Manuscript accepted on :15-05-2025
Published online on: 05-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Raju Thenge
Second Review by: Dr. Moumita Hazra
Final Approval by: Dr. Mariia Shanaida
Abdisalim A. Zaripov1,3 , Inoyat Z. Zhumaev1*
, Inoyat Z. Zhumaev1* , Pulat B. Usmanov 1
, Pulat B. Usmanov 1 , Yulduzkhon T. Mirzaeva1
, Yulduzkhon T. Mirzaeva1 , Shavkat Yu. Rustamov1
, Shavkat Yu. Rustamov1 , Sadriddin N. Boboev1
, Sadriddin N. Boboev1 , Adilbay T. Esimbetov1
, Adilbay T. Esimbetov1 , Gulnaz S. Begdullaeva1
, Gulnaz S. Begdullaeva1 , Sardor B. Sobirov1
, Sardor B. Sobirov1 , Eldor B. Ibragimov1
, Eldor B. Ibragimov1 , Sherzod N. Zhurakulov2
, Sherzod N. Zhurakulov2 , Dilbar D. Safarova3
, Dilbar D. Safarova3 , Ra'no I. Yusupova3
, Ra'no I. Yusupova3
1Institute of Biophysics and Biochemistry, National University of Uzbekistan, Tashkent, Uzbekistan.
2Institute of the Chemistry of Plant Substances, Uzbek Academy of Sciences, Tashkent, Uzbekistan.
3Uzbek State University of Physical Education and Sports, Chirchiq, Uzbekistan
Corresponding Author E-mail: inoyat8585@mail.ru
DOI : https://dx.doi.org/10.13005/bpj/3195
Abstract
The development of cardiovascular diseases is primarily associated with impaired activity of ion transport systems that ensure Ca2+ homeostasis in vascular smooth muscle cells. Modulation of the function of Ca2+ ion transport systems in smooth muscle cells with biologically active compounds allows the development of new approaches to the pharmacological regulation of Ca2+-dependent processes in cardiovascular diseases. This article studies the mechanisms of the vasorelaxant effect of the isoquinoline alkaloid 1-(3¢-Bromo-4¢-hydroxyphenyl-5¢-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-19) on the force of aortic ring contraction. It was found that the alkaloid F-19 exerts a concentration-dependent (5-100 μM) potent vasorelaxant effect on the force of aortic ring contraction induced by KCl and PE. The involvement of the L-type Ca2+ channel in the vasorelaxant effect of the alkaloid F-19 was investigated under conditions of incubation with its specific blocker verapamil. It was found that the role of the L-type Ca2+ channel in the vasorelaxant effect of this alkaloid is minor. The effect of the alkaloid F-19 on Ca2+ transport systems in the SR was also investigated. In this case, PE and caffeine-induced transient aortic contraction in the absence of Ca2+ significantly reduced. At the same time, a decrease in the vasorelaxant effect on aortic contractility was observed under the conditions of incubation with the SERCA inhibitor CPA, the alkaloid F-19. The vasorelaxant effect of the alkaloid F-19 on rat aortic smooth muscle contractility is mediated by several complex mechanisms. The vasorelaxant effect of this alkaloid is provided by inhibiting the entry of Ca2+ ions into the cytosol through plasma membrane potential-dependent L-type Ca2+ channels and the exit of Ca2+ ions into the cytosol through the SR Ca2+ transport systems, and by activating SERCA.
Keywords
Alakaloid; Ca-channels; endothelium; nitric oxide; smooth muscle
Download this article as:| Copy the following to cite this article: Zaripov A. A, Zhumaev I. Z, Usmanov P. V, Mirzaeva Y. T, Rustamov S. Y, Boboev S. N, Esimbetov A. T, Begdullaeva G. S, Sobirov S. B, Ibragimov E. B, Zhurakulov S. N, Safarova D. D, Yusupova R. I. Mechanism of Vasorelaxant Action of Isoquinoline Alkaloid F-19 on Rat Aorta. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Zaripov A. A, Zhumaev I. Z, Usmanov P. V, Mirzaeva Y. T, Rustamov S. Y, Boboev S. N, Esimbetov A. T, Begdullaeva G. S, Sobirov S. B, Ibragimov E. B, Zhurakulov S. N, Safarova D. D, Yusupova R. I. Mechanism of Vasorelaxant Action of Isoquinoline Alkaloid F-19 on Rat Aorta. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4kUzLrD |
Introduction
Cardiovascular diseases still occupy a leading position in the overall structure of morbidity and mortality in the world.1-3 The most common pathology of the cardiovascular system is arterial hypertension, which is one of the main pathophysiological risk factors for the development of most cardiovascular diseases.4 The development of arterial hypertension is mainly associated with a disruption of Ca2+ homeostasis, which regulates the contractile activity of vascular smooth muscle cells.5 In maintaining Ca2+ homeostasis in smooth muscle cells (SMCs), Ca2+ influx through voltage-gated L-type Ca2+ channels and the inositol 1,4,5-triphosphate receptor (IP3R) in the sarcoplasmic reticulum (SR) plays an important role.6,7 Also, participation of endothelium in SMC Ca2+ homeostasis and regulation of functional activity of blood vessels is considered important. Endothelial dysfunction leads to an increase in intracellular Ca2+ concentration through a decrease in NO synthase activity and disruption of the function of the SR Ca2+-ATPase and K+ channels.8 In this regard, the search for new approaches to correcting disorders of Ca2+ homeostasis in SMC is currently considered an urgent problem in cardiology and pharmacology. The solution to this problem will allow the development of effective methods for the prevention and treatment of arterial hypertension.9 The literature has shown that many isoquinoline alkaloids modulate cardiovascular diseases,10-12 have neuroprotective,13 analgesic,14 and other pharmacological properties.15 In addition, it has been described that it has a vasorelaxant effect on vascular SMСs.16 Therefore, the purpose of this study is to investigate the mechanisms of the vasorelaxant effect of the alkaloid F-19 on the contractile activity of the rat aortic ring.
Materials and Methods
Animals and aortic rings preparation.
White rats weighing 200-250 g were anesthetized with sodium pentobarbital, and the thorax was opened with scissors and the aorta was carefully dissected. Aortic vessels were cleaned from the upper layer of excess fat using tweezers and scissors, and aortic rings ~3-4 mm long were prepared. Aortic rings were set up in a custom-made Graz Glass Tissue Bath, a 5 ml vessel that was perfused with Krebs-Henseleit solution (mM): NaCl 120.4; КСl 5; NaHCO3 15.5; NaH2PO4 1.2; МgCl2 1.2; СaСl2 2.5; С6Н12О6 11.5; HEPES (рН 7.4).17,18 The Krebs-Henseleit solution was constantly aerated using a gas mixture of O2–95/СO2–5% at +37 °C. Aortic rings were attached to the cell using two stainless steel wire hooks. During the experiment, the international principles of the Declaration of Helsinki and the rules of human treatment of animals were followed when working with animals.
Aortic-ring contraction studies.
The force of aortic ring contraction was transmitted to a signal amplifier via a Grass FT.03 mechanotransducer (Grass-Telefactor, USA) and recorded on a computer using special software (PowerLab, Instruments). Each aortic ring was subjected to an initial tension corresponding to 1 g (∼10 mN) and incubated for 60 min before starting the experimental procedure. After incubation, experiments were conducted to study the vasorelaxant effect of the alkaloid F-19.
Experimental Protocols
Aortic smooth muscle contraction activity was measured with KCl and the α1-adrenoceptor agonist phenylephrine (PE), and the resulting contraction was set as 100% as a control.19 After the contraction force induced by KCl (50 mM) and PE (1 μM) reached a steady state, we studied their vasorelaxant effect by adding alkaloid F-19 at various concentrations (from 1 μM to 100 μM). Also, to determine the role of the endothelium in the vasorelaxant effect of F-19, experiments were conducted on aortic rings from which the endothelial layer was mechanically removed.
To determine the role of voltage-gated L-type Ca2+ channels in the vasorelaxant effect of the alkaloid F-19, the specific blocker of this channel, verapamil, was used and determined by varying the CaCl2 concentration (0.5-2.5 mM) in the Krebs-Henseleit solution.
The effect of alkaloid F-19 on sarcoplasmic reticulum (SR) Ca2+ transport systems (IP3R, RyR) was determined by measuring the contraction force induced by PE or caffeine in calcium-free Krebs solution. The maximum concentration of the alkaloid was incubated for 20 minutes and the contraction was studied by inducing PE or caffeine. The IP3R blocker heparin and the RyR blocker ruthenium red were also used. Cyclopiazonic acid (CPA), a blocker of this ATPase, was used to determine the role of SR Ca2+ ATPase (SERCA) in the vasorelaxant effect of alkaloid F-19. In the experiments, the vasorelaxant effect of F-19 on PE-induced contraction force was investigated under CPA incubation conditions.
Chemicals
Every chemical used was commercially available and of analytical grade. Phenylephrine (PE), verapamil, EGTA, Acetylcholine (Ach), L-NAME, indomethacin, heparin, ruthenium red, and CPA were acquired from Sigma Ltd. Alkaloid F-19 (Fig. 1) was synthesized by the staff of the Institute of Chemistry of Plant Compounds of Uzbek Academy of Sciences.
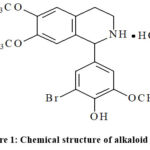 |
Figure 1: Chemical structure of alkaloid F-19. |
Statistical analysis
IC50 values, which indicate the drug concentration needed to achieve 50% of the maximum effect (EMax) for contraction or relaxation, were derived from concentration-response curves. These values were computed with the sigmoidal curve fitting feature within Origin 9.1 software (Microcal, Northampton, MA, U.S.A.). The outcomes of experiments, carried out repeatedly, are shown as M±m. Here, M is the average of the results and m represents the standard error. We used Student’s t-test to determine the statistical significance of the experimental outcomes compared to the control, with significance levels set at *p<0.05 and **p<0.01.
Results
Vasorelaxant effect of alkaloid F-19 on aortic contractions induced by KCl and PE
In preliminary experiments, the vasorelaxant effect of the isoquinoline alkaloid F-19 was studied on the contractile activity of the rat aortic vascular ring induced by a hyperpotassium solution. As a result, it was found that the alkaloid F-19 at a concentration of 5 μM attenuated the aortic contractile activity induced by 50 mM KCl by 8.6±3.8% compared to the control. It was also noted that at a concentration of 100 μM, this value was 93.5±3.7%. The IC50 value (concentration that reduces the contraction force by 50% compared to the maximum) of the alkaloid F-19 was 44.6 μM (Fig. 2, A). Similarly, in experiments, it was found that the alkaloid F-19 reduced the contractile activity of the aortic ring induced by 1 μM PE by 10.6±3.0% at a concentration of 5 μM compared to the control, and by 94.2±3.2% at a concentration of 100 μM, and the half-maximal concentration was equal to IC50=26.1 μM (Fig. 2, B).
 |
Figure 2: Dose-dependent vasorelaxant effect of the alkaloid F-19 on the contractile activity of endothelium-positive (E+) and endothelium-negative (E-) aortic rings induced by 50 mM KCl (A) and 1 μM PE (B). |
Based on the IC50 values obtained, it can be concluded that the alkaloid F-19 exhibits a stronger vasorelaxant effect on PE-induced contractions. It was found that the endothelial layer is very poorly involved in the vasorelaxant effect of the alkaloid F-19 (Fig. 2, A, B), and this was confirmed in experiments with L-NAME and indomethacin. From these results, it was observed that the vasorelaxant effect of the alkaloid F-19 is not endothelium-dependent and acts mainly through direct activation of the vasorelaxant mechanisms of vascular SMCs.
The role of voltage-gated L-type Ca2+ channels in the vasorelaxant effect of the alkaloid F-19
In this series of experiments, the effect of the alkaloid F-19 on the aortic ring in response to changes in the concentration of CaCl2 in the environment was studied. Cumulative addition of CaCl2 under conditions of incubation with KCl (50 mM) in Krebs’ solution, which lacks Ca2+ ions, led to a concentration-dependent increase in aortic ring contraction. Under these conditions, it was found that the alkaloid F-19 significantly reduced aortic contractility in a CaCl2 concentration-dependent manner (Fig. 3, A). In this case, preincubation with 100 μM of the alkaloid F-19 reduced the aortic contractile force during the maximal concentration of CaCl2 to 76.6±3.3% compared to the control. This suggests that it occurs through inhibition of voltage-gated L-type Ca2+ channels. To further clarify this, the effect of the L-type Ca2+ channel blocker verapamil on the concentration-dependent force of aortic contraction was compared with that of CaCl2. Verapamil (1 μM) reduced the CaCl2-dependent force of aortic ring contraction by 94.9±3.6% compared to control. Also, in subsequent experiments, the combined vasorelaxant effect of the specific the L-type Ca2+ channel blocker verapamil and the alkaloid F-19 was studied. In these experiments, it was noted that during incubation with verapamil (IC50=0.1 μM), the aortic ring reduced the force of contraction induced by KCl by 50±3.8% compared to the control, and the addition of the alkaloid F-19 (IC50=44.6 μM) under these conditions further attenuated the force of contraction by 28.5±3.3% (Fig. 3, B).
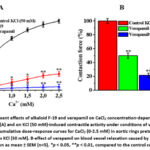 |
Figure 3: Vasorelaxant effects of alkaloid F-19 and verapamil on CaCl2 concentration-dependent contractions in rat aortic rings (A) and on KCl (50 mM)-induced contractile activity under conditions of verapamil (0.1 μM) incubation (B). |
The results of this study indicate that voltage-gated L-type Ca2+ channels are involved in the vasorelaxant effect of the alkaloid F-19. The additional decrease in aortic ring contractility under the influence of the alkaloid F-19 in the presence of verapamil indicates the involvement of other ion transport systems.
Effect of alkaloid F-19 on IP3R
In subsequent studies, PE (1 μM) in Ca2+-free Krebs solution supplemented with EGTA (1 mM) caused a transient contraction in aortic rings. This contraction occurs mainly as a result of the release of Ca2+ from the SR into the cytosol via the IP3R.20 This reduction was 66.7±4.2% of the contractile force of aortic rings induced by PE (1 μM) in normal Kreb’s saline (control). Preincubation of aortic rings with the alkaloid F-19 (100 μM) in Krebs’ solution lacking Ca2+ ions reduced the force of PE (1 μM)-induced contraction of the SMC from 66.7±4.2% to 24.3±3.6% (Fig. 4).
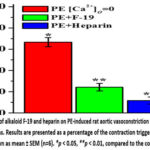 |
Figure 4: Effect of alkaloid F-19 and heparin on PE-induced rat aortic vasoconstriction in Krebs solution lacking Ca2+ ions. Results are presented as a percentage of the contraction triggered by PE (1μM). |
This result suggests that the vasorelaxant effect of the alkaloid F-19 may be related to IP3R blockade. To further clarify this, the effect of F-19 was compared with the IP3R inhibitor heparin (10 mg/ml). It was found that heparin (10 mg/ml) reduced the force of PE-induced contraction of aortic rings in Ca2+-free Krebs solution from 66.7±4.2% to 11.7±3.4% (Fig. 4). The results show that the vasorelaxant effects of F-19 and heparin are similar, indicating that this is achieved by inhibiting IP3R and reducing intracellular Ca2+ concentration.
Effect of alkaloid F-19 on SR RyR
It is known that in vascular smooth muscle cells, RyR, along with IP3R, is partially involved in the release of Ca2+ ions from the SR into the cytosol.21 In our experiments, we examined the effect of the F-19 alkaloid on RyR activity and caffeine-induced aortic contraction. In Krebs solutions without Ca2+ ions, the force of aortic ring contraction induced by caffeine (10 mM) was 41.4±3.1% compared to the force of aortic ring contraction induced by PE (1 μM) under normal conditions. In these experiments, it was found that preincubation with the alkaloid F-19 (100 μM) reduced the force of contraction of aortic rings induced by caffeine from 41.4±3.1% to 21.1±3.4% (Fig. 5).
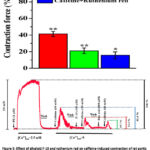 |
Figure 5: Effect of alkaloid F-19 and ruthenium red on caffeine-induced contraction of rat aortic blood vessel rings in Krebs solution lacking Ca2+ ions. |
According to these results, the vasorelaxant effect of the alkaloid F-19 on the force of contraction of the aortic rings induced by caffeine may be due to the attenuation of RyR activity. This was also proven when compared with the vasorelaxant effect of the RyR inhibitor ruthenium red. That is, preincubation of aortic rings with ruthenium red (100 μM) reduced the contraction force induced by caffeine from 41.4±3.1% to 15.7±4.2% (Fig. 5). The results obtained indicate that the vasorelaxant effect of the alkaloid F-19 on the contraction force induced by caffeine is associated with a decrease in the amount of Ca2+ released from the SR through the RyR.
Effect of alkaloid F-19 on SERCA
To assess the involvement of SERCA in the vasorelaxant effect of the alkaloid F-19 on the contractile activity of aortic smooth muscle cells, its specific blocker cyclopiazonic acid (CPA) was used. In the presence of CPA (10 μM), it was found that the maximum vasorelaxant effect of alkaloid F-19 was reduced compared to the control. In experiments, it was observed that the vasorelaxant effect of the alkaloid F-19 on the PE-induced contraction force decreased from 94.2±3.2% to 82.8±4.2% under conditions of incubation with CPA (10 μM) (Fig. 6, A). The results revealed that SERCA is involved in the vasorelaxant effect of the tested alkaloid. To further confirm this, in subsequent experiments, the addition of CPA (10 μM) in the presence of verapamil (1 μM) resulted in a transient contraction of the aortic annulus, which was 36.7±3.9% compared to the aortic contraction induced by 1 μM PE. Inhibition of SERCA by CPA, which reduces the entry of Ca2+ ions into the SR, increases the concentration of [Ca2+]in the cell and leads to a decrease in the SMC. In these experiments, it was found that preincubation of the aortic ring with the alkaloid F-19 reduced the force of aortic contraction induced by CPA by 23.6±3.6% (Fig. 6, B).
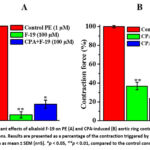 |
Figure 6: Vasorelaxant effects of alkaloid F-19 on PE (A) and CPA-induced (B) aortic ring contractions under CPA incubation conditions. Results are presented as a percentage of the contraction triggered by PE (1μM). |
These results indicate the involvement of SERCA in the vasorelaxant effect of the alkaloid F-19.
Discussion
This study presents the mechanisms of vasorelaxant action of the alkaloid F-19 in rat aortic rings. We have experimentally demonstrated that the alkaloid F-19 exerts a potent vasorelaxant effect on aortic rings induced by KCl and PE. It was observed that the vasorelaxant effect of the F-19 alkaloid was not endothelium-dependent in aortic blood vessels, that is, it remained unchanged in the conditions of mechanical removal of the endothelial layer or in the conditions of incubation with L-NAME and indomethacin, in KCl or PE-induced contractions.
Сa2+ ions play a key role in the contraction and relaxation of smooth muscles that support vascular function, as well as in the control of vascular tone.22 The intracellular [Са2+]in concentration is maintained by various Ca2+ transport systems located in the cell plasmalemma and the SR.23,24 In SМСs voltage-gated L-type Ca2+ channels play a key role in ensuring the entry of Ca2+ ions into the cell and activate the influx of Ca2+ in response to membrane depolarization, which in turn causes muscle contraction.25,26 In our experiments, the alkaloid F-19 significantly reduced the contractile activity of aortic rings induced by KCl. This suggests that the vasorelaxant effect of F-19 may be due to a decrease in the entry of Ca2+ ions into the cell by blocking L-type Ca2+ channels. This assumption was confirmed by the fact that the alkaloid F-19 reduced the force of contraction of aortic rings induced by changing the CaCl2 concentration in the [Ca2+]out medium. These results showed that the vasorelaxant effect of F-19 is mediated by the blockade of L-type Ca2+ channels. However, the additional vasorelaxant effect of F-19 in conditions where L-type Ca2+ channels are blocked by verapamil indicates the involvement of other ion transport systems.
SR Ca2+ transport systems, such as IP3R, RyR, and SERCA play an important role in controlling the contraction and relaxation processes of smooth muscles.27-29 IP3R plays a leading role in the release of Ca2+ from the SR into the cytosol in vascular smooth muscle cells.30 Therefore, in our experiments, we examined the effect of F-19 on PE-induced aortic ring contractions in the absence of Ca2+ ions. These studies showed that the PE-induced contraction was significantly reduced by the alkaloid F-19, indicating that it inhibits the release of Ca2+ ions from the SR through the IP3R. When we compared this to the vasorelaxant effect of the IP3R inhibitor heparin, we found that they had similar effects. It is known that heparin inhibits the release of Ca2+ from the SR via IP3R in reducing systems.31-33 In experiments, heparin was also observed to reduce PE-induced contractions of aortic rings in Krebs solution, which lacks Ca2+ ions. Therefore, based on these results, the vasorelaxant effect of the alkaloid F-19 is dependent on IP3R, which inhibits the release of Ca2+ ions from the SR and leads to relaxation of the aortic vascular smooth muscle.
In the absence of Ca2+ ions, [Са2+]out=0, the participation of RyR along with IP3R in the contraction of the SMR has been noted.34,35 The experiments studied the effect of the alkaloid F-19 on the force of aortic contraction induced by caffeine in Krebs solutions lacking Ca2+ ions. It is known that under the influence of caffeine, the release of Ca2+ ions from the SR into the cytosol occurs due to the activation of RyR in the SМС.36,37 In experiments, it was found that incubation with the alkaloid F-19 reduced the contraction force induced by caffeine. This is explained by the reduction of Ca2+ release from the SR through blockade of RyR. These results indicate that RyR is partially involved in the vasorelaxant effect of the alkaloid F-19.38 This was confirmed by comparing the vasorelaxant effect of the RyR inhibitor ruthenium red39 on caffeine-induced aortic ring contraction. It was found that the effects of the alkaloid F-19 and ruthenium red on reducing the force of contraction of the aortic rings induced by caffeine are similar.
SERCA plays a key role in the regulation of Ca2+ ions in the SR of the SМС.40,41 SERCA plays an important role in ensuring the accumulation of Ca2+ ions in the SR, maintaining Ca2+ homeostasis in the SМС, and in the process of smooth muscle relaxation.42,43 It is known that in studies to examine the effect of biologically active compounds on SERCA in smooth muscle cells, the selective blocker of this pump CPA is used.44 In our experiments, it was observed that the vasorelaxant administration of the alkaloid F-19 reduced PE-induced aortic contraction under CPA incubation conditions. The reduced vasorelaxant effect of F-19 may be due to the inhibition of SERCA by CPA. To further clarify this, in subsequent experiments, the effect of the F-19 alkaloid on transient contractions induced by incubation of aortic rings with CPA for a period of time was examined. It is known that CPA blocks SERCA and inhibits the entry of Ca2+ ions into the SR, causing a transient decrease in [Ca2+]in in the SМС.45 It was found that preincubation of aortic rings with alkaloid F-19 reduced the contraction induced by CPA. According to these results, the vasorelaxant effect of the F-19 alkaloid is explained by its reduction in [Ca2+]in in the CSF through SERCA activation.
Conclusions
These studies provided the first evidence that the vasorelaxant effect of the isoquinoline alkaloid F-19 on rat aortic rings is mediated by several complex mechanisms. This inhibits the release of Ca2+ ions into the cytosol through voltage-gated L-type Ca2+ channels and the SR Ca2+ transport systems and is provided by the activation of SERCA. The information obtained on the mechanism of action of the alkaloid F-19 plays an important role in the regulation of vascular tone. Modulating the function of SR Ca2+ transport systems could help develop new effective approaches to treating many cardiovascular diseases.
Acknowledgment
This work was supported by the Science and Technology Development Coordination Committee.
Funding source
This work was financed by the grant F-OT-2021-154 of the Science and Technology Development Coordination Committee under the Cabinet of Ministers of the Republic of Uzbekistan.
Conflict of interest
The author(s) do not have any conflict of interest
Data Availability
This statement does not apply to this article.
Ethics Statement
The experimental protocols complied with the standards and requirements for the humane treatment of animals and the provisions of the Ethical Commission of the IBB at the National University of Uzbekistan. (Protocol No. 7 BEC/IBB-NUU of 04/07/2022) on the use of laboratory animals. Preparations of isolated aortic segments were obtained using a known method.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources – Not Applicable
Authors’ Contribution
- Abdisalim A. Zaripov: Conceptualization, Methodology, Writing – review & editing, Writing – Original Draft, Supervision.
- Inoyat Z. Zhumaev: Methodology, Writing – review & editing, Writing – original draft, Validation, Formal analysis.
- Pulat B. Usmanov: Conceptualization, Methodology, Writing – review & editing, Supervision, Resources, Funding acquisition, Project Administration.
- Yulduzkhon T. Mirzaeva: Formal analysis.
- Shavkat Yu. Rustamov: Visualization, Resources.
- Sadriddin N. Boboev: Visualization, Resources.
- Adilbay T. Esimbetov: Data Collection, Analysis, Writing – Review & Editing.
- Gulnaz S. Begdullaeva: Data Collection, Analysis, Writing – Review & Editing.
- Sardor B. Sobirov: Resources, Data curation.
- Eldor B. Ibragimov: Resources, Data curation.
- Sherzod N. Zhurakulov: Isolation of alkaloid F-19.
- Dilbar D. Safarova: Investigation, Formal analysis.
- Ra’no I. Yusupova: Investigation, Formal analysis.
References
- Braunwald E. The war against heart failure: the Lancet lecture. The Lancet. 2015;38:1838-1845.
CrossRef - Zhumaev I.Z., Boboev S.N., Usmanov P.B., et al. Mechanism of Positive Inotropic Effect of Vincanine on Cardiac Muscle Contraction Activity. Biomedical & Pharmacology. 2022;15(4):1-8.
CrossRef - Ziyavitdinov Zh. F., Ishimov U. Zh., Berdiev N. Sh., et al. Supramolecular complex of lappaconitine hydrobromide and the monoammonium salt of glycyrrhizic acid: synthesis, physicochemical characteristics, and antiarrhythmic activity. Pharmaceutical Chemistry Journal. 2022;56(2):167-173.
CrossRef - Wright P., Thomas M. Pathophysiology and management of heart failure. Clinical Pharmacist. 2018;10;261-269.
- Dassanayaka S., Jones, S.P. Recent developments in heart failure. Circulation Research. 2015;117:58-63.
CrossRef - Horovitz A., Menice C., Laporte R., Morgan K. Mechanism of smooth muscle contraction. Pharmacological Rev. 1996;70(4):967-992.
CrossRef - Somlio A.V., Bond M., Somlio A.P., Scarpa A. Inositol trisphosphate-induced calcium release and contraction in vascular smooth muscle. Nati. Acad. Sci. USA. 1985;82: 5231-5235.
CrossRef - Francis S., Busch J., Corbin J. cGMP–dependent protein kinases and cGMP–phosphodiesterases in nitric oxide and cGMP action. Rev. 2010;62(3):525-563.
CrossRef - Zaripov A.A., Jumayev I.Z., Usmanov P.B., et al. Protective Effect of DHQ-11 against Hypoxia-induced Vasorelaxation. Trends in Sciences; 2024;21(11):1-12.
CrossRef - Khushmatov Sh.S., Zhumaev I.Z., Zhurakulov Sh.N., Saidov A.Sh., Vinogradova V.I. Synthesis and Comparative Inotropic Effects of Several Isoquinoline Alkaloids. Pharmaceutical Chemistry Journal. 2020;54(1):7-11.
CrossRef - Sanaa Almowallad, Rehab Al-Massabi. Berberine modulates cardiovascular diseases as a multitarget-mediated alkaloid with insights into its downstream signals using in silico prospective screening approaches. Saudi Journal of Biological Sciences. 2024;31(5):103977.
CrossRef - Jumayev I., Usmanov P., Rustamov Sh., Zhurakulov Sh. Comparative inotropic effects of the some isoquinoline alkaloids. Biomedical and Pharmacology Journal. 2020;13(1):325- 333.
CrossRef - Jinhua Li, Yarong Wu, Shuze Dong, et al. Research Progress on Neuroprotective Effects of Isoquinoline Alkaloids. 2023;8(12):4797.
CrossRef - Rakhmanova K.A., Zhurakulov S.N., Tursunkhodjayeva. F.M., Azamatov A.A., Saidkhodjayeva D.M., Jumayev I. Z. Analgesic and Anti-Inflammatory Effects of 1-(4′-Dimethylaminophenyl)-6, 7-Dimethoxy-1,2,3,4-Tetrahydroisoquinoline Hydrochloride. Biomedical and Pharmacology Journal.2022;15(2):891-895.
CrossRef - Guli M.R., Nozim N.Kh., Kabil E.N., et al. Anti-thrombotic action of sulfated polysaccharides on thrombosis caused by thromboplastin. Research Journal of Pharmacy and Technology. 2021; 14(11):6085-8.
CrossRef - Silva D. F., Porto D.L., Araújo I.A., et al. Endothelium-derived nitric oxide is involved in the hypotensive and vasorelaxant effects induced by discretamine in rats. 2009;64(5):327-331.
- Oshchepkova Yu.I., Uzbekov V.V., Jumayev I.Z., et al. Comparative study of antiarrhythmic and inotropic activity of amiodarone hydrochloride and its complexes with glycyrrhizic acid and monoammonium salt of glycyrrhizic acid. Eksperimental’naya i Klinicheskaya Farmakologiya. 2023; 86( 4):15 – 22.
- Mirzayeva Y.T., Zaripov A.A., Zhumaev I.Z., et al. The Protective Effect of Indole Alkaloid Vincanine Against Hypoxia-Induced Vasorelaxation Model of Rat Aorta. Biomedical & Pharmacology Journal. 2024;17(1):483-491.
CrossRef - Macêdo C.L., Vasconcelos L.C., Correia A.C., et al. Mechanisms Underlying Vasorelaxation Induced in Rat Aorta by Galetin 3,6-Dimethyl Ether, a Flavonoid from Piptadenia stipulacea (Benth.) Ducke. 2014;19:19678-19695.
CrossRef - Usmanov P.B., Jumayev I.Z., Rustamov Sh.Yu., et al. The Combined Inotropic and Vasorelaxant Effect of DHQ-11, a Conjugate of Flavonoid Dihydroquercetin with Isoquinoline Alkaloid 1-aryl-6,7-dimethoxy- 1,2.3,4-tetrahydroisoquinoline. Biomedical & Pharmacology Journal. 2021;14(2):651-661.
CrossRef - Omonturdiev S.Z., Abdullaev I.Z., Inomjonov D.R., et al. The Effect of Ájuga Turkestánica on the Rat Aortic Smooth Muscle Ion Channels. Biomedical and Pharmacology Journal.2024;17(2):1213-1222.
CrossRef - Tykocki N.R., Boerman E.M., Jackson W.F. Smooth Muscle Ion Channels and Regulation of Vascular Tone in Resistance Arteries and Arterioles. Compr Physiol. 2017;7(2):485-581.
CrossRef - Karaki H., Ozaki H., Hori M., Mitsui-Saito M., Amano K., Harada K. Calcium movements, distribution, and functions in smooth muscle. Pharmacol Rev. 1997;49:157-230.
CrossRef - Jorge Reyes-García, Edgar Flores-Soto, Abril Carbajal-García, Bettina Sommer, Luis M Montaño. Maintenance of intracellular Ca2+ basal concentration in airway smooth muscle (Review). Int J Mol Med. 2018;42(6):2998-3008.
CrossRef - Cribbs L.L. Vascular smooth muscle calcium channels. Circulation Research. 2001;89: 560-562.
CrossRef - Nelson, M.T., Quayle, J.M. Physiological roles and properties of potassium channels in arterial smooth muscle. J. Physiol. 1995;268(37):799-822.
CrossRef - Chalmers S., Olson M.L., MacMillan D., Rainbow R.D., McCarron J.G. Ion channels in smooth muscle: Regulation by the sarcoplasmicreticulum and mitochondria. Cell Calcium. 2007;42(4):447-466.
CrossRef - Wray S., Burdyga T. Sarcoplasmic reticulum function in smooth muscle. Rev. 2010;90(1):P.113-178.
CrossRef - Jumayev I.Z., Boboev S.N., Usmanov P.B., et al. Role of RyR2 and SERCA2a in the Cardioprotective Effects of Vincanine and Pyrazoline Alkaloids. Trends in Sciences. 2025;22(1):1-11.
CrossRef - Brozovich F.V., Nicholson C.J., Degen C.V., Gao Y.Z, Aggarwal M, Morgan K.G. Mechanisms of Vascular Smooth Muscle Contraction and the Basis for Pharmacologic Treatment of Smooth Muscle Disorders. Pharmacological Reviews. 2016;68(2):476-532.
CrossRef - Kobayashi, S., Somlyo A.V., Somlyo A.P. Heparin inhibits the inositol 1,4,5-trisphosphate-dependent, but not the independent, calcium release induced by guanine nucleotide in vascular smooth muscle. Biochemical and Biophysical Research Communications. 1988;153;625-631.
CrossRef - Narayanan D., Adebiyi A., Jaggar J.H. Inositol trisphosphate receptors in smooth muscle cells. American Journal of Physiology-Heart and Circulatory Physiology. 2012;302;2190-2210.
CrossRef - Yamamoto H., Kanaide H., Nakamura M. Heparin specifically inhibits the inositol 1,4,5-trisphosphate-induced Ca2+ release from skinned rat aortic smooth muscle cells in primary culture. Naunyn-Schmiedeberg’s Arch Pharmacol. 1990;341:273-278.
CrossRef - Tykocki N.R., Thompson J.M., Jackson W.F., Watts S.W. Ryanodine receptors are uncoupled from contraction in rat vena cava. Cell Calcium. 2013;53(2):112-119.
CrossRef - Thomas N.L., Williams A.J. Pharmacology of ryanodine receptors and Ca2+-induced Ca2+ WIREs Membr Transp Signal. 2012;1:383-397.
CrossRef - Watanabe С., Yamamoto I., Hirano K., Kobayashi S., Kanaide H. Mechanisms of caffeine-induced contraction and relaxation of rat aortic smooth muscle. J Physiol. 1992;456;193-213.
CrossRef - Huihui Kong, Peter P Jones, Andrea Koop, Lin Zhang, Henry J Duff, Wayne Chen. Caffeine Induces Ca2+Release by Reducing the Threshold for Luminal Ca2+ Activation of the Ryanodine Receptor. Biochem J. 2008;414(3):441-452.
CrossRef - Kanmura Y., Raeymaekers L., Casteels R. Effects of doxorubicin and ruthenium red on intracellular Ca2+ stores in skinned rabbit mesenteric smooth-muscle fibers. Pflügers Arch – Eur J Physiol. 1998;435:645-653.
- Zhang Z.D., Kwan C.Y., Daniel E.E. Subcellular membrane characterization of ryanodine-binding sites in smooth muscle. Biochem J. 1993;290:259-266.
CrossRef - Matthew A, Shmygol A, Wray S. Ca2+ entry, efflux and release in smooth muscle. Biological Research. 2004;37(4):617-624.
CrossRef - Hirota, P. Helli, L.J. Janssen. Ionic mechanisms and Ca2+ handling in airway smooth muscle. European Respiratory Journal. 2007;30(1):114-133.
CrossRef - Floyd R., Wray S. Calcium transporters and signalling in smooth muscles. Cell Calcium. 2007;42(45):467-476.
CrossRef - Floyd, Wray S. Calcium transporters and signalling in smooth muscles. Cell Calcium.2007;42(4):467-76.
CrossRef - Uyama Y., Imaizumi Y., Watanabe M. Effects of cyclopiazonic acid, a novel Ca2+-ATPase inhibitor, on contractile responses in skinned ileal smooth muscle. J. Pharmacol. 1992;106:208-214.
CrossRef - Nobel D., Borisova L., Wray S., Burdyga T. Store-operated Ca2+ entry and depolarization explain the anomalous behaviour of myometrial SR: Effects of SERCA inhibition on electrical activity, Ca2+ and force. Cell Calcium. 2014;56(3):188-194.
CrossRef







