Manuscript accepted on :03-07-2025
Published online on: 24-07-2025
Plagiarism Check: Yes
Reviewed by: Dr. Subhasis Chakraborty
Second Review by: Dr. Samara Sameer Yonus
Final Approval by: Dr. Anton R Keslav
Venkatesan Kotteeswaran* , Sasi Kumar K
, Sasi Kumar K , Mukhil M
, Mukhil M , Tarini Bardeja
, Tarini Bardeja , Trisha Saha
, Trisha Saha , R. Vasantha Narayanan
, R. Vasantha Narayanan , Ramakrishnan Govindaraj
, Ramakrishnan Govindaraj
Department of Biotechnology, School of Bioengineering, SRM Institute of Science and Technology, Kattankulathur, India
Corresponding Author E-mail:venkatek@srmist.edu.in
DOI : https://dx.doi.org/10.13005/bpj/3221
Abstract
Carbon nanodots have emerged as a promising nanomaterial in the biotech sector, leveraging their low cytotoxicity and remarkable biocompatibility. Their intriguing properties, including nanoscale dimensions, bioresorbability, safety, environmental friendliness, cost-effectiveness, facile chemical and surface functionalization, and photoluminescence, underpin a wide array of potential applications. This review offers a comprehensive exploration of CND synthesis methodologies, encompassing top-down approaches like organic precursor carbonization and bottom-up strategies employing molecular precursors. While significant studies have focused on the characterization, synthesis, and application of CNDs, this outline summarizes recent advancements in their preparation and utilization. Furthermore, we delve into prospective future applications, particularly within healthcare. The substantial attention garnered by carbon dots in the past decade has spurred extensive investigation, with carbon nanodots, the newest evolution of nanocarbon materials, currently under intense scrutiny for their diverse potential.
Keywords
Carbon nanodots; Characterization; Properties; Photoluminescence; Sources; Synthesis
Download this article as:| Copy the following to cite this article: Kotteeswaran K, Kumar K. S, Mukhil M, Bardeja T, Saha T, Narayanan R. V, Govindaraj B. A Comprehensive Review on Carbon Nanodots: From Synthesis to Applications. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Kotteeswaran K, Kumar K. S, Mukhil M, Bardeja T, Saha T, Narayanan R. V, Govindaraj B. A Comprehensive Review on Carbon Nanodots: From Synthesis to Applications. Biomed Pharmacol J 2025;18(3). |
Introduction
A carbon nanodot is a dispersed, quasi-spherical carbon nanoparticle that is less than 10nm in size.1 Carbon nanodots comprise two types of carbon atoms, sp2 and sp3, and have many polymer chains or functional structures attached to their surfaces. Carbon nanodots are made up of two types of carbon atoms, sp2 and sp3, and have many polymer chains or functional structures attached to their surfaces. Due to their remarkable qualities, carbon nanodots have drawn a lot of attention from researchers. These qualities include great electron conductivity, resistance to breaking down when exposed to light, high light-emitting efficiency, glowing properties, good mixing in water, excellent compatibility with living organisms, long-lasting chemical stability, low cost, minimal harmful effects, and a large surface area that is effective.2 Different synthesis routes influence the diverse architectures of carbon nanodots, broadly categorised as bottom-up and top-down approaches.3 The top-down strategy, a subtractive methodology, involves the meticulous deconstruction of macroscopic carbonaceous materials into nanoscale fragments, a process frequently employed in the synthesis of carbon nanodots. Conversely, the bottom-up approach, an additive technique, centres on the controlled assembly of nanostructures from molecular precursors through a variety of sophisticated chemical methodologies. Carbon nanodots can be used to track how scaffold nanomaterials break down and work in tissue regeneration because of their unique optical features, such as changing fluorescence brightness, ability to emit light, and strong resistance to light damage. They possess advantageous photothermal properties that can eradicate tumour and cancer cells.4 In this literature review, we discussed how the unique properties of carbon nanodots aid their application. We intend to showcase recent developments in carbon nanodot research, focusing on production, characterisation, and applications. This study stands out from other review articles on similar topics because it shows the biological uses of various carbon nanodots all together in one clear picture, which is not often seen in published research.
 |
Figure 1: Properties of the Carbon Nanodot
|
Synthesis
The synthesis of carbon nanodots is categorised into two primary types: Top-down approaches for synthesising carbon nanodots from precursor materials necessitate the imposition of extreme physical or chemical conditions. Bottom-up methodologies employ external energy sources, including pyrolysis, heating, and ultrasonication, to synthesise carbon dots from small molecules like glucose and fructose.5 The primary variables influencing the synthesis of carbon nanodots are surface state, cellular state, and quantum entanglement effects. The synthesis techniques of carbon nanodots enable exact control of these variables. Several functional groups can be added to carbon nanodots, including hydroxyl, carbonyl, carboxyl, epoxy, and amine. Using different biological, polymeric, and organic substances, the surface of carbon nanodots can be changed by adding heteroatoms like nitrogen, phosphorus, sulphur, and boron. Changing the size and number of functional groups on the surface of carbon nanodots can change their properties. This can be done by using different production methods or starting materials.2
Table 1: Sources of Carbon Nanodots
| S.No. | SOURCE | METHOD | Interpretation | REFERENCE |
| 1. | Biomass | Hydrothermal/Solvothermal | Utilizes organic precursors like glucose, citric acid, or biomass waste. | 6 |
| 2. | Carbon- rich Materials | Pyrolysis | The thermal decomposition of carbon-rich materials, such as coal or biomass | 7 |
| 3. | Graphite | Electrochemical Exfoliation | Exfoliates graphite using electrochemical methods to produce C-dots. | 8 |
| 4. | Carbon Soot | Laser Ablation | Ablates carbon soot into nanodots using a laser. | 7 |
| 5. | Carbohydrates | Microwave Irradiation | Carbon dots are formed from carbohydrates using microwave energy. | 6 |
| 6. | Citric acid | Thermal Decomposition | Citric acid is heated to a high temperature under a vacuum to form carbon dots. | 3 |
| 7. | Food Waste | Hydrothermal Carbonization | Converts food waste, such as fruit peels, into C-dots using hydrothermal processes. | 6 |
| 8. | Polymer Precursors | Solvothermal | Polymers such as polyethylene glycol are heated to high temperatures and pressures, and then carbonized to form carbon nanodots. | 8 |
Top-down Approach
Laser Ablation
The laser ablation technique utilises a powerful laser pulse to expose substantial carbonaceous materials to a thermodynamic environment characterised by elevated pressure and temperature. As a result, there is an increase in heat and the evaporation of plasma. A subsequent crystallisation process converts the generated vapour into carbon nanodots.2 Laser ablation is known for being simple and effective and for producing high-quality nanodots, making it an important method for creating nanomaterials. Researchers have conducted recent studies to examine the impact of varying laser wavelengths on the synthesis process. It was found that using lasers at 532 nm and 1064 nm created many clumps of carbon nanodots and big clusters seen under a transmission electron microscope. Samples ablated at 355 nm had produced smaller agglomerates. 9 In a specific study, various carbon precursor materials demonstrated the ability to produce CNDs with varying doping elements. Toluene and pyridine were selected and tested as carbon precursors. After the laser ablation process, the toluene has produced undoped CNDs, whereas the pyridine has produced N-doped CNDs. This experiment demonstrates that the nitrogen atom in the pyridine attaches itself to the CNDs to produce N-doped CNDs. 10
Ultrasonic Method
The ultrasonication method is a simple approach to synthesising carbon nanodots with controllable properties like surface charge, solubility, size, fluorescence colour and quantum yield. Using this approach, CNDs with desired properties can be obtained for various applications, e.g., bioimaging/sensing, drug delivery, etc. 11 The acoustic cavitation process is the mechanism that the ultrasonication method uses to synthesise CNDs. This process is divided into three steps: bubble formation, implosion, and nanodot formation. The first step, bubble formation, uses ultrasound to generate pressure variations in the dispersion medium, which leads to the formation of micro-vacuum bubbles. In the second step, the micro-vacuum bubbles burst, causing very high temperatures (about 5000 K), intense pressures (around 1000 atm), hydrodynamic shear forces and rapid liquid jet penetration. Third step, nanodot formation: In this step, the large carbon is broken down into tiny CNDs by hydrodynamic shear forces that break down carbon structures and the rapid liquid jet penetration to keep them from clumping together. Advantages of the ultrasonication method are cost-effectiveness, eco-friendliness, scalability, rapid synthesis, and uniform particles. 2 Researchers have recently conducted experiments with this method on various substances and carbon precursors. In the year 2018, crab shells will be used for the synthesis of CNDs through ultrasonic methods, which aim to raise environmental awareness and produce valuable material through waste. The successful synthesis of CNDs has proven beneficial for fluorescent imaging. 12 Additionally, it is shown that CNDs can also be produced using tender coconut water, which was found to have strong green fluorescence.13
Arc Discharge Method
A lesser-known method for synthesising carbon dots is arc discharge, which converts electrical energy to thermal energy to generate plasma while dissociating nanoparticles. An electrical current at elevated voltage is produced by submerging two electrodes made of carbon at the ideal distance apart in water in a chamber. Then the chamber is filled with inert gas such as helium or argon. The dispersion of high-energy plasma within the reactor can elevate the core temperature beyond 3500 °C. The elevated temperature facilitates the sublimation of carbon atoms from the anode, leading to their reassembly in the cathode to create carbon dots. Carbon dots created with arc discharge often have oxygen groups like carboxylic (-COOH) and hydroxyl (-OH) attached, and they can still glow steadily even if they haven’t been treated on the surface. 14 While making single-walled carbon nanotubes with the arc discharge method, researchers unexpectedly found three different kinds of carbon nanoparticles, each having unique molecular weights and glowing properties. When exposed to 365 nm light, carbon nanodots emit blue-green, yellow, and orange fluorescence. 15
Electrochemical Method
The electrochemical method is a common way to make carbon nanodots due to its flexibility and other benefits. This method’s key features are that it controls CND size and obtains them with high purity. 2 Researchers have conducted a study on the impact of this method’s varying synthesis potential. Ethanol has been used as a medium in which two platinum plates are used as anodes and cathodes. The synthesis of CNDs took place at 3, 4.5, 6 and 7.5 V. Following the synthesis process, a significant difference was observed in the size of the produced CNDs. Researchers found that the size ranged of this study concludes that an increase in the voltage of the process leads to the synthesis of carbon nanodots of larger size. And that an increase in the voltage of the process leads to the synthesis of carbon nanodots of larger size. 16
Chemical Oxidation
The chemical oxidation method involves oxidising precursor materials from carbon sources using oxidants to produce carbon nanodots. This method does not include any advanced apparatus for large-scale production. The carbon nanodots demonstrate considerable hydrophilicity and chemical reactivity due to the abundance of oxygen-containing functional groups (e.g., hydroxyl and carboxyl groups) generated on their surface during oxidation, thus bestowing remarkable hydrophilicity and chemical activity to the carbon nanodots. 17 Carbon nanodots are synthesised electrochemically or chemically by oxidation-reduction reactions under standard pressure and temperature. It requires the use of strong oxidising agents, such as hydrogen peroxide (H₂O₂), sulphuric acid (H₂SO₄), and nitric acid (HNO₃). You can change the surfaces of carbon dots by adding water-attracting groups like NH₂, COOH, and OH by controlling redox processes and the makeup of the electrolyte. 18
 |
Figure 2: Synthesis of the Carbon Nanodots
|
Bottom-up Approach
Pyrolysis
Pyrolysis is usually done in an inert atmospheric environment and by providing a very high temperature from 300°C to 1000°C. The process of carbonisation of organic substances is generally done through the pyrolysis process. 14 During the synthesis process, to produce CNDs, highly concentrated acid or base is used. With the help of these chemicals, carbon precursors can be reduced to CNDs. The optical characteristics of the carbon dots were improved by altering various essential factors such as reaction temperature, reaction time, and pH of the mixture. Recent research has shown that a variety of materials and substances can be used as a carbon source, such as biomass material and fruit waste. 19 One of the substances used was found to be watermelon peels. These watermelon peels were carbonised, and the produced products were filtered without any difficulties. The produced CNDs had shown strong blue luminescence. 20
Hydrothermal Method (HTC)
HTC is a cost-effective, environmentally friendly, and non-toxic method for the production of carbon dots. The process starts with organic solvents reacting with each other and being kept in a hydrothermal reactor at high temperatures and pressures. The HTC technique can synthesise carbon dots from a diverse range of raw materials, such as chitosan, citric acid, glucose, proteins, and many more. 2 Carbon nanodots emissive properties are determined by the heating temperature and reaction time. Higher temperatures contribute to red-emitting carbon nanodots, whereas lower temperatures produce blue or green emissive colours. Recent study analysis indicates that hydrothermal treatment of organic carbon precursors, especially from natural sources, seldom achieves a quantum yield of 50% or above. 14 A sugarcane juice-soluble aqueous media serves as a plant-based source of luminous carbon dots, measuring less than 5 nm in size. These carbon dots detected Cu²⁺ with high sensitivity and specificity. 6
Microwave-assisted Method
Microwaves cover a broad spectrum of electromagnetic waves, which can range from 1 mm to 1 m, to facilitate the rapid dissociation of chemical bonds in precursor molecules. The microwave-assisted technique is comparatively simple, cost-effective, and efficient, yielding shorter reaction times while generating uniform heat for the homogeneous dispersion of carbon nanodots. 2 Microwaves are waves that fall between 1 cm and 1 m in wavelength and belong to the electromagnetic spectrum of light. Their frequency range is 300 MHz to 300 GHz. Two perpendicular oscillating fields, which are employed as carriers of energy and information, combine to form these waves. The interaction of microwaves with particular materials, which can absorb some of their electromagnetic energy and transform it into heat, is their first application. For this purpose, commercial microwaves require 2450 MHz of energy, which is roughly equal to 600–700 W. 21 They developed a straightforward microwave-assisted method for the synthesis of carbon dots from Mangifera indica leaves in a single container. Their carbon dots demonstrated the ability to detect intracellular temperature, excellent photostability, favourable biocompatibility, and excitation-independent near-infrared emission, as well as enhanced cellular uptake. The carbon nanoparticles exhibited exceptional chemical stability, fluorescence intensity, and excitation-dependent fluorescence in biological matrices. 20
Thermal Decomposition
The thermal degradation of massive carbonaceous precursors through carbonisation or pyrolysis at high temperatures is referred to as thermal decomposition, and it is among the most efficient methods for synthesising carbon nanodots. Mass production of carbon nanodots, reduced reaction time, easier synthesis, solvent-free strategies, and broader precursor tolerance are all advantages of this method. Moreover, by adjusting variables such as reaction-mix pH, reflux duration, and reaction temperatures, the thermal technique can improve the fluorescence properties of carbon nanodots. 2 A carbon-based precursor, like citric acid, glucose, or natural gas, undergoes high-temperature treatment in an inert or reducing environment throughout this procedure. Carbon nanodots and other byproducts are formed when the precursor decomposes due to the heat. Techniques such as centrifugation, dialysis, or chromatography are used to remove impurities and achieve uniformity in the produced carbon nanodots. 8 Bioimaging, drug delivery, and sensors are potential uses for the highly regarded and easily controllable carbon nanodots that can be synthesised using this method. 22
Template Method
The synthesis of nano-sized carbon nanodots involves two stages: the generation of carbon nanodots through calcination within mesoporous silicon spheres or templates, after which etching is done to remove supports and produce nano-sized carbon dots. 2 Researchers devised a method utilising mesoporous silica spheres as rigid templates, yielding photostability and monodispersion with exceptional fluorescent characteristics. 2 Template-based carbon nanodot synthesis uses mesoporous silica. Template pores receive organosilane, a carbon-rich precursor. The template with precursor in its pores thermally decomposes in an inert or reducing atmosphere. The precursor decomposes at high temperatures, forming carbon nanodots in template pores. After making carbon nanodots, remove the template. Without damaging nanodots, chemical etching or calcination dissolves or burns the template. Purifying carbon nanodots after template removal removes residual template material and byproducts. To ensure uniform size and shape, carbon nanodots are formed within the template’s consistent and defined spaces. 23
Characterization of Nanodots
 |
Figure 3: Characterization of the Carbon Nanodot
|
UV-visible Spectroscopy
UV-visible spectroscopy operates on the principle of distinct spectra produced by the absorption of ultraviolet light or visible light due to chemical compounds. When the chemical compound absorbs the UV, it undergoes excitation and de-excitation, producing a distinct spectrum. Spectroscopy is based on the light and matter interaction. C-dots have wide and stable absorption bands in the ultraviolet to visible wavelength spectrum. The optical absorbance spectrum of C-Dots is generally around 230–340 nm with a tail spanning into the visible region. 11, 19, 24 The absorption of UV peaks located in the range of 220-270 nm can be attributed to π-π* transition C=C and C=N bonds of the carbon core. The peak in the range of 280-350 can be ascribed to the n-π* transition of C-O and C=O bonds of the CO surface group. Absorption peaks positioned in the region of 350–600 nm wavelength are usually due to the transition of functional groups on the surfaces of C-dots. 19,24 CNDs synthesised from citric acid using thermolysis techniques and applied them to sense protein molecules. To characterise the carbon nanodots, they studied UV-VIS spectroscopy from 265 nm to 450 nm at room temperature and concluded that the peak at 340 nm is attributed to the n-π* transition of C=O bonds and may also involve contributions from additional functional groups present on the surface of the synthesised functional groups. 25
High-Resolution Transmission Electron Microscopy (HR-TEM)
HR-TEM is a valuable tool for gaining insights into the size distribution, structural pattern, and direct imaging of atoms in nanoparticles. 26 HR-TEM can observe the atomic structures of nanoparticles with high accuracy, usually obtaining an image resolution of better than 0.2 nm. 27 The carbon nanodots are dispersed in a suitable solvent and then deposited onto a TEM grid. The sample needs to be ultra-thin to allow electrons to pass through. It employs a high-energy electron beam that is transmitted through the sample. As the electrons interact with the atoms in the carbon nanodots, they are scattered, creating an image. The scattered electrons are collected to form this image. The high resolution allows it to reveal atomic-level details of the carbon nanodots. 24 It can produce intricate pictures of the carbon nanodots’ lattice structure, including the arrangement of carbon atoms and any defects or irregularities. 28
Atomic Force Microscopy (AFM)
Atomic force microscopy comprises a 4-quadrant photodiode, a scanner, a laser, and a cantilever with a small tip (probe) at its free end. In this high-resolution scanning probe microscopy method, a topographical image of the sample surface is obtained through the interactions that take place between the tip and the sample surface. 2 Atomic force microscopy-based force spectroscopy is a technology that effectively quantifies the interactions between biomolecules and a wide range of material interfaces. These material interfaces can range from hard inorganic surfaces to soft bio-substrates. 29 The primary advantage of the AFM technique in biotechnology is its capacity to examine biological specimens in their natural environment. This includes the ability to analyse biological samples in buffer solutions in vitro, in situ, and even in vivo without the need for sample preparation, which was previously a time-consuming and laborious process. 30 In a study where a comparison of interaction between mercury ions and CNDs wasproduced through two different methods. The average size was found to be 2nm in the bottom-up approach of synthesis, and the average size of CNDs was found to be 5nm for the top-down approach of synthesis. 13
Fourier-transform Infrared Spectroscopy (FTIR)
FTIR can identify the energy of molecular motions by shining infrared light on a sample and measuring the amount of energy it absorbs. The transferred radiation is picked up, while the sample absorbs the rest. The chemical fingerprints of polymers are determined by using the different bands created by the spectra. 31 It helps to identify the functional groups on the surface of carbon nanodots. When a carbon precursor is partially oxidised to form carbon dots, its surface contains a wide variety of oxygen-containing groups, including hydroxyl groups, carboxyl or carboxylic acid groups, and ether/epoxy groups. Prior to utilisation, carbon dots were altered to equilibrate potential wells on the energy surface, reduce cytotoxicity, and enhance fluorescence quantum yield. Infrared spectroscopy can ascertain the adequacy of passivation in modified carbon dots. 2 The presence of carboxyl groups on the surface was indicated by absorption peaks at 1724 cm⁻¹ and 3307 cm⁻¹.. A double bond was indicated by a peak at 1579 cm-1, and the presence of ether linkages was suggested by an absorption peak at 1097 cm-1.20
Nuclear Magnetic Resonance (NMR)
NMR spectroscopy can be used to study structure, verify purification, and reveal molecular species of carbon nanodots. It is used to identify the hybridisation of carbon atoms within a crystalline lattice and the nature and mode of their bonding. By separating sp³ carbons from sp² carbons, NMR measurements can be employed to determine the structural insights of carbon nanodots. Arenes (sp²) display resonance between 90 and 180 ppm, while aliphatic (sp³) carbons resonate between 8 and 80 ppm. The carbon-13 (¹³C) NMR range revealed the absence of aliphatic carbons, with no single peak below 120 ppm. 32 The majority of the peaks that emerged within the 120–150 ppm range were caused by aromatic carbons. The development of the carbon dots from sp² carbons was confirmed by ¹³C NMR spectroscopic estimations. 20
X-ray Diffraction (XRD)
XRD is an effective procedure for characterising carbon nanodots, phase purity, and crystal structure. 20 It generally describes the clustered nature of the surface of synthesised carbon nanodots. The crystalline or amorphous nature of the carbon nanodots can be ascertained using XRD. Crystalline carbon nanodots will show sharp diffraction peaks, whereas amorphous carbon nanodots will have broad humps. 33 The wide peak obtained in the XRD study illustrates a significant degree of disorder among carbon atoms on the surface of the carbon nanodots. The (002) plane of graphitic carbon is represented by the peak that frequently appears in XRD patterns of carbon nanodots, located at approximately 26° 2θ. Some carbon nanodots may have broad peaks, indicating a primarily amorphous structure with some graphitic domains.26
Challenges of Carbon Nanodots
Table 2: Challenges of Carbon Nanodots
| S. No. | Challenges | Descriptions | Reference |
| 1. | Synthesis Complexity | During the synthesis process, uniform size and consistent properties can be challenging due to multiple steps such as surface passivation and post-synthetic modifications. | 1 |
| 2. | Surface Functionalization | It can be difficult to modify the surface of carbon nanodots for particular uses, and it involves exact control over the chemical processes involved. | 34 |
| 3. | Biocompatibility | Surface functional groups or impurities have the potential to be toxic. | 1 |
| 4. | Size and Surface Area | Propensity to collect, which may have an impact on stability and properties | 6 |
| 5. | Purity | Maintaining high purity and removing impurities from the finished product can be complicated. Impurities can impact the performance and safety of carbon nanodots. | 35 |
| 6. | Environmental Impact | The environmental implications of large-scale carbon nanodot synthesis and disposal are insufficiently recognized, raising concerns about long-term safety and sustainability. | 35 |
| 7. | Solubility | Solubility can vary depending on surface chemistry and the synthesis method. | 36 |
| 8. | Quantum Yield | Defects and surface states can reduce quantum yield. | 36 |
| 9. | Optical Properties | Environmental factors like pH, temperature, and ionic strength can have an impact on the optical properties of Carbon nanodots. This sensitivity may complicate their use in practical applications. | 25 |
| 10. | Photostability | Carbon nanodots are frequently photobleached after prolonged light exposure, limiting their use in long-term imaging and sensing applications. | 25 |
| 11. | Regulatory and Safety Issues | There is concern about the potential for carbon nanodots to accumulate in biological systems and the environment, which could have unexpected repercussions. | 5 |
Application of Carbon Nanodots
Drug Delivery
Carbon nanodots are essential as nanocarriers in the development of innovative medication delivery systems. Innovative nanomaterials-based targeted drug delivery technologies provide a multitude of benefits, including the ability to visualise drug sites using imaging agents on the carrier, transport macromolecular therapeutics, implement a variety of therapeutic strategies, administer multiple drugs simultaneously, enhance the delivery of hydrophobic medications, and facilitate targeted delivery to specific cells, tissues, or organs.2
Carbon nanodots have demonstrated efficacy in drug delivery and the management of specific disorders related to the central nervous system (CNS). However, getting carbon dots into the central nervous system remains the main obstacle for these applications. To test carbon dots’ capacity to penetrate the blood-brain barrier (BBB) in the zebrafish model through transferrin receptor-mediated delivery, they were covalently linked to transferrin and labelled with dye. Transferrin-conjugated carbon dots, unlike carbon dots alone, may traverse the blood-brain barrier to access the CNS.37 Moreover, further study indicates that the mitomycin-loaded carbon dots nanocarrier may efficiently penetrate tumor cells, have pH-dependent release characteristics, and exhibit biocompatibility with MCF-7 cancer cells.11 The distinctive optical and physicochemical features of carbon dots facilitate their application as an efficient drug delivery system, evaluation of cancer therapy efficacy, and determination of accurate medication dosage. However, the ability of carbon dots to target specific disease states is unknown, limiting its therapeutic application.37
Table 3: Applications of Carbon Nanodot
| SL. No. | Applications | Mechanism | Significance | Reference |
| 1. | Biomedical Imaging | Carbon nanodots emit specific wavelengths of light when excited, allowing tracking of cells and processes. | Provides a safer alternative to traditional fluorescent dyes for live-cell and in vivo imaging. | 38 |
| 2. | Drug Delivery | Carbon nanodots can encapsulate drugs and target specific cells, releasing drugs upon triggers. | Enhances therapeutic effect while minimizing side effects, particularly in cancer therapy. | 39 |
| 3. | Gene Therapy | Carbon nanodots encapsulate nucleic acids, facilitating cellular uptake and deliver CRISPR/Cas9 systems for precise gene editing. | Biocompatible and low toxicity, suitable for gene delivery. It also allows monitoring of efficiency and localization of gene therapy. | 40,41 |
| 4. | Environmental Remediation | Carbon nanodots act as photocatalysts to degrade pollutants under light irradiation. | Offers a sustainable method for cleaning contaminated environments, such as water. | 42 |
| 5. | Tissue engineering | Carbon nanodots serve as carriers for growth factors or drugs in engineered tissues. | Allows controlled release of therapeutic agents and aids to monitoring of cell behaviour and integration. | 43 |
| 6. | Photothermal Therapy | Carbon nanodots produce heat when exposed to light, effectively eliminating cancer cells. | Offers a least-invasive method for cancer therapy. | 44 |
Tissue Engineering
Tissue engineering aims to create functional constructions capable of repairing, preserving, or improving damaged tissues. Carbon nanodots have a distinctive range of physicochemical features that enable them suitable for tissue engineering applications. Carbon nanodots can be included into scaffolds to enhance their mechanical strength and biocompatibility. These scaffolds promote cell adhesion, proliferation, and differentiation, which are crucial for tissue regeneration.45
It is proved that nitrogen-doped carbon dots (NCDs) made through hydrothermal/coprecipitation method in conjunction with hydroxyapatite (HA) nanoparticles (NCDs-HA) show potential in zebrafish (ZF) jaw regeneration (JBR).This research indicates that NCDs-HA may promote osteogenesis by modifying osteoblast proliferation, differentiation, and mineralization.46 Carbon dots can be used to create hydrogels, which are highly hydrated networks that resemble the extracellular matrix. Recent research demonstrated that hydrogels made using CNDs can create an environment that is conducive to cell growth and tissue formation when used as scaffolds in tissue engineering, which facilitates cartilage regeneration and chondrogenic differentiation. Then these carbon nanodots can be degraded into smaller, harmless compounds so that the body can be easily eliminated. In the future, it is vital to conduct comprehensive and extended in vivo studies to examine the biodegradation, bio-clearance, and accumulation of a variety of carbon nanodots in primary organs, such as the brain, kidneys, intestines, and liver. The biodegradability of carbon nanodots is a critical determinant in their application in tissue engineering. To accurately evaluate the impact and biosafety of carbon nanodots in tissue engineering, it is imperative to conduct additional research on their biocompatibility and cytotoxicity in a variety of tissues.43
Gene Therapy
Gene therapy is defined as the treatment of disease by introducing genetic material into cells. Incorporating a tumor-suppressor gene into tumor cells inhibits growth during gene therapy or gene delivery. However, nanoparticle-based carriers, such as carbon nanodots, have demonstrated efficacy in gene therapy due to their superior biocompatibility and safety. Currently, highly efficient carbon nanodots are extensively utilized for the delivery of nucleic acids which includes siRNA, noncoding RNAs, and DNA, as they facilitate gene delivery through endocytosis, macro-pinocytosis, and phagocytosis.2 Hydrothermally synthesized polymeric carbon nanodots were employed in a study to improve the efficiency of gene delivery during transfection. The luciferase assay conducted in HeLa cells has shown that the synthesised polymeric carbon nanodots may function as multifunctional gene vectors, with high biocompatibility, low cytotoxicity, and increased transfection efficiency. The positively charged DNA was efficiently condensed and its degradation was inhibited by the synthesised polymeric carbon nanodots.47 In conclusion, carbon nanodots, especially in cancer gene therapy, can serve as versatile transmembrane nanocarriers for the efficient delivery of drugs, genes, or both.48
Bioimaging
Another interesting application of carbon nanodots is their potential as bioimaging agents for cells and organisms in both in vivo and in vitro environments. This is due to their photoluminescence, which is a fundamental characteristic of carbon nanodots.22 This technology enables detection, screening, diagnosis, and image-guided treatment of life-threatening disorders, including cancer, as well as cellular and single-molecule imaging. The imaging capabilities of carbon nanodots are derived from their distinctive optical characteristics, such as their inherent fluorescence, or from functional agents that are present in their interior or on their surface.49 Recent study has shown that carbon nanodots are functionalized with quaternary ammonium lauryl betaine to create positively charged moieties which are examined on three gram-positive and three gram-negative bacterial species. The findings indicated that carbon nanodots effectively label gram-positive organisms, whereas no gram-negative species were tagged. The variation in labelling resulted from differences in cell wall architectures and composition. The substantial peptidoglycan layer in the cell membranes of gram-positive species has several anionic moieties, promoting electrostatic interactions between the positively charged carbon nanodots and anionic species.49 The bioimaging of cellular compartments and organelles improves understanding of the initial symptoms and progression of a variety of diseases, such as Alzheimer’s, Parkinson’s, diabetes, and malignancies. Carbon nanodots were implemented for intracellular imaging of HeLa, MCF-7, Caco-2, HepG2, PC12, lung cancer, and pancreatic progenitor cell lines.50
Photodynamic Therapy
Photodynamic therapy (PDT) consists of three key components: a light source, a photocatalyst (PS), and oxygen. PS absorbs energy from a light source, which causes it to transition from ground state to excited state, resulting in three reactions: a Type I reaction involving electron transfer between PS and intracellular substrates, which produces free radicals, and a Type II reaction involving direct energy transfer to oxygen molecules, which produces singlet oxygen. These reactions can occur simultaneously, producing reactive oxygen species (ROS). The Type III reaction mechanism specifically targets biomolecules, including nucleic acids and proteins, leading to the direct and efficient annihilation of biological target molecules. The resulting ROS are collectively known as ROS. ROS refers to H2O2, O2−, •OH, and 1O2.51 ROS serve as mediators of cytotoxic effects, with their effects restricted to specific target tissues that have been exposed to light. This light-induced toxicity can be employed to selectively eradicate malignant tumours or cancer cells. A thorough examination of the application of PDT in the treatment of cancer. It is also identified as a novel integration of PDT and imaging systems for a variety of disorders, such as cancer.22 Selenium and nitrogen co-doped carbon nanodots (Se/N-CDs) were synthesised by researchers in52 to enhance photodynamic treatment and overcome its in vivo limitations. Cell labelling with RNA probes, isothermal titration microcalorimetry, and digesting methods have all demonstrated that Se/N-CDs exhibit selective binding to RNA. Se/N-CDs produced ROS in the vicinity of the nuclear membrane. The membrane degraded under light irradiation, enabling the absorption of supplementary Se/N-CDs. The capacity for photosensitisation was augmented consequently. Utilising Se/N-CDs as photosensitisers holds the potential to effectively inhibit tumour growth.2
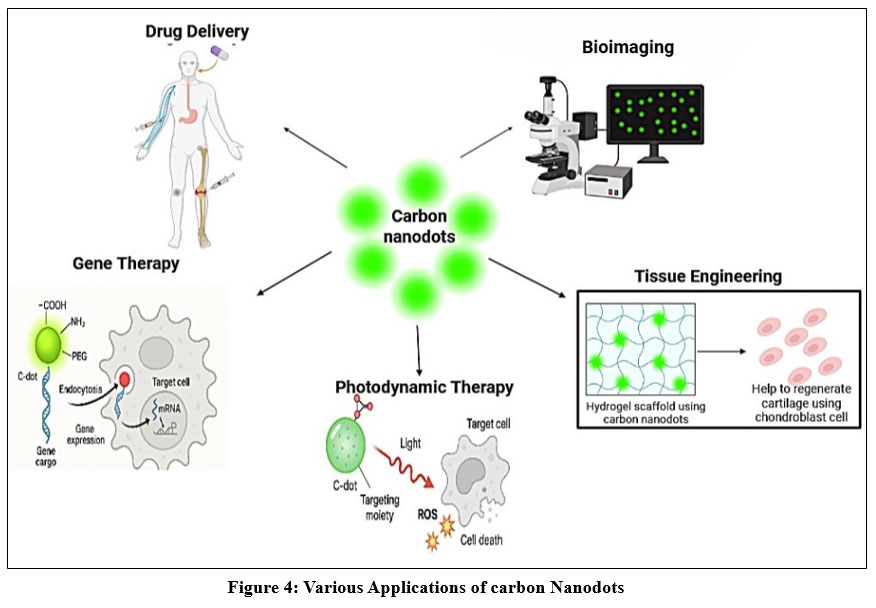 |
Figure 4: Various Applications of carbon Nanodots
|
Conclusion
This literature review investigates the synthesis of carbon nanodots by both top-down and bottom-up methodologies. Comprehending the structural, morphological, and chemical features of carbon nanodots necessitates the application of characterization techniques like TEM, XRD, and FTIR. For the purpose of adapting their properties to particular applications, these insights are absolutely necessary. Carbon nanodots have the potential to revolutionize a variety of fields, as evidenced by their wide-ranging applications in bioimaging, drug delivery, gene therapy, and photodynamic therapy, among other areas. Because of their one-of-a-kind fluorescence properties, high biocompatibility, and low toxicity, these substances are promising prospects for conducting research and development in the future. We have the ability to propel innovation and make a contribution to the development of environmentally friendly technological solutions if we adopt green synthesis techniques and make progress in our understanding of carbon nanodots. The incorporation of carbon nanodots into applications that are used in the real world represents a significant step toward the achievement of technological advancements that are both more environmentally friendly and more efficient. The most recent version of carbon-based nanomaterials, carbon nanodots, has attracted considerable interest from researchers owing to their beneficial properties, including cost-effectiveness, remarkable fluorescence, high solubility in water, low toxicity, and ease of synthesis and surface functionalization. Despite significant advancements in carbon nanodots, their synthesis processes still necessitate improvement. Numerous biotechnological domains remain unexplored and necessitate focused research. Carbon nanodots have an exciting future ahead of them.
Acknowledgement
We would like to convey our heartfelt gratitude to the SRM Institute of Science and Technology, Kattankulathur, whose efforts and assistance have significantly improved the quality and rigor of our research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Venkatesan Kotteesaran: Conceptualization, Analysis, Supervision, Project Administration, Review & Editing.
- Tarini Bardeja: Data Collection, Methodology, Analysis & Writing
- Trisha Saha: Data Collection, Methodology, Analysis & Writing
- Sasi Kumar: Data Collection, Methodology, Analysis & Writing
- Mukhil M: Data Collection, Methodology, Analysis & Writing
- Vasantha Narayanan: Data Collection, Methodology, Analysis & Writing
- Ramakrishnan Govindaraj: Analysis &Writing
References
- Chauhan DS, Quraishi MA, Verma C. Carbon nanodots: recent advances in synthesis and applications. Carbon Lett. 2022;32(7):1603-1629. doi:10.1007/s42823-022-00359-1
CrossRef - Mansuriya BD, Altintas Z. Carbon dots: Classification, properties, synthesis, characterization, and applications in health care-an updated review (2018–2021). Nanomaterials. 2021;11(10). doi:10.3390/nano11102525
CrossRef - Cui, L.; Ren, X.; Sun, M.; Liu, H.; Xia L. Carbon Dots: Synthesis, Properties, and Application. Nanomaterials. 2021;52(1):01053-01063. doi:10.3390/nano11123419
CrossRef - Farshidfar N, Fooladi S, Nematollahi MH, Iravani S. Carbon dots with tissue engineering and regenerative medicine applications. RSC Adv. 2023;13(21):14517-14529. doi:10.1039/d3ra02336b
CrossRef - Roy P, Chen PC, Periasamy AP, Chen YN, Chang HT. Photoluminescent carbon nanodots: Synthesis, physicochemical properties and analytical applications. Mater Today. 2015;18(8):447-458. doi:10.1016/j.mattod.2015.04.005
CrossRef - Banger A, Gautam S, Jadoun S, et al. Synthetic Methods and Applications of Carbon Nanodots. Catalysts. 2023;13(5):1-25. doi:10.3390/catal13050858
CrossRef - Lemberger MM, Hirsch T, Wegener J. Carbon Nanodots: Synthesis, Characterization, and Bioanalytical Applications. Bioanal Rev. 2014;5:135-175. doi:10.1007/11663_2014_11
CrossRef - Khan A, Vishvakarma R, Vimal A, Sharma P, Usman H, Kumar A. Carbon Nanodots: A Novel Carbon Material with Multifacet Applications in Healthcare. Published online 2023:145-167. doi:10.1007/978-3-031-28263-8_6
CrossRef - Reyes D, Camacho M, Camacho M, et al. Laser Ablated Carbon Nanodots for Light Emission. Nanoscale Res Lett. 2016;11(1). doi:10.1186/s11671-016-1638-8
CrossRef - Mas N, Hueso JL, Martinez G, et al. Laser-driven direct synthesis of carbon nanodots and application as sensitizers for visible-light photocatalysis. Carbon N Y. 2020;156:453-462. doi:10.1016/j.carbon.2019.09.073
CrossRef - Jing HH, Bardakci F, Akgöl S, Kusat K, Adnan M, Sasidharan S. Green Carbon Dots: Synthesis, Characterization, Properties and Biomedical Applications. J Funct Biomater. Published online 2023.
CrossRef - Dehvari K, Liu KY, Tseng PJ, Gedda G, Girma WM, Chang JY. Sonochemical-assisted green synthesis of nitrogen-doped carbon dots from crab shell as targeted nanoprobes for cell imaging. J Taiwan Inst Chem Eng. 2019;95:495-503. doi:10.1016/j.jtice.2018.08.037
CrossRef - Bruno F, Sciortino A, Buscarino G, et al. A comparative study of top-down and bottom-up carbon nanodots and their interaction with mercury ions. Nanomaterials. 2021;11(5). doi:10.3390/nano11051265
CrossRef - Ghasemlou M, Navya PN, Alexander K, et al. Fluorescent Nanocarbons: From Synthesis and Structure to Cancer Imaging and Therapy. Adv Mater. 2024;36(19):1-66. doi:10.1002/adma.202312474
CrossRef - Wang X, Feng Y, Dong P, Huang J. A Mini Review on Carbon Quantum Dots: Preparation, Properties, and Electrocatalytic Application. Front Chem. 2019;7(October):1-9. doi:10.3389/fchem.2019.00671
CrossRef - Deng J, Lu Q, Mi N, et al. Electrochemical synthesis of carbon nanodots directly from alcohols. Chem – A Eur J. 2014;20(17):4993-4999. doi:10.1002/chem.201304869
CrossRef - Yang HL, Bai LF, Geng ZR, et al. Carbon quantum dots: Preparation, optical properties, and biomedical applications. Mater Today Adv. 2023;18:100376. doi:10.1016/j.mtadv.2023.100376
CrossRef - Pan M, Xie X, Liu K, Yang J, Hong L, Wang S. Fluorescent carbon quantum dots-synthesis, functionalization and sensing application in food analysis. Nanomaterials. 2020;10(5). doi:10.3390/nano10050930
CrossRef - Kang C, Huang Y, Yang H, Yan XF, Chen ZP. A review of carbon dots produced from biomass wastes. Nanomaterials. 2020;10(11):1-24. doi:10.3390/nano10112316
CrossRef - Zuo P, Lu X, Sun Z, Guo Y, He H. A review on syntheses, properties, characterization and bioanalytical applications of fluorescent carbon dots. Microchim Acta. 2016;183(2):519-542. doi:10.1007/s00604-015-1705-3
CrossRef - Ghanem A, Al-Qassar Bani Al-Marjeh R, Atassi Y. Novel nitrogen-doped carbon dots prepared under microwave-irradiation for highly sensitive detection of mercury ions. Heliyon. 2020;6(4):e03750. doi:10.1016/j.heliyon.2020.e03750
CrossRef - Sivasankarapillai VS, Vishnu Kirthi A, Akksadha M, et al. Recent advancements in the applications of carbon nanodots: Exploring the rising star of nanotechnology. Nanoscale Adv. 2020;2(5):1760-1773. doi:10.1039/c9na00794f
CrossRef - Kurdyukov DA, Eurov DA, Stovpiaga EY, et al. Template synthesis of monodisperse carbon nanodots. Phys Solid State. 2016;58(12):2545-2549. doi:10.1134/S1063783416120167
CrossRef - Khairol Anuar NK, Tan HL, Lim YP, So’aib MS, Abu Bakar NF. A Review on Multifunctional Carbon-Dots Synthesized From Biomass Waste: Design/ Fabrication, Characterization and Applications. Front Energy Res. 2021;9(April):1-22. doi:10.3389/fenrg.2021.626549
CrossRef - Carneiro Cruz AA, Freire RM, Froelich DB, et al. Fluorescence Based Platform to Discriminate Protein Using Carbon Quantum Dots. ChemistrySelect. 2019;4(19):5619-5627. doi:10.1002/slct.201901014
CrossRef - John BK, Abraham T, Mathew B. A Review on Characterization Techniques for Carbon Quantum Dots and Their Applications in Agrochemical Residue Detection. J Fluoresc. 2022;32(2):449-471. doi:10.1007/s10895-021-02852-8
CrossRef - Wang ZL, Lee JL. Electron Microscopy Techniques for Imaging and Analysis of Nanoparticles. Dev Surf Contam Clean Second Ed. 2008;1:395-443. doi:10.1016/B978-0-323-29960-2.00009-5
CrossRef - Thota SP, Kolli NK, Kurdekar A, Thota SM, Vadlani P V., Kumar BS. One-step synthesis of biocompatible luminescent carbon dots from Cuscuta for bio-imaging application. Biomass Convers Biorefinery. 2023;(0123456789). doi:10.1007/s13399-023-05211-8
CrossRef - Li Q, Zhang T, Pan Y, Ciacchi LC, Xu B, Wei G. AFM-based force spectroscopy for bioimaging and biosensing. RSC Adv. 2016;6(16):12893-12912. doi:10.1039/c5ra22841g
CrossRef - Vahabi S, Nazemi Salman B, Javanmard A. Atomic force microscopy application in biological research: A review study. Iran J Med Sci. 2013;38(2):76-83.
- Mattsson K, Jocic S, de Lima JA, Hansson LA, Gondikas A. Nanoplastics in aquatic environments—Sources, sampling techniques, and identification methods. Microplastic Contam Aquat Environ An Emerg Matter Environ Urgency. Published online January 1, 2024:381-397. doi:10.1016/B978-0-443-15332-7.00003-X
CrossRef - Han L, Ghosh D, Chen W, Pradhan S, Chang X, Chen S. Nanosized carbon particles from natural gas soot. Chem Mater. 2009;21(13):2803-2809. doi:10.1021/cm900709w
CrossRef - El-Shafey AM. Carbon dots: Discovery, structure, fluorescent properties, and applications. Green Process Synth. 2021;10(1):134-156. doi:10.1515/gps-2021-0006
CrossRef - Sciortino A, Cannizzo A, Messina F. Carbon Nanodots: A Review—From the Current Understanding of the Fundamental Photophysics to the Full Control of the Optical Response. C. 2018;4(4):67. doi:10.3390/ c4040067
CrossRef - Eco-Friendly Fluorescent Carbon Nanodots: Characteristics and Potential … – Mohammed Muzibur Rahman – Google Books. Accessed December 23, 2024. https://books.google.co.in/books/about/Eco_ Friendly_ Fluorescent _Carbon_Nanodots.html?id=KHEh0AEACAAJ&redir_esc=y
- Singh I, Arora R, Dhiman H, Pahwa R. Carbon quantum dots: Synthesis, characterization and biomedical applications. Turkish J Pharm Sci. 2018;15(2):219-230. doi:10.4274/tjps.63497
CrossRef - Li S, Peng Z, Dallman J, et al. Crossing the blood-brain-barrier with transferrin conjugated carbon dots: A zebrafish model study. Colloids Surfaces B Biointerfaces. 2016;145:251-256. doi:10.1016/j. colsurfb.2016. 05.007
CrossRef - Boakye-Yiadom KO, Kesse S, Opoku-Damoah Y, et al. Carbon dots: Applications in bioimaging and theranostics. Int J Pharm. 2019;564:308-317. doi:10.1016/J.IJPHARM.2019.04.055
CrossRef - Sameer M, Arif Y, Aqil A, et al. Carbon nanodots as a remedial nanovesicles for drug delivery. Eur Polym J. 2023;200:112515. doi:10.1016/J.EURPOLYMJ.2023.112515
CrossRef - Mohammadinejad R, Dadashzadeh A, Moghassemi S, et al. Shedding light on gene therapy: Carbon dots for the minimally invasive image-guided delivery of plasmids and noncoding RNAs – A review. J Adv Res. 2019;18:81-93. doi:10.1016/J.JARE.2019.01.004
CrossRef - Hasanzadeh A, Radmanesh F, Kiani J, et al. Photoluminescent functionalized carbon dots for CRISPR delivery: synthesis, optimization and cellular investigation. Nanotechnology. 2019;30(13). doi:10.1088/1361-6528/AAFBF9
CrossRef - Smrithi SP, Kottam N, Arpitha V, Narula A, G N A, Subramanian KRV. Tungsten oxide modified with carbon nanodots: Integrating adsorptive and photocatalytic functionalities for water remediation. J Sci Adv Mater Devices. 2020;5(1):73-83. doi:10.1016/J.JSAMD.2020.02.005
CrossRef - Zong Q, Chen H, Zhao Y, Wang J, Wu J. Bioactive carbon dots for tissue engineering applications. Smart Mater Med. 2024;5(1):1-14. doi:10.1016/j.smaim.2023.06.006
CrossRef - Lee C, Kwon W, Beack S, et al. Biodegradable Nitrogen-Doped Carbon Nanodots for Non-Invasive Photoacoustic Imaging and Photothermal Therapy. Theranostics. 2016;6(12):2196-2208. doi:10.7150/THNO.16923
CrossRef - Zhang R, Hou Y, Sun L, et al. Recent advances in carbon dots: synthesis and applications in bone tissue engineering. Nanoscale. 2023;15(7):3106-3119. doi:10.1039/D2NR05951G
CrossRef - Zhu L, Kong W, Ma J, et al. Applications of carbon dots and its modified carbon dots in bone defect repair. J Biol Eng. 2022;16(1):1-17. doi:10.1186/s13036-022-00311-x
CrossRef - He X, Chen P, Zhang J, et al. Cationic polymer-derived carbon dots for enhanced gene delivery and cell imaging. Biomater Sci. 2019;7(5):1940-1948. doi:10.1039/C8BM01578C
CrossRef - Yazdani S, Mozaffarian M, Pazuki G, et al. Carbon-Based Nanostructures as Emerging Materials for Gene Delivery Applications. Pharmaceutics. 2024;16(2). doi:10.3390/pharmaceutics16020288
CrossRef - Wang B, Cai H, Waterhouse GIN, Qu X, Yang B, Lu S. Carbon Dots in Bioimaging, Biosensing and Therapeutics: A Comprehensive Review. Small Sci. 2022;2(6). doi:10.1002/smsc.202200012
CrossRef - Das S, Mondal S, Ghosh D. Carbon quantum dots in bioimaging and biomedicines. Front Bioeng Biotechnol. 2023;11(January):1-22. doi:10.3389/fbioe.2023.1333752
CrossRef - Sun L, Zhao Y, Peng H, et al. Carbon dots as a novel photosensitizer for photodynamic therapy of cancer and bacterial infectious diseases: recent advances. J Nanobiotechnology. 2024;22(1):1-24. doi:10.1186/s12951-024-02479-4
CrossRef - Xu X, Ray R, Gu Y, et al. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J Am Chem Soc. 2004;126(40):12736-12737. doi:10.1021/ja040082h
CrossRef







