Manuscript accepted on :04-06-2025
Published online on: 01-07-2025
Plagiarism Check: Yes
Reviewed by: Dr. Sonam Bhutia
Second Review by: Dr. Hany Akeel
Final Approval by: Dr. Prabhishek Singh
Syed Mahabubur Rahman1 , Fahim Salek Gowrab1
, Fahim Salek Gowrab1 , Shaiful Islam1
, Shaiful Islam1 , Sabiha Tabassum1
, Sabiha Tabassum1 , Md. Shafiul Hossen1,2
, Md. Shafiul Hossen1,2 , Arafat Miah1
, Arafat Miah1 , Mohammad Safiqul Islam1
, Mohammad Safiqul Islam1 and Md Abdul Barek1*
and Md Abdul Barek1*
1Department of Pharmacy, Faculty of Science, Noakhali Science and Technology University, Sonapur, Noakhali, Bangladesh
2Department of Pharmacy, State University of Bangladesh, South Purbachal, Kanchan, Dhaka, Bangladesh
Corresponding Author Email: barekpharma@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3209
Abstract
This study examines the anti-inflammatory, analgesic, and antipyretic properties of the methanolic extract of the Zingiber barbatum (ZbME) rhizome to validate traditional applications and explore its potential as an alternative to NSAIDS. Standard assays were used to evaluate the extract's antioxidant capacity as well as its phenolic and flavonoid levels. Swiss albino mice were used to test their analgesic, antipyretic, and anti-inflammatory properties utilizing models of carrageenan-induced edema, acetic acid-induced writhing, formalin, hot plate, and yeast-induced pyrexia. GraphPad Prism with ANOVA and Bonferroni's post hoc test were used to examine the data. Quantitative analysis showed the crude extract contained 70.675 mg GAE (Gallic Acid Equivalent) per gram of total phenolic content and 25.365 mg QE (Quercetin Equivalent) per gram of total flavonoid content, with a total antioxidant capacity of 339.258 mg AAE (Ascorbic Acid Equivalent)/g DW (Dry Weight). In dose-dependent analgesic testing, ZbME inhibited acetic acid-induced writhing by 47.18%, 64.08%, and 66.67% at 100, 200, and 400 mg/kg. Significant reductions in paw-licking responses were noted in the formalin test, with the 400 mg/kg dose showing the highest inhibition. The hot plate test also confirmed ZbME's analgesic effects, with the 400 mg/kg dose significantly extending latency times compared to ketorolac. Like paracetamol, ZbME at 200 and 400 mg/kg lowered body temperatures in a yeast-induced fever model. Thus, ZbME 400 mg/kg inhibited carrageenan-induced paw edema better than Indomethacin in anti-inflammatory testing. The methanolic extract of Zingiber barbatum rhizomes demonstrates notable anti-inflammatory, analgesic, and antipyretic effects, validating its traditional uses. It may serve as a potential alternative to NSAIDS with less adverse effects.
Keywords
Analgesic; Anti-inflammatory; Antipyretic; Antioxidant; In vivo; Phytochemical screening; Zingiber barbatum
Download this article as:| Copy the following to cite this article: Rahman S. M, Gowrab F. S, Islam S, Tabassum S, Hossen S, Miah A, Islam M. S, Barek M. A. Screening of Antioxidant, Analgesic, Antipyretic and Anti-inflammatory Effect of Zingiber barbatum Methanolic Extracts in Mice Model: In Vitro & In Vivo Study. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Rahman S. M, Gowrab F. S, Islam S, Tabassum S, Hossen S, Miah A, Islam M. S, Barek M. A. Screening of Antioxidant, Analgesic, Antipyretic and Anti-inflammatory Effect of Zingiber barbatum Methanolic Extracts in Mice Model: In Vitro & In Vivo Study. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4nz9XDK |
Introduction
Ginger plays a crucial role in traditional medicine systems, including Chinese, Ayurvedic, and Unani-Tibb. It has been employed to address a range of conditions such as muscular aches, dyspepsia, nausea, diabetes, gingivitis, sore throats, constipation, toothaches, nervous disorders, and fever.1 Its carminative qualities and ability to stimulate the gastrointestinal tract are noticeable.2,3 For respiratory disorders, Ayurvedic practitioners use ginger with honey, and for infantile colic, they use milk or water.4 Western medicine employs ginger for digestive issues, and traditional uses extend to blood purification, aphrodisiac effects, anti-nausea, and antispasmodic properties.4 The Yin-Yang philosophy associates ginger with reducing Yin effects and providing body nutrients.5 Traditional Chinese medicine regards ginger as a treatment for cold extremities and weak pulse, and in Eastern medicine, it is valued for its antiemetic effects and cardiovascular benefits.5 In Iran, ginger is traditionally used to enhance male sexuality and regulate menstrual cycles.5,6 The genus Zingiber, encompassing 144 species, is recognized for its medicinal properties.7 Traditional medicine in Asia and Southeast Asia makes use of Zingiber species, which are also popular as seasonings, foods, and nutritional supplements. Zingiber officinale, Zingiber montanum, Zingiber zerumbet, and Zingiber mioga are renowned about their antimicrobial, antioxidant, and anti-inflammatory properties.7 The rhizome’s value is attributed to its bioactive properties, linked to volatile and phenolic compounds like gingerols, shogaols, and paradols.8
Finding effective alternative therapies with fewer side effects than conventional pharmaceuticals has been the subject of growing interest in recent decades. Zingiber barbatum is a perennial or rhizomatous geophyte and grows primarily in the wet tropical biome. It is an aromatic, medicinal and endemic species. The plant naturally grows in hilly regions and it thrives in light to moderate shade and highly humid conditions. Within the vast array of medicinal plants, Zingiber barbatum Wall, a ginger plant found in the wild and part of the Zingiberaceae family, shows great potential. Z. barbatum is a species that has not received much attention in Myanmar, also known as “Meik-thalin” or “Pwe-au.”9An essential aspect of modern healthcare, traditional medicine in Myanmar is deeply rooted in the country’s rich cultural history.10 The rhizomes of Z. barbatum have been traditionally used in ethnomedicine to address various conditions, including gout and to provide relief from joint, bone, and muscle discomfort.11 Despite the potential advantages, Z. barbatum is not being fully utilized, as changes in culture and a lack of extensive research pose risks to its ongoing use and preservation. 12 Z. barbatum has been utilized for medicinal purposes for a long duration of time due to its powerful anti-inflammatory and pain-relieving capabilities.10,11 It can be applied either topically or taken orally.10
Inflammation, characterized by pain, heat, redness, and swelling, is a complex response to harmful stimuli.13 When pro-inflammatory cytokines are released, they induce fever and other inflammatory responses, which trigger the activation of inflammatory pathways and ultimately cause the immune system to get out of harmony. Through the activation of immune cells and the production of mediators like prostaglandin E2 (PGE2), these cytokines mediate inflammation.13 Fever, a marker of viral and inflammatory disorders, is mediated by peripheral vasoconstriction, reduced sweating, shivering, and possibly non-shivering thermogenesis, often initiated by cytokines in response to pathogenic microorganisms.14 Prostaglandin, a potent vasodilator and hyperalgesic agent, is crucial for regulating inflammation, pain, and fever induction.15 NSAIDS alleviate pain, reduce inflammation, and lower high body temperature by inhibiting cyclooxygenase (COX) enzymes responsible for prostaglandin synthesis.13 Considering the detrimental consequences that are linked to long-term NSAID use,16 there is growing interest in alternative therapies, including those derived from natural sources. Herbal medications, which have been used for centuries for effective pain relief, are gaining popularity because they generally have fewer side effects.
Ginger constituents exhibit pharmacological properties similar to dual-acting NSAIDS but with fewer side effects.1 Gingerols effectively suppress prostaglandins, inhibit cytokine synthesis and release, and modulate biochemical pathways involved in chronic inflammation.17 They inhibit the expression of genes related to inflammation, including those coding for cytokines, chemokines, and COX enzymes. Preclinical studies show ginger’s anti-inflammatory, anti-nociceptive, and antipyretic effects, including reduced serum prostaglandin E2. Additionally, 6-gingerol and its metabolites inhibit iNOS expression and macrophage activity.18 The antioxidant, anti-inflammatory, analgesic, and antipyretic benefits of ginger have been highlighted in recent research. The active phytochemicals in it, such as zingerone, 6-shogaol, 6-gingerol, and other flavonoids and phenolic compounds, are mainly responsible for these positive effects.19,20
This research provides an in-depth analysis of the therapeutic potential of Zingiber barbatum, highlighting its significance in modern medicine. By focusing on the methanolic extract of the plant’s rhizome, the study aims to validate its traditional uses and explore new applications in managing pain, inflammation, and fever. The findings contribute to the existing knowledge of medicinal plants and offer valuable insights into developing alternative therapies that are not only effective but also safe. In addition to these pharmacological assessments, the research also includes in vitro studies to evaluate the total flavonoid and phenolic content as well as the antioxidant activity of the extract. The findings of this investigation could facilitate the development of novel herbal formulations that capitalize on the therapeutic properties of Zingiber barbatum, providing an alternative to conventional drugs with minimal side effects. Ultimately, this research not only confirms the traditional uses of Z. barbatum but also uncovers new insights, laying the groundwork for its future applications in modern medicine.
Materials and Methods
Plant material and extraction
In the Chittagong hill area of Bangladesh, the rhizomes of Zingiber barbatum were obtained, and the Bangladesh National Herbarium verified a voucher specimen (DACB 94773). The rhizomes were shade-dried for ten days to preserve essential oils, then the material was crushed and retained in a hermetic container. To extract the desired components, a quantity of 600 grams of powdered leaves was submerged in 3 liters of 80% methanol solution within a sealed glass container. For 16 days, the mixture was left undisturbed, with occasional shaking. The combination underwent a sequential filtration process using a clean cloth, followed by cotton, and ultimately Whatman filter paper, resulting in the acquisition of a transparent methanolic extract. The extract was condensed using a rotary evaporator, then dried with forced air to get a dark brown crude methanolic extract of Zingiber barbatum.21
Experimental animals
Swiss albino mice (22–27 g), aged six to seven weeks, had been purchased from the animal facility at Jahangirnagar University in Bangladesh. Only animals exhibiting normal behaviour and free from any signs of illness, injury, or infection were included. Exclusion criteria involved animals showing signs of disease, injury, abnormal behaviour, or stress; pregnant or lactating females; those not meeting the specified age or weight range; and animals with prior exposure to test substances or experimental procedures. Following a 12-hour light/dark cycle, they were housed in plastic enclosures approximately 40 cm × 30 cm × 17 cm. The temperature was preserved at 25.0 ± 2ºC, and they had unrestricted access to food and water, as well as adequate air circulation. Before the experiments, mice were subjected to a seven-day course of adaptation. During the experiment, the mice were divided into five groups (Group I to Group V) of five animals each. Group I was the control group, Group II was the positive control and Groups III, IV, and V received 100 mg/kg, 200 mg/kg and 400 mg/kg methanolic extract of Z. barbatum, respectively. The Ethics Committee of Noakhali Science & Technology University, Bangladesh, pre-approved all operations (ethical number– NSTU/SCI/EC/2023/173).
Reagents and chemicals
We used high-quality, analytical-grade chemicals from multiple suppliers. The Folin-Ciocalteu reagent and methanol were obtained from Merck (Germany), while gallic acid, ascorbic acid, and carrageenan were obtained from Sigma-Aldrich (USA). Loba Chemie (India) supplied formaldehyde and acetic acid. Aspirin was employed as the reference drug in the acetic acid-induced writhing and formalin-induced paw lapping experiments, ketorolac in the heated plate test, and paracetamol in the antipyretic test. All of these drugs were obtained from Beximco Pharmaceuticals (Bangladesh). ACI Limited (Bangladesh) supplied the indomethacin used in the carrageenan-induced paw edema test, while Popular Pharmaceuticals Limited provided the experimental media, a 0.9% NaCl solution.
Quantitative analysis
Total phenolic content estimation
The Folin-Ciocalteu reagent method was employed to determine the total phenolic content.22 The calibration curve was set up by utilizing gallic acid at concentrations extending from 0 to 300 µg/ml. The Folin-Ciocalteu reagent was diluted with refined water by a factor of 10. A 1 mg/ml plant extract was combined with a 7.5% sodium carbonate solution, vortexed for 15 seconds, and that left to sit in the dark at room temperature for 30 minutes. The absorbance was measured at 760 nm employing a blank as a reference. The overall phenolic substance was measured three times for precision and evaluated in milligrams of gallic acid equivalents per gram of extract.
Total flavonoid content estimation
The aluminium chloride colorimetric technique was utilized to calculate the total flavonoid content (TFC) of the plant extract.23 We merged 500 µL of plant extract with 1.5 mL of 95% ethanol, 0.1 mL of 1M potassium acetate, 0.1 mL of 10% aluminum chloride, and 2.8 mL of distilled water. The absorbance was obtained at 415 nm after 40 minutes of exposure to ambient temperature. The Total Flavonoid Content (TFC) in mg quercetin equivalents (QE) per gram of extract has been calculated by employing a quercetin calibrated curve (0-50 µg/mL).
Total antioxidant capacity (TAC)
The method was followed with slight adjustments to assess the antioxidant activity of Zingiber barbatum extract.24 A 2:4:4 reagent solution has been created by mixing sodium phosphate (28 mM), ammonium molybdate (4 mM), and sulfuric acid (0.6 M). The reagent solution was blended with a 0.3 mL test sample (1 mg/mL) and heated at 95°C for 90 minutes. The absorbance was obtained at 695 nm with methanol as the reference after settling to ambient temperature. A standard curve has been generated by utilizing ascorbic acid (5.0–500 µg/mL), and the antioxidant activity of the extract is expressed as milligram ascorbic acid equivalent per gram of dried extract (mg AAE/g).
Analgesic activity
Acetic acid induced the writhing model in mice
The methodology for this study was adapted from Sigmund’s approach.25 Five groups of mice, each with five animals, were used after an 18-hour fast. The control group, denoted as Group 1, received oral saline at a dosage of 10 ml/kg, while Group 2 was delivered aspirin at a dosage of 100 mg/kg BW. The other subjects were administered Zingiber barbatum methanol extract (ZbME) at amounts of 100, 200, and 400 mg/kg BW. To induce pain, all groups were intraperitoneally injected with 0.2 ml of 3% acetic acid one hour after treatment. Writhing responses were tracked for a period of 5 to 15 minutes. The writhing response was identified by specific behaviors, such as abdominal contractions and limb extensions. Data were analyzed to calculate the average percentage inhibition of writhing (PIW):
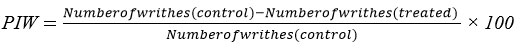
Formalin induced nociception model
For this experiment, we followed the methodology of Hunskaar and Hole, 1987 with slight modifications.26 The twenty-five animals were divided into five groups of five. Normal saline (10 ml/kg) was provided to the control group, whereas aspirin (100 mg/kg) was provided to the second group. ZbME was given to the remaining three groups at doses of 100, 200, and 400 mg/kg. Each treatment was given orally one hour beforehand to the formalin test, which comprised a subcutaneous injection of 20 µl of 1% formalin into the lateral left hind paw of the respective mouse. The neurogenic phase (0–5 minutes) was characterized by licking or biting behavior, while the inflammatory phase was observed 20–30 minutes after injection. The findings have been displayed as the average percentage inhibition of licking (PIL):
![]()
Hot plate test
The study involved 25 mice divided into five groups of five. A heated plate analgesiometer (Orchid, India) maintained at 55 ± 0.5ºC was used for testing. Mice responding to the heat stimulus within 10 seconds were selected. The control group, Group 1, received 10 ml/kg of normal saline. Group 2 received 10 mg/kg BW of ketorolac (reference or standard). Groups 3, 4, and 5 received the ZbME at 100, 200, and 400 mg/kg, respectively. Response times to the heat stimulus were recorded at 0, 0.5, 1, and 2 hours after treatment, measured as the time between placement on the plate and a pain response (jumping or paw licking). A 20-second cutoff was used to prevent tissue injury.27 Results were expressed as the average percentage of the maximum effect (%MPE) to assess the analgesic effect.28
![]()
Antipyretic test
The test was performed with minor alterations to the protocol described by Liu et al., 2017.29 A glycerin-coated digital thermometer was used for accurate rectal temperature measurements. Baseline temperatures were recorded 18 hours before treatment. The experiment involved five groups of five mice each. A 30% Baker’s yeast suspension in saline was injected subcutaneously (10 ml/kg). Mice with at least a 0.5°C/1.5°F rise in rectal temperature after 18 hours were selected, indicating hyperthermia. The initial group was given 10 ml/kg of saline during the treatment, while the second group was provided 150 mg/kg of Paracetamol. ZbME was provided to the third, fourth, and fifth groups at concentrations of 100, 200, and 400 mg/kg, respectively. Rectal temperatures were recorded at 30 minutes, 1 hour, 2 hours, 3 hours, and 24 hours post-treatment.
Anti-inflammatory activity
The anti-inflammatory activity was assessed using the carrageenan-induced paw edema test, as described by Igbe and Inarumen, 2013.30 The experiment involved five groups, each containing five rats. The control group received saline at a dose of 10 mg/kg, while the standard group was treated with indomethacin. The other three groups were administered ZbME at doses of 100, 200, and 400 mg/kg. Edema was induced by injecting 1% carrageenan (in 0.9% NaCl) into the hind paws of the rats after treatment. Paw thickness was recorded at 0, 1, 2, 3, and 4 hours, with changes measured relative to the initial readings.
Statistical analysis
GraphPad Prism 8.0.1 was utilized to carry out the data assessment. The statistical value was calculated using an ANOVA complemented by Bonferroni’s post hoc test. The significance levels are stated as P < 0.0001 (****), P <0.001 (***), P <0.01 (**), and P <0.05 (*). The results are displayed as the mean ± SEM.
Results
Quantitative analysis
Total flavonoid and phenolic contents
An analysis was conducted on the methanolic extract of the Zinziber barbatum rhizome to determine its total phenolic and flavonoid contents. The results were represented in terms of gallic acid and quercetin equivalents, respectively. [Figure 1 and Figure 2] represent the Standard curve of Total phenolic Content and Total flavonoid Content of Z. barbatum methanol extract (ZbME), respectively.
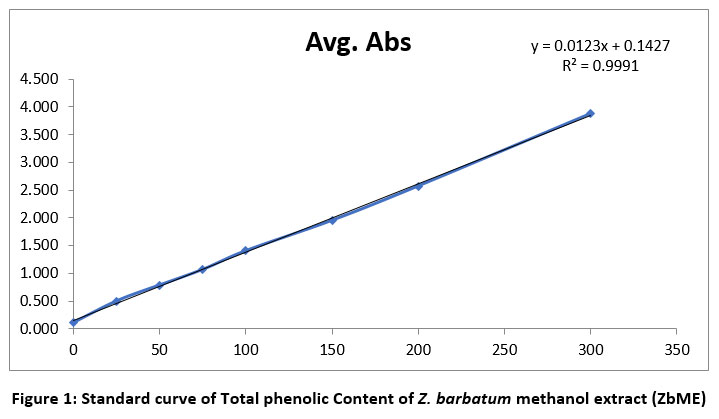 |
Figure 1: Standard curve of Total phenolic Content of Z. barbatum methanol extract (ZbME) |
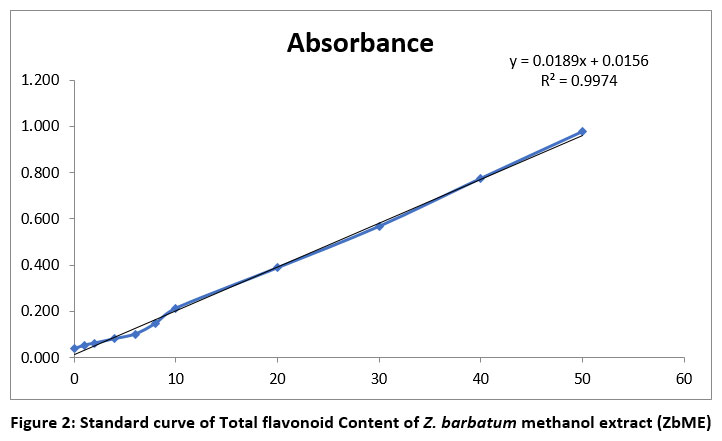 |
Figure 2: Standard curve of Total flavonoid Content of Z. barbatum methanol extract (ZbME) |
The crude extract had an average total phenolic content of 70.675 mg GAE per gram, and the dried sample had a total flavonoid content of 25.365 mg QE per gram [Table 1 and Figure 3].
Table 1: Total phenolic, flavonoid Content and Total antioxidant capacity of Z. barbatum methanol extract (ZbME) and Standard
| Total Phenolic Content | Total Flavonoid Content | Total Anti-oxidant Capacity | ||||
| Standard | Avg. Abs | Stdv. | Avg. Abs | Stdv. | Avg. Abs | Stdv. |
| 0.104 | 0.007 | 0.039 | 0.006 | 0.000 | 0.000 | |
| 0.496 | 0.085 | 0.054 | 0.011 | 0.059 | 0.014 | |
| 0.784 | 0.085 | 0.063 | 0.011 | 0.070 | 0.019 | |
| 1.064 | 0.057 | 0.082 | 0.007 | 0.103 | 0.012 | |
| 1.403 | 0.150 | 0.102 | 0.022 | 0.161 | 0.027 | |
| 1.951 | 0.075 | 0.148 | 0.037 | 0.209 | 0.040 | |
| 2.572 | 0.219 | 0.213 | 0.021 | 0.338 | 0.017 | |
| 3.879 | 0.376 | 0.388 | 0.014 | 0.469 | 0.055 | |
| Extract | 1.012 | 0.167 | 0.495 | 0.016 | 1.003 | 0.039 |
| Results | 70.675 mg GAE/g DW | 25.365 mg QE/g DW | 339.258 mg AAE/g DW | |||
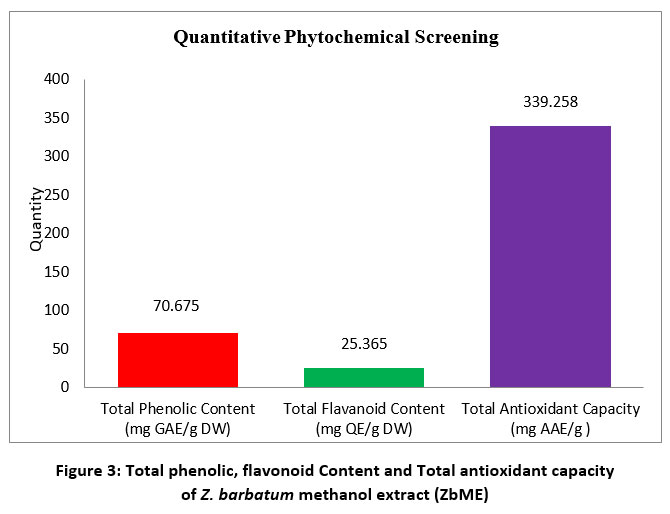 |
Figure 3. Total phenolic, flavonoid Content and Total antioxidant capacity of Z. barbatum methanol extract (ZbME) |
Total antioxidant capacity (TAC)
The standard curve of Total antioxidant capacity of Z. barbatum methanol extract (ZbME) was shown in [Figure 4]. The total antioxidant capacity of the extract was found to be 339.258 mg AAE/g DW extract [Figure 1].
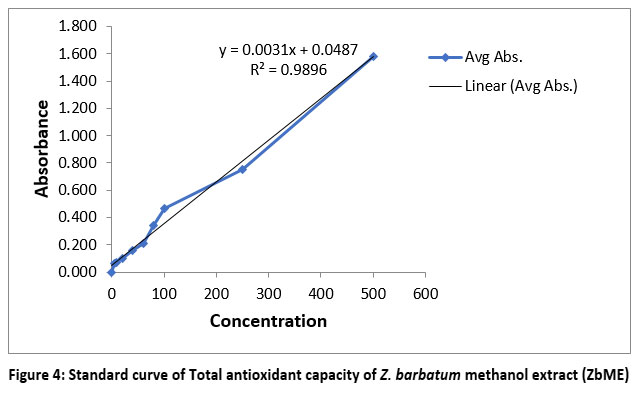 |
Figure 4: Standard curve of Total antioxidant capacity of Z. barbatum methanol extract (ZbME) |
Analgesic activity
Acetic acid induced writhing method
The writhing response was notably lowered in a dose-dependent manner by the methanolic extract of Zingiber barbatum rhizomes. [Table 2 and Figure 5] illustrate that the reduction in writhes was 47.18% at a dosage of 100 mg/kg, 64.08% at 200 mg/kg, and 66.67% at 400 mg/kg, with significance levels of P < 0.01, P < 0.001, and P < 0.001, respectively. For comparison, the standard reference drug, aspirin, showed a 76.92% inhibition of the writhing response in the same test, with a significance level of P < 0.001, indicating a strong analgesic effect.
Table 2: The effects of ZbME and Aspirin on the acetic acid-induced writhing test in mice
| Group | Writhing count | % Inhibition of wreathing (PIW) |
| Control | 39.00± 2.429 | _ |
| Standard (Aspirin) | 9.00 ± 1.517*** | 76.92 |
| ZbME 100mg/kg | 20.60 ± 5.046** | 47.18 |
| ZbME 200mg/kg | 14.40 ± 2.977*** | 63.08 |
| ZbME 400mg/kg | 13.00 ± 1.304*** | 66.67 |
All values are given in mean ± SEM. N = 5. P < 0.001 (***) and P < 0.01 (**) P < 0.05 (*) as compared to control (One-way ANOVA followed by Bonferroni’s test).
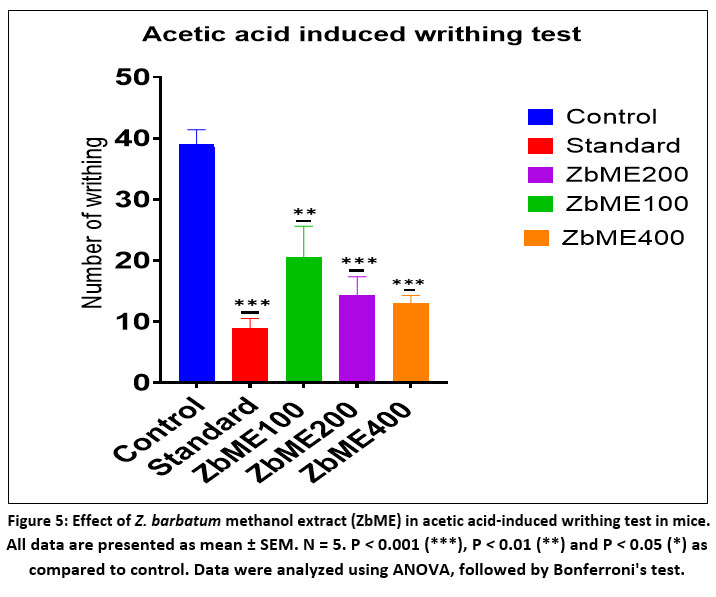 |
Figure 5: Effect of Z. barbatum methanol extract (ZbME) in acetic acid-induced writhing test in mice. |
Formalin induced paw-licking test
Zingiber barbatum rhizomes methanolic extract (ZbME) substantially decreased the licking response time in both stages of the test. Aspirin, the standard drug, significantly reduced licking response time in both phases (P < 0.001) with inhibition rates of 63.43% in the early phase and 63.05% in the late phase. The 200 mg/kg and 400 mg/kg ZbME doses had highly significant effects (P < 0.001) in the early phase, with the 400 mg/kg dose demonstrating the greatest reduction. The late phase results were comparable, with the 400 mg/kg dose once more displaying the most pronounced effect (P < 0.001), while the 200 mg/kg dose also had a significant impact (P < 0.01). The percentage inhibition of licking in the early phase was 34.37%, 48.75%, and 62.05% for dosages of 100, 200, and 400 mg/kg, respectively. In the late phase, these percentages were 32.52%, 53.04%, and 63.80%, respectively [Table 3 and Figure 6].
Table 3: Effect of ZbME and aspirin on the percentage inhibition of paw licking response.
| Group | % Inhibition of licking (PIL) | |
| Early phase (0-5 min) | Late phase (20-30 min) | |
| Standard (Aspirin) | 63.43 | 63.05 |
| ZbME 100mg/kg | 34.37 | 32.52 |
| ZbME 200mg/kg | 48.75 | 53.04 |
| ZbME 400mg/kg | 62.05 | 63.80 |
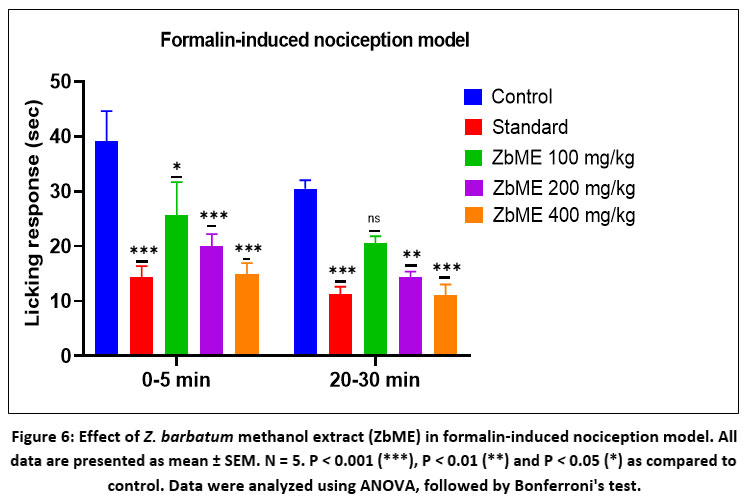 |
Figure 6. Effect of Z. barbatum methanol extract (ZbME) in formalin-induced nociception model. |
Hot plate test
The pre-treatment of animals with the extract at a dose of 400 mg/kg and with the standard drug ketorolac at a dose of 10 mg/kg resulted in a significantly prolonged response in terms of latency time. This effect was observed at 30,60,90 and 120 min post-administration, with a significance level of P < 0.001 [Figure 7].
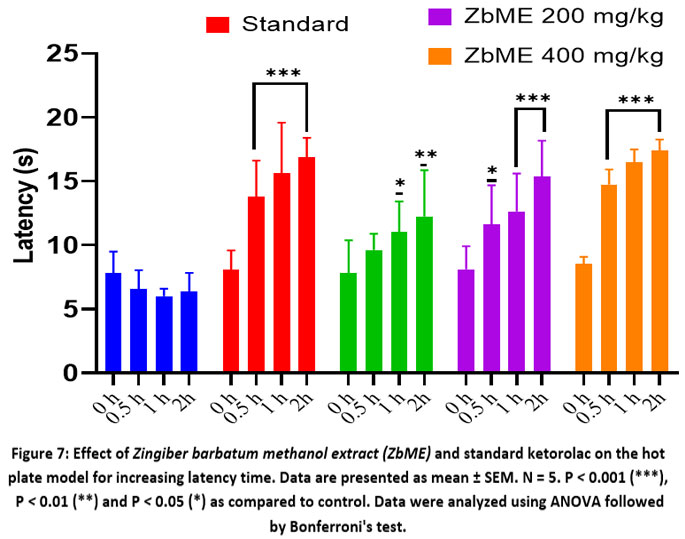 |
Figure 7: Effect of Zingiber barbatum methanol extract (ZbME) and standard ketorolac on the hot plate model for increasing latency time. |
Notably, the 400 mg/kg extract exhibited a slightly longer latency response compared to ketorolac across these time points. Additionally, animals pre-treated with the extract at 200 mg/kg also demonstrated a significant (P < 0.001) increase in latency time, particularly at the 90 and 120-minute marks [Figure 8].
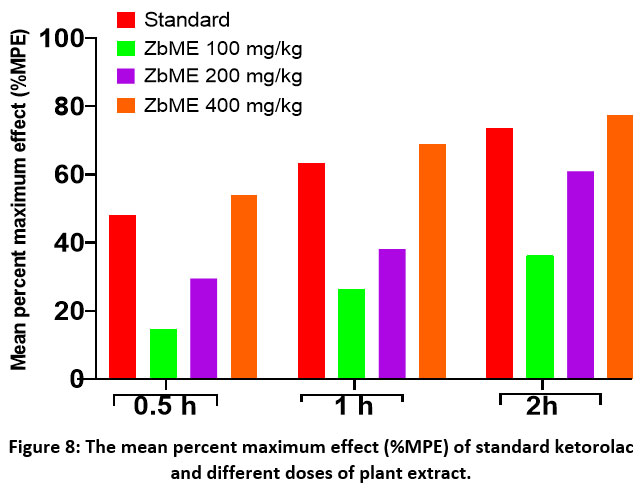 |
Figure 8: The mean percent maximum effect (%MPE) of standard ketorolac and different doses of plant extract. |
Furthermore, the ZbME extract at 400 mg/kg showed a greater mean percent maximal effect at 0.5, 1, and 2 hours compared to the standard. Specifically, the standard drug produced mean percent maximal effects of 48.12%, 63.35%, and 73.72% at 0.5, 1, and 2 hours, respectively. In contrast, the ZbME extract at 400 mg/kg yielded superior effects with mean percent maximal effects of 54.01%, 69.02%, and 77.40% at the corresponding time points [Figure 8].
Antipyretic test
Eighteen hours after administering Baker’s yeast, all animals exhibited elevated rectal temperatures due to the release of pyrogens [Table 4].
Table 4: Antipyretic effect of Zingiber barbatum methanol extract (ZbME) on Baker’s yeast-induced pyrexia in mice
| Treatment group | Rectal temperature (◦F) # | Rectal temperature recorded after respective treatment (◦F) | ||||
| 0.5h | 1h | 2h | 3h | 24h | ||
| Control | 99.54± 0.20 | 99.62±0.11 | 100.36±0.20 | 100.46±0.17 | 100.84±0.15 | 101.16±0.28 |
| Standard (Paracetamol
150mg/kg) |
99.18±0.32 | 98.80±0.24 | 98.56±0.24*** | 98.02±0.29*** | 96.66±0.22*** | 97.48±0.26*** |
| ZbME 100mg/kg | 99.48±0.31 | 98.92±0.32 | 99.20±0.30 | 99.36±0.35 | 99.88±0.47 | 99.44±0.32*** |
| ZbME 200mg/kg | 99.48±0.32 | 99.28±0.30 | 98.42±0.25*** | 97.90±0.33*** | 96.90±0.22*** | 97.42±0.20*** |
| ZbME 400mg/kg | 98.98±0.63 | 98.22±0.47 * | 97.56±0.28*** | 96.90±0.22*** | 96.32±0.24*** | 96.76±0.15*** |
#Rectal temperature after 18 h of yeast injection. All data are presented as mean ± SEM. N = 5. P < 0.001 (***), P < 0.01 (**) and P < 0.05 (*) as compared to control. Data were analyzed by using ANOVA followed by Bonferroni’s test.
Paracetamol and plant extracts were given to subjects at concentrations of 100 mg/kg, 200 mg/kg, and 400 mg/kg to assess their antipyretic effects. Body temperature changes were monitored at 0.5, 1, 2, 3, and 24 hours following the treatment. In comparison to the control, the results showed that both paracetamol and plant extracts at 200 mg/kg and 400 mg/kg significantly lowered elevated rectal temperatures (P < 0.001) [Table 4]. Paracetamol at 150 mg/kg resulted in a temperature reduction of 2.52°F three hours after treatment, while plant extracts at 200 mg/kg and 400 mg/kg reduced it by 2.58°F and 2.66°F, respectively.
Anti-inflammatory activity
This study evaluated the anti-inflammatory effects of crude Zingiber barbatum rhizome extract at doses of 100 mg/kg, 200 mg/kg, and 400 mg/kg, using Indomethacin as the standard reference drug [Table 5].
Table 5: Effect of the Zingiber barbatum methanol extract (ZbME) on carrageenan-induced paw oedema in mice
| Treatment mg/kg | Paw thickness (mm) | ||||
| 0h | 1h | 2h | 3h | 4h | |
| Control | 3.09± 0.12 | 4.21±0.25 | 4.16±0.30 | 4.01±0.23 | 4.02±0.30 |
| Standard
(Indomethacin) |
2.91±0.17 | 3.92±0.34(06.89%) | 3.52±0.13**** (15.38%) | 3.17 ±0.10**** (20.95%) | 3.06±0.11**** (23.88%) |
| RpME
100mg/kg |
3.06±0.11 | 4.10±0.27 (02.61%) | 3.76±0.34*
(09.61%) |
3.46±0.27*** (13.72%) | 3.36±0.15**** (16.42%) |
| RpME
200mg/kg |
2.99±0.14 | 4.07±0.30(03.33%) | 3.60±0.21*** (13.46%) | 3.38±0.13*** (15.71%) | 3.10±0.12**** (22.89%) |
| RpME
400mg/kg |
2.95±0.19 | 3.90±0.30*(07.36%) | 3.38±0.28**** (18.75%) | 3.16±0.11**** (21.20%) | 3.00±0.18**** (25.37%) |
Result data are reported as mean ± SEM. N = 5. P < 0.0001(****) P < 0.001 (***), P < 0.01 (**) and P< 0.05 (*) as compared to control.
The 400 mg/kg extract dose and Indomethacin both exhibited the most significant anti-inflammatory activity, particularly at the 2nd, 3rd, and 4th hours following carrageenan administration. Specifically, the 400 mg/kg extract dose demonstrated inhibition rates of 18.75%, 21.20%, and 25.37% at the 2nd, 3rd, and 4th hours, respectively, which were greater than those observed with Indomethacin (15.38%, 20.95%, and 23.88%) at the same time points (P < 0.0001). The 100 mg/kg dose of the extract showed significant anti-inflammatory activity at the 3rd hour (P < 0.001), with a maximum inhibition of 16.42% at the 4th hour (P < 0.0001). Meanwhile, the 200 mg/kg dose exhibited significant activity at both the 2nd and 3rd hours (P < 0.001) and achieved maximum inhibition of 22.89% at the 4th hour (P < 0.0001) [Table 5].
Discussion
The objective of this investigation was to verify the conventional application of Zingiber barbatum by verifying its antipyretic, analgesic, and anti-inflammatory properties. The phenolic and flavonoid chemicals found in ginger were mostly responsible for its pharmacological activity.19,20 There are a number of pharmacological actions exhibited by the phenolic compound, such as antioxidant, analgesic, anti-inflammatory, and antipyretic capabilities. Furthermore, flavonoids have a number of pharmacological effects, one of which is an anti-inflammatory effect.31,32 A study found that ginger is rich in flavonoids, key compounds contributing to its anti-inflammatory effects.33 Additionally, the phenolic-rich extract of Zingiber barbatum showed a total antioxidant capacity of 339.258 mg ascorbic acid equivalent per gram, highlighting its potential as a valuable antioxidant source.
To assess analgesic activity in animals, various methods have been developed that observe changes in response to painful stimuli after administering an active substance. To induce peripheral pain in animals, the writhing test is a chemical assay that involves the injection of irritants such as acetic acid. This method assesses the efficacy of analgesics by observing a decrease in the frequency of painful behaviors, such as back arching, hind limb extension, and abdominal muscle contractions.34,35 Acetic acid triggers the release of prostaglandins, histamine, and pro-inflammatory mediators which contribute to peripheral pain. This response is employed to evaluate the efficacy of analgesics that operate on both peripheral and central levels. Zingiber barbatum’s analgesic properties are attributed to its capacity to impede the release of endogenous pain mediators, such as prostaglandins. This implies that Zingiber barbatum may have an additional inhibitory effect on the cyclooxygenase pathway, which is critical for prostaglandin biosynthesis.36
The Zingiber plant extract diminished writhing in a dose-dependent way compared to the control. The ZbME at 200 mg/kg and 400 mg/kg appeared significant writhing inhibition, with P-values < 0.001 (***), achieving percentage inhibitions of 63.08% and 66.67% respectively [Table 2 and Figure 5]. This impact may be due to interference with pain receptor activation or disruption of mediators involved. The formalin test is an invaluable model for the investigation of chronic pain, as it replicates tissue injury and illustrates a variety of pain mechanisms, such as central, neurogenic, and inflammatory pain. A biphasic pain response is the consequence of a localized paw lesion that is induced in animals in this test. Neurogenic pain takes place in the initial phase as a result of direct nociceptor stimulation, whereas inflammatory pain is present in the later phase as a result of the release of pro-inflammatory mediators. During the early stage, the licking response was substantially reduced (P < 0.001) by ZbME administration at 200 mg/kg and 400 mg/kg in comparison to the control group [Figure 6]. As outlined in [Figure 6], the licking response was moreover significantly diminished by ZbME at 400 mg/kg within the late stage (P < 0.001). Noteworthy, ZbME at 400 mg/kg shown a comparable rate inhibition of paw licking to the conventional aspirin in both stages (Table 3). The impact may be linked to a decline in the release or blocking of pro-inflammatory mediators. The Hot Plate test is employed to identify compounds that possess central antinociceptive effects.37 It functions as an efficient model for the evaluation of centrally acting analgesics, which are recognized for their capacity to extend the pain threshold in animals when they are subjected to heat. Compared to the control group, the 400 mg/kg Zingiber barbatum rhizome extract significantly increased the mean maximal effect (% MPE) [Figure 8]. The extract’s powerful pain-relieving action, which is likely intervened through central mechanisms, is proposed by the considerable increase in % MPE. Therefore, Z. barbatum rhizomes exhibit analgesic properties that are mediated by both peripheral and central pathways, suggesting their potential as a supplement to current analgesics.
Endogenous cytokines mediate fever, also known as pyrexia, a protective response that is induced by a number of clinical diseases.38 Symptoms include a febrile reaction—an abnormally high body temperature caused by an elevation in the body’s temperature set point. Infection, tissue damage, inflammation, cancer, graft rejection, and other diseases can trigger fever as a secondary symptom. Fever is the body’s way of protecting itself from harmful invaders or injured tissues by making the surrounding environment too hot for them to survive. The extract exhibited the most substantial antipyretic effect at an amount of 400 mg/kg among the concentrations that were evaluated, with a maximal temperature decrease of 2.66°F at the 3-hour point. This extract also consistently outperformed the standard paracetamol in reducing temperature at all observed time points [Table 4]. Most antipyretic drugs work by inhibiting the expression of COX-2, which in turn prevents the biosynthesis of PGE2 and helps lower the elevated body temperature. Two research found similar results, suggesting that ginger might be an even more potent inhibitor of prostaglandin (PGE2) synthesis.39,40 The components of ginger also impede the metabolism of arachidonic acid, which in turn hinders the formation of prostaglandins.41
The conventional utilization of Zingiber barbatum (ginger) in for the management of inflammation is now supported by the current research, which provides a pharmacological rationale. In the initial and later stages of inflammation, the methanol extract of ginger demonstrated substantial anti-inflammatory properties, successfully minimizing edema in the rat paw tissues. Inflammatory chemical mediators, including histamine, serotonin, and bradykinin, accelerate the development of edema. The initial stage, which occurs within one hour, is mainly driven by histamine and serotonin. The subsequent stage, which occurs three to four hours after treatment, encompasses bradykinin, prostaglandins, and other mediators.42,43
This investigation demonstrated that ginger extract significantly reduced paw edema and the proportion of edema inhibition in contrast to the control group and the group that received indomethacin. It is noteworthy that ginger exhibited anti-inflammatory properties that were comparable to those of indomethacin when delivered at an amount of 400 mg/kg, with enhanced efficacy observed at specific time intervals [Table 5]. The active constituents of ginger tend to inhibit the actions of other inflammatory mediators and to suppress the secretion of histamine and serotonin from mast cells, as indicated by the outcomes. Prior research has corroborated these findings; Sharma et al., 1994 demonstrated that ginger suppressed paw edema in rats with experimentally induced arthritis,44 while Srivastava and Mustafa, 1992 reported that 75% of individuals with arthritis, osteoarthritis, or muscular pain experienced a reduction in pain and swelling as a result of extended ginger therapy.45 In a separate investigation, it was shown that the anti-inflammatory properties of ginger on osteoarthritic cow chondrocytes were linked to its ability to suppress COX-2, pro-inflammatory cytokines, and prostaglandins.46
Conclusion
This study demonstrates that the methanolic extract of Zingiber barbatum rhizomes exhibits significant anti-inflammatory, analgesic, and antipyretic activities, supporting its traditional medicinal applications. The findings suggest its potential as a safer and more natural alternative to conventional NSAIDs, potentially minimizing adverse effects associated with synthetic drugs. Molecular docking analyses further identified promising interactions between key phytoconstituents and relevant biological targets, highlighting bioactive compounds that merit further isolation and characterization. Overall, this research advances the scientific basis for the therapeutic use of Z. barbatum and opens new avenues for the development of evidence-based phytomedicines in modern healthcare.
Acknowledgement
The authors would like to express their heartfelt obligation, indebtedness, Gramercy and profound appreciation to the Department of Pharmacy, Noakhali Science and Technology University, Sonapur-3814, Noakhali, Bangladesh for their general facilities continuous support, untiring inspiration, scholastic supervision, constructive criticism, affectionate feeling and optimistic counselling for the conduction of this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The Noakhali Science & Technology University, Bangladesh, Ethics Committee approved the experimental methodology in conformity with animal research rules (NSTU/SCI/EC/2023/173).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Syed Mahabubur Rahman: Conceptualisation, Methodology, Data Collection, Writing – Original Draft;
- Fahim Salek Gowrab: Data Collection, Writing – Original Draft;
- Shaiful Islam: Data Collection, Analysis;
- Sabiha Tabassum: Editing, Project Administration;
- Md. Shafiul Hossen: Project Administration;
- Arafat Miah: Writing – Original Draft, Editing;
- Mohammad Safiqul Islam: Software, Visualisation, Supervision;
- Md Abdul Barek: Conceptualization, Methodology, Analysis, Supervision
References
- Talebi M, İlgün S, Ebrahimi V, et al. Zingiber officinale ameliorates Alzheimer’s disease and Cognitive Impairments: Lessons from preclinical studies. Biomed Pharmacother. 2021;133:111088.
CrossRef - Srinivasan K. Ginger rhizomes (Zingiber officinale): A spice with multiple health beneficial potentials. PharmaNutrition. 2017;5(1):18-28.
CrossRef - Ali BH, Blunden G, Tanira MO, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): a review of recent research. Food Chem Toxicol an Int J Publ Br Ind Biol Res Assoc. 2008;46(2):409-420.
CrossRef - Mahboubi M. Zingiber officinale Rosc. essential oil, a review on its composition and bioactivity. Clin Phytoscience. 2019;5(1):6.
CrossRef - Mishra RK, Kumar A, Kumar A. Pharmacological activity of Zingiber officinale. Int J Pharm Chem Sci. 2012;1:1073-1078.
- Hafez DA. Effect of extracts of ginger goots and cinnamon bark on fertility of male diabetic rats. J Am Sci. 2010;6(10):940-947.
- Shukurova MK, Asikin Y, Chen Y, Kusano M, Watanabe KN. Profiling of volatile organic compounds in wild Indigenous medicinal ginger (Zingiber barbatum Wall.) from Myanmar. Metabolites. 2020;10(6).
CrossRef - Mao QQ, Xu XY, Cao SY, et al. Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods (Basel, Switzerland). 2019;8(6).
CrossRef - Aung MM. Taxonomic study of the genus Zingiber Mill. (Zingiberaceae) in Myanmar. Published online 2016.
- Awale S, Linn TZ, Than MM, Swe T, Saiki I, Kadota S. The healing art of traditional medicines in Myanmar. J Tradit Med. 2006;23(2):47-68. doi:10.11339/jtm.23.47
- Wicaksana N. Characterization and diversity assessment of Zingiber species with special reference to underutilized medicinal Z. Barbatum species from Myanmar. Tsukuba University; 2012.
- Wicaksana N, Gilani SA, Ahmad D, Kikuchi A, Watanabe KN. Morphological and molecular characterization of underutilized medicinal wild ginger (Zingiber barbatum Wall.) from Myanmar. Plant Genet Resour. 2011;9(4):531-542.
CrossRef - Chen L, Deng H, Cui H, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9(6):7204.
CrossRef - Conti B, Tabarean I, Andrei C, Bartfai T. Cytokines and fever. Front Biosci. 2004;9:1433-1449.
CrossRef - Wautier JL, Wautier MP. Pro-and anti-inflammatory prostaglandins and cytokines in humans: a mini review. Int J Mol Sci. 2023;24(11):9647.
CrossRef - Mukhopadhyay N, Shukla A, Makhal PN, Kaki VR. Natural product-driven dual COX-LOX inhibitors: Overview of recent studies on the development of novel anti-inflammatory agents. Heliyon. 2023;9(3):e14569.
CrossRef - Sukatta U, Rugthaworn P, Punjee P, Chidchenchey S, Keeratinijakal V. Chemical Composition and physical properties of oil from Plai (Zingiber cassumunar Roxb.) Obtained by Hydro Distillation and Hexane Extraction. Kasetsart J – Nat Sci. 2009;43:212-217.
- Sharifi-Rad M, Varoni EM, Salehi B, et al. Plants of the genus Zingiber as a source of bioactive phytochemicals: from tradition to pharmacy. Molecules. 2017;22(12).
CrossRef - Danciu C, Vlaia L, Fetea F, et al. Evaluation of phenolic profile, antioxidant and anticancer potential of two main representants of Zingiberaceae family against B164A5 murine melanoma cells. Biol Res. 2015;48(1):1.
CrossRef - Ha SK, Moon E, Ju MS, et al. 6-Shogaol, a ginger product, modulates neuroinflammation: a new approach to neuroprotection. Neuropharmacology. 2012;63(2):211-223.
CrossRef - Reza MS, Jashimuddin M, Ahmed J, et al. Pharmacological investigation of analgesic and antipyretic activities of methanol extract of the whole part of Aeginetia indica. J Ethnopharmacol. 2021;271:113915.
CrossRef - Meda A, Lamien CE, Romito M, Millogo J, Nacoulma OG. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005;91(3):571-577.
CrossRef - Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colometric methods. J Food Drug Anal. 2002;10(3):178-182.
CrossRef - Kubola J, Siriamornpun S. Phenolic contents and antioxidant activities of bitter gourd (Momordica charantia L.) leaf, stem and fruit fraction extracts in vitro. Food Chem. 2008;110(4):881-890.
CrossRef - Sandhiutami NM, Khairani S, Dewi RS, Hakim ZR, Pradani AR. Anti-inflammatory and analgesic activity of musa balbisiana peels in vivo. Borneo J Pharm. 2022;5(2):81-92.
CrossRef - Hunskaar S, Hole K. The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain. 1987;30(1):103-114.
CrossRef - Shahnaz M, Biswas P, Akhter M, Rafiq K, Ali T. Effects of Spirulina platensis on neuropathic pain in Wistar rats. J Bangladesh Soc Physiol. 2021;16(1):1-0.
CrossRef - Dewey WL, Harris LS, Howes JF, Nuite JA. The effect of various neurohumoral modulators on the activity of morphine and the narcotic antagonists in the tail-flick and phenylquinone tests. J Pharmacol Exp Ther. 1970;175(2):435-442.
CrossRef - Liu C, Su H, Wan H, et al. Forsythoside A exerts antipyretic effect on yeast-induced pyrexia mice via inhibiting transient receptor potential vanilloid 1 function. Int J Biol Sci. 2017;13(1):65-75.
CrossRef - Igbe I, Inarumen GO. The effect of leaf aqueous extract of Brachystegia Eurycoma Harms (Fabaceae) in acute and chronic inflammatory animal models. J Pharm Res Int. 2013;3(3):391–400.
CrossRef - Al-Khayri JM, Sahana GR, Nagella P, et al. Flavonoids as potential anti-inflammatory molecules: A review. Molecules. 2022;27(9):2901.
CrossRef - Read MA. Flavonoids: naturally occurring anti-inflammatory agents. Am J Pathol. 1995;147(2):235-237.
- Ilavarasan R, Mallika M, Venkataraman S. Anti-inflammatory and free radical scavenging activity of Ricinus communis root extract. J Ethnopharmacol. 2006;103(3):478-480.
CrossRef - Fish R. Anesthesia and analgesia in laboratory animals. Academic press; 2015.
- Gawade SP. Acetic acid induced painful endogenous infliction in writhing test on mice. J Pharmacol Pharmacother. 2012;3(4):348.
CrossRef - Ma RH, Ni ZJ, Zhu YY, et al. A recent update on the multifaceted health benefits associated with ginger and its bioactive components. Food Funct . 2021;12(2):519-42.
CrossRef - Kim HY, Lee HJ, Zuo G, et al. Antinociceptive activity of the Caesalpinia eriostachys Benth. ethanolic extract, fractions, and isolated compounds in mice. Food sci nutr. 2022;10(7):2381-9.
CrossRef - Blomqvist A, Engblom D. Neural mechanisms of inflammation-induced fever. The Neuroscientist. 2018;24(4):381-99.
CrossRef - Gupta J, Sharma B, Sorout R, Singh RG, Sharma MC. Ginger (Zingiber officinale) in Traditional Chinese Medicine: A Comprehensive Review of Its Anti-Inflammatory Properties and Clinical Applications. Pharmacol Res Mod Chin Med. 2024:100561.
CrossRef - Thomson M, Al-Qattan KK, Al-Sawan SM, Alnaqeeb MA, Khan I, Ali M. The use of ginger (Zingiber officinale Rosc.) as a potential anti-inflammatory and antithrombotic agent. Prostaglandins Leukot Essent Fatty Acids. 2002;67(6):475-478.
CrossRef - Shen CL, Hong KJ, Kim SW. Effects of ginger (Zingiber officinale Rosc.) on decreasing the production of inflammatory mediators in sow osteoarthrotic cartilage explants. J Med Food. 2003;6(4):323-328.
CrossRef - Maling HM, Webster ME, Williams MA, Saul W, Anderson WJ. Inflammation induced by histamine, serotonin, bradykinin and compound 48-80 in the rat: antagonists and mechanisms of action. J Pharmacol Exp Ther. 1974;191(2):300-310.
CrossRef - Wallace JM. Nutritional and botanical modulation of the inflammatory cascade–eicosanoids, cyclooxygenases, and lipoxygenases–as an adjunct in cancer therapy. Integr Cancer Ther. 2002;1(1):7-37.
CrossRef - Sharma JN, Srivastava KC, Gan EK. Suppressive effects of eugenol and ginger oil on arthritic rats. Pharmacology. 1994;49(5):314-318.
CrossRef - Srivastava KC, Mustafa T. Ginger (Zingiber officinale) in rheumatism and musculoskeletal disorders. Med Hypotheses. 1992;39(4):342-348.
CrossRef - Murugesan S, Venkateswaran MR, Jayabal S, Periyasamy S. Evaluation of the antioxidant and anti-arthritic potential of Zingiber officinale Rosc. by in vitro and in silico analysis. S Afr J Bot. 2020;130:45-53.
CrossRef
ZbME; Zingiber barbatum Methanolic Extract,
NSAIDS; Non-Steroidal Anti-Inflammatory Drugs;
GAE; Gallic Acid Equivalent,
QE; Quercetin Equivalent, and
AAE; Ascorbic Acid Equivalent,
DW; Dry Weight.
TFC; Total Flavonoid Content,
TAC; Total Antioxidant Capacity,
PIW; Percentage Inhibition of Writhing,
PIL; Percentage Inhibition of Licking,
%MPE; Percent Maximal Possible Effect,
COX; Cyclooxygenase,
PGE2; Prostaglandin E2,
iNOS; Inducible Nitric Oxide Synthase,
ANOVA; Analysis of Variance,
SEM; Standard Error of the Mean,
BW; Body Weight







