Manuscript accepted on :16-05-2025
Published online on: 29-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Exbrayat Jean-Marie
Second Review by: Dr. Pravinkumar Darji
Final Approval by: Dr. Prabhishek Singh
Wenyan Xu1 ,Bo Zou1
,Bo Zou1 ,Guohao Xie2
,Guohao Xie2 ,Yeqing Wu2
,Yeqing Wu2 and H. Fai Poon 2*
and H. Fai Poon 2*
1School of Medicine, Sun Yat-Sen University, Shenzhen, China.
2Research and Development Department,Quacell Biotechnology Co., Ltd., Zhongshan, China.
Corresponding Author E-mail: hungfaipoon@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3198
Abstract
Cell culture plays a pivotal role in the biomedical industry, with a growing shift from traditional serum-containing adherent systems to serum-free suspension culture platforms. This transition underscores the importance of developing robust cell suspension adaptation protocols and serum-free culture media to meet industrial demands. In this study, we employed a combination of cell suspension acclimation, differential screening of culture media, and factorial design methodology to rapidly adapt adherent human embryonic kidney (HEK293) cells to suspension culture. Additionally, we developed a chemically defined medium optimized for these cells within a short timeframe. By systematically adjusting the concentrations of key medium components and integrating an optimized adaptation protocol, we successfully achieved efficient suspension culture of HEK293 cells. Notably, this approach significantly reduced cell aggregation and enhanced cell growth density, thereby advancing the potential for industrial-scale applications.
Keywords
Cell adaptation; HEK293 cells; Medium optimization; Plackett - Burman experiment; Serum-free suspension culture
Download this article as:| Copy the following to cite this article: Xu W, Zou B, Xie G, Wu Y, Poon H. F. Development of Serum-Free Medium and Suspension Culture for HEK293 Cells Using Systematic Optimization Strategies. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Xu W, Zou B, Xie G, Wu Y, Poon H. F. Development of Serum-Free Medium and Suspension Culture for HEK293 Cells Using Systematic Optimization Strategies. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/43gTP1s |
Introduction
Cell culture is a cornerstone of the biomedical industry, playing a critical role in drug discovery, vaccine production, and the development of biopharmaceuticals. Among the various cell types used, mammalian cells are particularly valuable due to their ability to express recombinant proteins and produce virus particles, making them indispensable for therapeutic applications. In recent years, the industry has witnessed a significant transition from traditional serum-containing adherent cell culture systems to serum-free suspension culture platforms. This shift is primarily driven by the need for enhanced reproducibility, reduced risk of contamination, and improved scalability, all of which are essential for large-scale industrial production.1
The development of serum-free culture media and the adaptation of cells to suspension culture are pivotal for advancing pharmaceutical manufacturing. Serum-free media offer greater control over medium composition and quality, while suspension culture technology enables scalable and cost-effective production processes. However, designing serum-free media that provide adequate nutrients and support optimal cell growth remains a challenge, particularly for cell lines with unique metabolic and physiological requirements. For instance, while significant progress has been made in optimizing serum-free media for Chinese hamster ovary (CHO) cells, 2HEK293 cells—a widely used cell line for recombinant protein production—require tailored optimization due to their distinct characteristics. This study addresses this gap by focusing on the development of a serum-free medium and an efficient adaptation protocol specifically for HEK293 cells.
In this study, we utilized a Design of Experiments (DOE) approach to develop a serum-free culture medium tailored for adherent HEK293 cells. The DOE methodology enables systematic analysis of the effects of multiple factors and their interactions, providing a robust framework for optimizing culture conditions. By strategically adjusting the concentrations of key medium components and implementing a structured adaptation protocol, we aimed to achieve successful suspension culture of HEK293 cells. Our specific objectives included reducing cell aggregation rates, enhancing cell growth density, and establishing a scalable platform to support industrial applications .3
Materials and Method
Cell lines and cell culture
Cell Line Source
The HEK293 cell line was obtained from the European Collection of Authenticated Cell Cultures (ECACC; Catalog Number: 85120602) on November 8, 2020. Following acquisition, the cells were expanded and cryopreserved in liquid nitrogen for long-term storage.
Culture Environment
For initial adherent culture, HEK293 cells were maintained in a carbon dioxide incubator (SANYO MCO-15AC) at 37 °C with a 5% CO₂ atmosphere. The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Gibco, 11995065) supplemented with 10% fetal bovine serum (FBS; Quacell, B21004) and seeded in T25 culture flasks (Corning, CLS430639). For suspension culture, cells were first digested with 0.25% trypsin (Gibco, 25200056) in T25 culture flasks. After digestion, they were transferred using a pipette in a biosafety cabinet conditions into Corning shake flasks (CLS430421). The cultures were then incubated in a shaking incubator (ZhiChu BS8V) at 37°C, 5% CO₂, with constant agitation at 140 rpm to maintain uniform suspension.The microscope used for cell photography was the Mshot MF53-N.
Cell suspension
Adherent cells were cultured in DMEM medium supplemented with 10% FBS. Subsequently, the FBS content was gradually decreased in a gradient of 10%, 5%, 2.5%,1% and 0.5%.4 At any stage when some cells started to grow in suspension,all cells in the T25 flasks were digested and then transferred via pipette into 125 mL shake flasks for suspension culture.
Chemical define Media screening
Following the establishment of suspension culture in T25 flasks, 20 serum-free media formulations were selected from the Quacell media panel for further evaluation. Cells were seeded into each of these media at an initial density of 1 × 10⁶ cells/mL. After 72 hours of culture, the cells were subcultured to maintain the same initial density. Cell density, viability, and aggregation rate were monitored at 24-hour intervals using a cell counter (Alit, Countstar) to assess growth performance. Cultures demonstrating poor proliferation or viability below 60% were terminated. Media formulations that supported robust cell growth and maintained high viability were identified and selected for subsequent optimization and cell adaptation studies.
Factor Selection and Experimental Design
Previous studies have demonstrated that the concentrations of ions such as iron, calcium, magnesium, and zinc significantly influence cell growth and metabolic activity.5,6 Based on these findings, this study selected iron, magnesium, calcium, and zinc as key factors for investigation. Additionally, ethylenediaminetetraacetic acid (EDTA) was included as a candidate component due to its ability to chelate metal ions and regulate the concentration of free metal ions in the culture medium.7 Linear regression analysis of 48-hour cell density versus medium components further demonstrated that specific amino acids and vitamins could significantly enhance cellular growth performance.8-10 Consequently, these amino acids and vitamins were also incorporated into the study.
A total of 11 factors were selected, each evaluated at two levels. The experimental design employed the Plackett-Burman (PB) methodology, a robust experimental design method for screening key factors. This approach tests each factor at two levels (high/low) and can only identify main effects without analyzing interactions, making it particularly useful for medium optimization. The factor levels were determined based on the optimal medium composition identified during the screening phase, with adjustments made to reflect the effective concentration ranges reported in the literature for cell culture applications .11-14
Serum-Free Medium Composition and Preparation
During the medium screening phase, a panel of commercially available customized media was selected for evaluation. For the media used in the Plackett-Burman (PB) experiment, an incomplete base medium was first prepared by omitting the 11 selected factors. This base medium was then divided into 13 aliquots. Each aliquot was supplemented with the corresponding factors at the concentrations specified for each experimental group. Factors with quantities greater than 10 mg were weighed using an analytical balance, while those with quantities less than 10 mg were added as 100X concentrated stock solutions.
Following the addition of factors, the pH of each medium was adjusted to 7.00 ± 0.10 using diluted hydrochloric acid (HCl) and sodium hydroxide (NaOH) solutions. The osmolality was also adjusted to 300 mOsm/kg to ensure physiological compatibility. The media were then sterilized by filtration through a 0.22 μm membrane filter and stored in the dark at 2–8 °C until further use.
Cell Growth and Aggregation Rate Determination
Cell growth was evaluated using trypan blue exclusion staining and an automated cell counter (Countstar IC1000). Briefly, a 20 μL aliquot of cell suspension was mixed with an equal volume of 0.2% trypan blue solution. The mixture was then loaded into the cell counter to determine total cell count and viability. Cell viability was calculated as the percentage of viable (unstained) cells relative to the total cell population. Additionally, the cell counter provided measurements of other parameters, including cell roundness and aggregation rate, which were used to further assess cell culture quality.
Results
Results of Serum-Containing Suspension Adaptation
Cells were cultured in T25 cell culture flasks. When cultured in DMEM supplemented with 10% FBS, the cells were in an adherent state. In the DMEM medium with 5% FBS, the doubling time of the cell are similar to that of the control group(25.73h to 26.54 h), without any obvious changes. After being passaged into the medium containing 2.5% FBS, the doubling time of the cells slowed down from 26.54 hr to 59.31 hr , and the cell viability decreased(Table 1). However, some cells began to grow in suspension. After three passages in the T25 flask, the cells were transferred to shake flasks for further suspension culture in DMEM medium with 2.5% FBS. Photographs of the cells at different stages during adherent culture are shown in Figure 1.
The cell growth conditions were evaluated in T25 flasks using varying serum concentrations. For each passage, the seeding density was maintained at 8 × 10⁵ cells/mL. “P” denotes the passage number under each serum concentration. Viable cell density (VCD) and viability were measured 48 hours post-passage. The doubling time was calculated for each passage, and the “mean” is the average value of the cell doubling times of the three passages at each stage.
Table 1: Cell Growth Conditions in T25 Flasks Under Different Serum Concentrations.
| FBS% | P | VCD(cells/ml) | Viability(%) | Doubling time(h) | Mean |
| 1 | 2.56E+06 | 89.7 | 28.60 | ||
| 10%FBS | 2 | 3.25E+06 | 94.2 | 23.73 | 25.73 |
| 3 | 3.05E+06 | 99.1 | 24.86 | ||
| 1 | 3.06E+06 | 93.3 | 24.80 | ||
| 5%FBS | 2 | 2.29E+06 | 92.3 | 31.63 | 26.54 |
| 3 | 3.36E+06 | 97.7 | 23.18 | ||
| 1 | 1.83E+06 | 85.3 | 40.20 | ||
| 2.5%FBS | 2 | 1.14E+06 | 72.6 | 93.93 | 59.31 |
| 3 | 1.71E+06 | 82 | 43.79 |
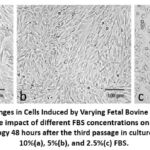 |
Figure 1: Morphological Changes in Cells Induced by Varying Fetal Bovine Serum (FBS) Concentrations.This figure demonstrates the impact of different FBS concentrations on cell morphology. |
The suspended cells in DMEM medium containing 2.5% FBS were counted, centrifuged(200g,5min), and then resuspended in the 20 media selected from the medium panel (see Figure [2]) and the DMEM medium with 2.5% FBS as Control respectively. The seeding cell density was controlled at 1.00E+06 cells/ml. Samples were taken for counting at 24 h and 48 h after seeding (Figure 3a). Based on cell growth and viability data collected from two independent counts, the results were ranked from high to low, and 11 culture media were selected for second passage culture. Among these, Panel 6 ranked fourth in terms of growth performance but was excluded from further experimentation due to contamination during the handling process. The cell growth conditions during the second passage are presented in Figure 3b. Based on the highest viable cell density(3.44E+06 cells/ml), cell viability(96.4%), and the lowest cell aggregation rate(30%) of Panel 10 at 48 hours after the second passage, this group of medium was selected for further optimization.
 |
Figure 2: Variation in Component Concentrations Among Medium Bank.This heatmap visually illustrates the differential composition of the twenty culture media within the medium bank. |
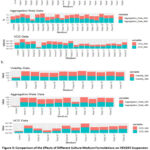 |
Figure 3: Comparison of the Effects of Different Culture Medium Formulations on HEK293 Suspension Culture. |
DOE Results and Analysis
Panel 10 medium was further selected for optimization of its components. The components were divided into 11 groups for the Plackett-Burman (PB) experiment, as detailed in Table 2. In the experimental design, “1” indicates that the concentration of components in a specific group was increased to twice the concentration in the original Panel 10 medium, while “-1” indicates that the concentration was reduced to half of the original level. Through these adjustments, 13 modified versions of Panel 10 medium were generated (Figure 3).
Cell growth and viability were measured for each modified medium, and the results are presented in Figure 4. Significant differences in cell growth rate and viability were observed among the different versions of Panel 10 medium, demonstrating the impact of component concentration adjustments on cell culture performance. The Pareto chart (Figure 5) highlights the components that exhibit a statistically significant impact on viable cell density (VCD). Specifically, an increase in the concentrations of MgSO₄ and EDTA negatively affects cell growth, whereas an increase in the concentration of Ammonium iron (III) citrate significantly enhances cell growth.
A comparison between predicted and actual values (Figure 6) demonstrates close agreement, with a coefficient of determination (R²) of 0.9111. This high correlation confirms the model’s strong predictive accuracy in forecasting cellular proliferation dynamics under the tested experimental conditions.
For Medium No. 13, the cell density reached 3.90×10⁶ cells per milliliter 48 hours after the second passage, and it further increased to 6.99×10⁶ cells per milliliter at 72 hours. It is worth noting that the cell viability remained above 90% during this period, which demonstrates a robust cell growth trend.
Finally, a response surface methodology (RSM) was employed to investigate the optimal concentration ranges of key factors identified in the Plackett-Burman (PB) experiment. However, no statistically significant improvement in cell growth or viability was observed, suggesting that the factor levels selected in the PB design were already near-optimal, and further refinement was unnecessary.
Cell morphology was analyzed during the adaptation process using an inverted microscope (Figure 7). The cells exhibited a smooth, spherical shape with uniform dispersion, confirming their stable adaptation to suspension culture conditions.A batch culture experiment was conducted in Medium No. 13 with an initial seeding density of 1.00 × 10⁶ cells/mL. Cells were cultured in a shaking incubator at 36.5°C with an agitation speed of 140 rpm. Daily sampling revealed a peak cell density of 1.12 × 10⁷ cells/mL on day 7 (Figure 8). Cell viability remained above 96% during the exponential growth phase, demonstrating robust proliferation. These results confirm the successful completion of the cell adaptation process to serum-free suspension culture.
Table 2: Plackett-Burman Experimental Design Matrix for Screening Factors Influencing Response Variables.The matrix encompasses 13 experimental runs (Run 1 to Run 13). Factor levels are indicated by -1 and 1, representing low and high levels.(AIC :Ammonium iron (III) citrate;THCl:Thiamin Hydrochloride;DS:Dextran Sulfate.)
| Run | A | B | C | D | E | F | G | H | J | K | L |
| ZnSO₄·7H₂O | CaCl₂ | MgSO₄ | AIC | VB₁₂ | THCl | Met | Thr | Val | DS | EDTA | |
| 1 | -1 | -1 | -1 | -1 | -1 | -1 | -1 | -1 | -1 | -1 | -1 |
| 2 | 1 | 1 | -1 | -1 | -1 | 1 | -1 | -1 | 1 | -1 | 1 |
| 3 | -1 | 1 | 1 | 1 | -1 | -1 | -1 | 1 | -1 | -1 | 1 |
| 4 | 1 | 1 | 1 | -1 | -1 | -1 | 1 | -1 | -1 | 1 | -1 |
| 5 | 1 | -1 | 1 | 1 | 1 | -1 | -1 | -1 | 1 | -1 | -1 |
| 6 | -1 | 1 | -1 | -1 | 1 | -1 | 1 | 1 | 1 | -1 | -1 |
| 7 | -1 | -1 | 1 | -1 | 1 | 1 | 1 | -1 | -1 | -1 | 1 |
| 8 | -1 | -1 | -1 | 1 | -1 | -1 | 1 | -1 | 1 | 1 | 1 |
| 9 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 10 | -1 | -1 | 1 | -1 | -1 | 1 | -1 | 1 | 1 | 1 | -1 |
| 11 | -1 | 1 | -1 | 1 | 1 | 1 | -1 | -1 | -1 | 1 | -1 |
| 12 | 1 | -1 | -1 | -1 | 1 | -1 | -1 | 1 | -1 | 1 | 1 |
| 13 | 1 | -1 | -1 | 1 | -1 | 1 | 1 | 1 | -1 | -1 | -1 |
 |
Figure 4: HEK293 Suspension Cell Growth and viability Curves Over Three Passages.This graph illustrates the cell growth curves for 13 different groups over a period of time, with passages performed every 72 hours for a total of three passages. |
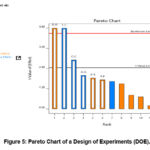 |
Figure 5: Pareto Chart of a Design of Experiments (DOE). |
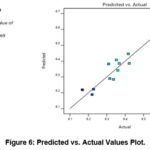 |
Figure 6: Predicted vs. Actual Values Plot. |
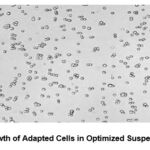 |
Figure 7: Growth of Adapted Cells in Optimized Suspension Medium |
This figure illustrates the morphology of HEK293 cells successfully adapted to suspension culture in an optimized medium. The cells demonstrate excellent dispersion and high viability, reflecting their effective adaptation to suspension culture conditions.
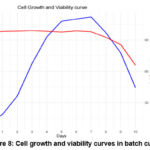 |
Figure 8: Cell growth and viability curves in batch culture. |
Discussion
The adaption of adherent cell culture to serum-free suspension culture is an emerging trend in the biomedical industry, and our study, focusing on HEK293 cell suspension culture and serum-free medium development, has unearthed several key insights.
During the serum-containing suspension adaptation process, the progressive reduction of fetal bovine serum (FBS) concentration exerted a pronounced influence on cell morphology, cell culture, double time etc (Figure 1,Table 1). When the FBS concentration decreased from 10% to 5%, the impact on cell culture was relatively minor. However, upon reaching 2.5% FBS, the cell growth rate slackened, and cell viability declined, while suspended cells began to emerge. This concentration appears to be a crucial turning point for HEK293 cell line. We speculate that this performance is due to insufficiency of nutrients provided by the serum that lead to cell growth is hampered. Moreover, it is pursable that since the factors in the serum that promote cell adhesion are also diminished, cells start to show a tendency to transition into a suspension state. It is thus essential to bridge the gap in suspension culture techniques and supplement the necessary nutrients at this critical stage, which is why we opted to screen for serum-free media at this juncture.
By screening 20 types of serum-free media and comparing the cell growth density, viability, and aggregation rate across them, we identified panel 10 as the prime candidate for further optimization. The Plackett – Burman experiment selected 11 components groups, enabled us to analyze the critical factors influencing cell growth in the culture medium. These factors presumably interact in complex ways,but the Plackett – Burman (PB) experiment can only analyze the main effects.15 After the screening, attempts were made to further optimize factor levels and cell growth conditions. However, compared to the PB experiment, no more favorable progress was achieved. This indicates that the factor levels selected in the PB experiment were already within a relatively suitable range, validating the rationality of our initial design based on previous screening data and known effective ranges. Nevertheless, there is still room for further refinement via more intricate experimental designs such as the response surface methodology.
Although the cell analysis methods provided useful data regarding growth and aggregation, they failed to present a comprehensive view of the cell metabolic state. Future research could integrate transcriptomic or proteomic analyses to gain a deeper insight of how cells adapt to different medium components.
In summary, our study successfully achieved the suspension of HEK293 cells and developed a serum-free medium suitable for transient transfection through a systematic approach. By enabling efficient cell suspension with reduced aggregation, it has laid a solid foundation for the industrial-scale production of biopharmaceuticals.
Conclusion
In conclusion, through a series of experiments, this study has systematically achieved the transformation of HEK293 cells from adherent culture to serum-free suspension culture and has made good progress in optimizing the cell growth state. We identified the appropriate time point to bridge serum-free culture and suspension adaptation, and further optimized the culture medium. This also provides a reference for the suspension culture of other mammalian cells. The Plackett – Burman experiment effectively analyzed the impacts of multiple factors on cell growth. The data show that the initially selected factor levels are already relatively suitable. This not only validates our research design method but also paves the way for future experimental designs.
Despite these achievements, we are also aware of the limitations of the cell analysis methods. Based on this work, subsequent research can incorporate transcriptomic or proteomic techniques to deepen our understanding of cell adaptation mechanisms. Overall, the serum-free medium developed in this study and the optimized cell suspension state have laid a solid foundation for the industrial-scale production of biopharmaceuticals using mammalian cells, facilitating more efficient and reliable production processes.
Acknowledgement
The authors wish to thank Quacell for providing HEK293cells and lab conditions
Funding Sources
The author(s) received no financial support for the research, authorship,and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Wenyan Xu and H. Fai Poon: Conceptualization and methodology
- Software, Validation, Investigation and Data curation: Wenyan Xu and Yeqing Wu
- Formal analysis and writing: Bo Zou. and Guohao Xie.
References
- Jang M, PeteES, Bruheim The impact of serum-free culture on HEK293 cells: From the establishment of suspension and adherent serum-free adaptation cultures to the investigation of growth and metabolic profiles. Front Bioeng Biotechnol. 2022, 10, 964397.
CrossRef - Kimiz-Gebologlu I, Saglam-Metiner P, Ozaslan O, Ayyildiz-Tamis D, Deliloglu-Gurhan SIand Gulce-Iz S. Development of a serum free medium for HUMIRA® biosimilar by design of experiment approaches. Turkish Journal of Biochemistry. 2020, 45, 283 – 294.
CrossRef - Wu SY,Rish AJ, Skomo A, Zhao YX, Drennen JK, Anderson Rapid serum‐free/suspension adaptation: Medium development using a definitive screening design for Chinese hamster ovary cells. Biotechnol Prog. 2021, 37(4), e3154.
CrossRef - Tsao YS,Condon R, Schaefer E, Lio P, Liu Development and improvement of a serum-free suspension process for the production of recombinant adenoviral vectors using HEK293 cells. Cytotechnology. 2001, 37(3), 189-98.
CrossRef - Stone AT,Dhara VG, Naik HM, et al. Chemical speciation of trace metals in mammalian cell culture media: looking under the hood to boost cellular performance and product q Curr Opin Biotechnol. 2021, 71, 216-224.
CrossRef - Prabhu A, Gadgil M. Trace metals in cellular metabolism and their impact on recombinant protein production. Process Biochemistry.2021, 110, 251-262.
CrossRef - Melero A,Senna A, Domingues J, et al. Chelating Effect of Cellulose Acetate Hydrogel Crosslinked with EDTA Dianhydride Used as a Platform for Cell Gro Advances in Materials Science and Engineering. 2019.
CrossRef - Salazar A, Keusgen M, Hagen J Amino acids in the cultivation of mammalian cells. Amino Acids. 2016, 48(5), 1161-71.
CrossRef - Yamamoto K, Niwa A. Amino acid and vitamin requirements in mammalian cultured cells.Amino Acids.1993, 5, 1-16.
CrossRef - Hosios AM, Hecht VC, Danai LV, et al. Amino Acids Rather than Glucose Account for the Majority of Cell Mass in Proliferating Mammalian Cells. Developmental cell. 2016, 36 (5), 540-9 .
CrossRef - Agarabi CD, Schiel JE, Lute SC, et al. Bioreactor process parameter screening utilizing a Plackett-Burman design for a model monoclonal antibody.Journal of pharmaceutical sciences.2015, 104 (6), 1919-1928 .
CrossRef - Ritacco FV, Wu Y, Khetan A. Cell culture media for recombinant protein expression in Chinese hamster ovary (CHO) cells: History, key components, and optimization strategies. Biotechnology progress. 2018, 34(6), 1407–1426.
CrossRef - Salim T, Chauhan G, Templeton N, Ling W. Using MVDA with stoichiometric balances to optimize amino acid concentrations in chemically defined CHO cell culture medium for improved culture performance. Biotechnology and bioengineering. 2022,119(2), 452–469.
CrossRef - Pereira S, Kildegaard HF, Andersen MR. Impact of CHO Metabolism on Cell Growth and Protein Production: An Overview of Toxic and Inhibiting Metabolites and Nutrients. Biotechnology journal. 2018,13(3), e1700499.
CrossRef - Analytical Methods Committee Amctb No (2013). Experimental design and optimisation (4): Plackett-Burman designs. Analytical methods : advancing methods and applications, 5(8), 1901–1903.
CrossRef







