 2019;17(1):72-84.
CrossRef - Taylor DA. Hypertensive crisis: a review of pathophysiology and treatment. Crit Care Nurs Clin North Am. 2015;27(4):439-447.
CrossRef - Konukoglu D, Uzun H. Endothelial dysfunction and hypertension. Adv Exp Med Biol. 2017;956:511-515.
CrossRef - Strilchuk L, Cincione RI, Fogacci F, Cicero AFG. Dietary interventions in blood pressure lowering current evidence in 2020. Kardiol Pol. 2020;78(7-8):659-666.
CrossRef - Hong D, Shan W. Improvement in hypertension management with pharmacological and non-pharmacological approaches: current perspectives. Curr Pharm Des. 2021;27(4):548-555.
CrossRef - Oliveros E, Patel H, Kyung S. Hypertension in older adults: assessment, management, and challenges. Clin Cardiol. 2020;43(2):99-107.
CrossRef - Helmer A, Slater N, Smithgall S. A review of ACE inhibitors and ARBs in Black patients with hypertension. Ann Pharmacother. 2018;52(11):1143-1151.
CrossRef - Novo S, Lunetta M, Evola S, Novo G. Role of ARBs in blood hypertension therapy and prevention of cardiovascular events. Curr Drug Targets. 2009;10(1):20-25.
CrossRef - Shah K, Seeley S, Schulz C, Fisher J, Gururaja Rao S. Calcium channels in the heart: disease states and drugs. Cells. 2022;11(6):943.
CrossRef - Zhu J, Chen N, Zhou M. Calcium channel blockers versus other classes of drugs for hypertension. Cochrane Database Syst Rev. 2022;1(1):CD003654.
CrossRef - Sekar D. Innovations in microRNA-based electrochemical biosensors for essential hypertension. Hypertens Res. 2024;47(7):2003-2005.
CrossRef - Blowey DL. Diuretics in the treatment of hypertension. Pediatr Nephrol. 2016;31(12):2223-2233.
CrossRef - McNally RJ, Faconti L, Cecelja M, Farukh B, Floyd CN, Chowienczyk PJ. Effect of diuretics on plasma renin activity in primary hypertension: a systematic review and meta-analysis. Br J Clin Pharmacol. 2021;87(5):2189-2198.
CrossRef - Li H, Xu TY, Li Y. Role of α1-blockers in the current management of hypertension. J Clin Hypertens (Greenwich). 2022;24(9):1180-1186.
CrossRef - Bavishi C, Bangalore S, Messerli FH. Renin angiotensin aldosterone system inhibitors in hypertension: is there evidence for benefit independent of blood pressure reduction? Prog Cardiovasc Dis. 2016;59(3):253-261.
CrossRef - Nyamagoud SB, Swamy AH, Hiremath J. Finerenone as a new potent resistant hypertension agent: a review. Int J Pharm Investig. 2024;14(1).
CrossRef - Marcath LA. Finerenone. Clin Diabetes. 2021;39(3):331-332.
CrossRef - Zhang Y, Jiang L, Wang J. Network meta-analysis on the effects of finerenone versus SGLT2 inhibitors and GLP-1 receptor agonists on cardiovascular and renal outcomes in patients with type 2 diabetes mellitus and chronic kidney disease. Cardiovasc Diabetol. 2022;21(1):232.
CrossRef - Grune J, Benz V, Brix S. Steroidal and nonsteroidal mineralocorticoid receptor antagonists cause differential cardiac gene expression in pressure overload-induced cardiac hypertrophy. J Cardiovasc Pharmacol. 2016;67(5):402-411.
CrossRef - Filippatos G, Anker SD, Agarwal R. Finerenone and cardiovascular outcomes in patients with chronic kidney disease and type 2 diabetes. Circulation. 2021;143(6):540-552.
CrossRef - Singh AK, Singh A, Singh R, Misra A. Finerenone in diabetic kidney disease: a systematic review and critical appraisal. Diabetes Metab Syndr. 2022;16(10):102638.
CrossRef - Frampton JE. Finerenone: first approval. Drugs. 2021;81(15):1787-1794.
CrossRef - Pitt B, Filippatos G, Agarwal R. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N Engl J Med. 2021;385(24):2252-2263.
CrossRef - Bakris GL, Agarwal R, Anker SD. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med. 2020;383(23):2219-2229.
CrossRef - Stier CT Jr, Koenig S, Lee DY, Chawla M, Frishman WH. Aldosterone and aldosterone antagonism in cardiovascular disease: focus on eplerenone (Inspra). Heart Dis. 2003;5(2):102-118.
CrossRef - Lv R, Xu L, Che L, Liu S, Wang Y, Dong B. Cardiovascular-renal protective effect and molecular mechanism of finerenone in type 2 diabetic mellitus. Front Endocrinol (Lausanne). 2023;14:1125693.
CrossRef - Toda N, Nakanishi S, Tanabe S. Aldosterone affects blood flow and vascular tone regulated by endothelium-derived NO: therapeutic implications. Br J Pharmacol. 2013;168(3):519-533.
CrossRef - Chaudhuri A, Ghanim H, Arora P. Improving the residual risk of renal and cardiovascular outcomes in diabetic kidney disease: a review of pathophysiology, mechanisms, and evidence from recent trials. Diabetes Obes Metab. 2022;24(3):365-376.
CrossRef - Lavall D, Selzer C, Schuster P. The mineralocorticoid receptor promotes fibrotic remodeling in atrial fibrillation. J Biol Chem. 2014;289(10):6656-6668.
CrossRef - Lavall D, Jacobs N, Mahfoud F. The non-steroidal mineralocorticoid receptor antagonist finerenone prevents cardiac fibrotic remodeling. Biochem Pharmacol. 2019;168:173-183.
CrossRef - Kolkhof P, Jaisser F, Kim SY. Steroidal and novel non-steroidal mineralocorticoid receptor antagonists in heart failure and cardiorenal diseases: comparison at bench and bedside. Handb Exp Pharmacol. 2017;243:271-305.
CrossRef - Shibata S, Fujita T. The kidneys and aldosterone/mineralocorticoid receptor system in salt-sensitive hypertension. Curr Hypertens Rep. 2011;13(2):109-115.
CrossRef - Shibata S, Ishizawa K, Uchida S. Mineralocorticoid receptor as a therapeutic target in chronic kidney disease and hypertension. Hypertens Res. 2017;40:221-225.
CrossRef - Chen W, Zheng L, Wang J, Lin Y, Zhou T. Overview of the safety, efficiency, and potential mechanisms of finerenone for diabetic kidney diseases. Front Endocrinol (Lausanne). 2023;14:1320603.
CrossRef - Agarwal R, Ruilope LM, Ruiz-Hurtado G. Effect of finerenone on ambulatory blood pressure in chronic kidney disease in type 2 diabetes. J Hypertens. 2023;41(2):295-302.
CrossRef - Kovesdy CP, Ebert N, Vizcaya D, Walsh M, Kosiborod MN, Layton JB, Ziemiecki R, Johannes CB, Pladevall-Vila M, Gee PO, Jefferson N, Chicoye A, Lopes M, Thapa BB, Curhan G, Rangel L, Bhartia M, Liu F, Farjat AE, Oberprieler NG. Change in Urine Albumin-Creatinine Ratio and Occurrence of Hyperkalemia in Patients Initiating Finerenone in the USA: A Cohort Study from the FOUNTAIN Platform. Nephron. 2025 Feb 27:1-13.
CrossRef - Agarwal R, Filippatos G, Pitt B. Cardiovascular and kidney outcomes with finerenone in patients with type 2 diabetes and chronic kidney disease: the FIDELITY pooled analysis. Eur Heart J. 2022;43(6):474-484.
CrossRef - Davies JI, Band M, Morris A. Spironolactone impairs endothelial function and heart rate variability in patients with type 2 diabetes. Diabetologia. 2004;47:1687-1694.
CrossRef - Yamaji M, Tsutamoto T, Kawahara C. Effect of eplerenone versus spironolactone on cortisol and hemoglobin A1c levels in patients with chronic heart failure. Am Heart J. 2010;160:915-921.
CrossRef - Zhao JV, Xu L, Lin S. Spironolactone and glucose metabolism: a systematic review and meta-analysis of randomized controlled trials. J Am Soc Hypertens. 2016;10:671-682.
CrossRef - Ruilope LM, Agarwal R, Anker SD. Blood pressure and cardiorenal outcomes with finerenone in chronic kidney disease in type 2 diabetes. Hypertension. 2022;79(12):2685-2695.
CrossRef - Agarwal R, Anker SD, Bakris G. Investigating new treatment opportunities for patients with chronic kidney disease in type 2 diabetes: the role of finerenone. Nephrol Dial Transplant. 2022;37(6):1014-1023.
CrossRef - Long A, Salvo M. Finerenone: a novel mineralocorticoid receptor antagonist for cardiorenal protection in CKD and T2DM. Ann Pharmacother. 2022;56(9):1041-1048.
CrossRef - Zhang H, Xie J, Hao C Finerenone in patients with chronic kidney disease and type 2 diabetes: the FIDELIO-DKD subgroup from China. Kidney Dis (Basel). 2023;9(6):498-506.
CrossRef - Ortiz A, Alcázar Arroyo R, Casado Escribano PP. Optimization of potassium management in patients with chronic kidney disease and type 2 diabetes on finerenone. Expert Rev Clin Pharmacol. 2023;16(6):519-531.
CrossRef







