Manuscript accepted on :March 10, 2015
Published online on: 05-12-2015
Plagiarism Check: Yes
Ali Asgari1 and Farzad Mehrabi2*
1Department of Infectious Diseases, School of Medicine, AJA University of Medical Sciences,Tehran , Iran.
2Department of Neurology, Imam Reza hospital, AJA University of Medical Sciences, Tehran, Iran.
Abstract
We investigated the epidemiology and demographic features of multiple sclerosis in military personnel and their family members. Demographic and clinical records of 122 MS patients based on the revised Mc-Donald criteria were reviewed, which included; age, sex, marital status, presenting symptoms, type of disease, disability status and frequency of relapses. Out of 122 cases, seventy-seven were females and forty-five males. The female/male ratio was 1.7. The mean age at onset of disease was 29.9±6.2 for all of the patients. The mean duration of illness was 6.17±4.5. The majority of patients(60%) had relapsing-remitting pattern. Average disability was 3.4±2.45 (3.06±2.34 in females and 3.59±2.53 in males, P=0.03) on the Kurtzke disability status scale. There was no difference in epidemiologic and demographic features of multiple sclerosis between military personnel and their families and general population, therefore it seemed that military status had no important role in the prevalence of multiple sclerosis and rate of disability.
Keywords
Multiple sclerosis; Demography; Iran
Download this article as:| Copy the following to cite this article: Asgari A, Mehrabi F. Epidemiology of Multiple Sclerosis (MS) in Military Personnel : Demographic Study in Iran. Biomed Pharmacol J 2015;8(March Spl Edition) |
| Copy the following to cite this URL: Asgari A, Mehrabi F. Epidemiology of Multiple Sclerosis (MS) in Military Personnel : Demographic Study in Iran. Biomed Pharmacol J 2015;8(March Spl Edition). Available from: http://biomedpharmajournal.org/?p=2161> |
Introduction
Multiple sclerosis(MS) is a chronic inflammatory and autoimmune demyelinating disorder of the central nervous system that mostly affects young adults, ages 20-40 years old and women more than men1,2. Many population based studies over various geographical regions suggested the complex interactions between genetic and environmental factors in the development of MS3,4,5 .Although many factors like variability in surveyed population sizes, age, ethnic origins and composition, and the difficult quantification of patients have faced published regional studies with limited validity; achieving a good knowledge of the epidemiology and demographic features of MS in different areas gives new insight into the underlying causes of the disease6,7. Kurtzke believed that latitude plays a role in the frequency of MS and divided the world into 3 zones by prevalence. These zones consist of: 1) high (30 to 60 per 100,000) for the areas located farther away from the equator , medium (5 to 15 per 100,000) and low risk
( less than 5 per 100,000 ) which are more near to equator.8,9 According to Kurtzke, Middle East countries had been classified as low risk regions, but nowadays, several studies show higher risk in this area 10 .Epidemiology of MS in Iran has been a major topic of concern in last decade and several population based studies revealed a rapid increase in the incidence and prevalence of MS especially in females, in Iran.11
In this study our objective was to determine the epidemiology and demographic features of MS in the military personnel and their family members.
Material and Methods
We evaluated the medical records of 122 consecutive military personnel and their family members with multiple sclerosis admitted since April 2009 to 2014 to the neurology ward of the Imam Reza Hospital, retrospectively. The diagnosis of multiple sclerosis was confirmed by one of the study neurologists based on the revised McDonald criteria by international panel for MS diagnosis12. Patients with known inflammatory, infectious, malignancy and autoimmune disease not included in the study. In the case of missing clinical data if possible, phone contact with the patients were made; otherwise, they were excluded from the study. Medical records of patients who previously diagnosed with multiple sclerosis, were reviewed by neurology resident and all demographics as well as clinical variables such as type of disease, duration of disease, type of first attack and expanded disability status scale (EDSS) were extracted from the records.
Data analysis
Statistical analysis was performed using SPSS software (21th edition). Applied methods composed of descriptive statistics (mean, median and standard deviation) for frequency, t-test and Anova for independent variables. Differences between the two groups were estimated using Chi-Square test and correlation between different clinical factors were evaluated using Pearson,s correlation for continuous variables or Spearman,s rank correlation for numerical ones. For all of the analyses, P value <0.05 were considered statistically significant.
Result
The total number of patients enrolled in this study was 122. Forty five were male and seventy seven were female with female/male ratio 1.71. In total, 64.8% of patients were married, 28.6% single and 6.6% widowed/divorced with mean EDSS(SD) 3.2(2.34), 2.31(2.1), 2.2(2.07) respectively, (Pvalue=0.133) . Unfortunately, as one of limitations of our study, it was not possible to calculate prevalence of MS in military personnel, because of lacking data about the exact number of military personnel and family members. The mean age was 36.1(7.7) years and the mean age of onset was 29.9(6.2) years(29.6±6 for female and 30.4±6.5 for male with Pvalue=0.8). As shown in Table 1, multiple sclerosis was most frequent in the 31-35 years age group in both females and males. The youngest patient was a 14-year-old girl and the oldest was a 46-year-old woman at the onset of disease. We considered the age below 16 years as an early-onset age and above 50 years as late-onset age of MS.13 In total, only two patients(1.6%) were in the early-onset age and none of the patients were in the late-onset age category. The mean duration of disease was 6.17±4.5year (6.20±4.9 for female and 6.11±4 for male, P=0.35). The frequency of disease type in 122 patients were: relapsing-remitting multiple sclerosis(RRMS) (60%), secondary progressive multiple sclerosis(SPMS) (36%) and primary progressive multiple sclerosis(PPMS) (4%); Although RRMS was more common among women with sensory symptoms as an initial attack, SPMS more frequently occurred in men with motor symptoms. Progressive relapsing multiple sclerosis(PRMS) cases were included in the PPMS group. In this study we looked that most patients with RRMS had sensory symptoms(41.1%) and most of the SPMS and PPMS had motor symptoms as their initial attack (36.4% and 80%, respectivetly) with Pvalue=0.003. The mean EDSS was 3.4± 2.45 (3.06±2.34 for female and 3.59±2.53 for male, P= .03). With respect to the initial feature of the disease, visual (35.1%) was the most common symptom followed by sensory(31.3%), cerebellar(17.2%) and motor symptom (16.4%); In addition, as illustrated in figure 1, on the basis of duration adjusted multivariate analysis , patients with first attack of sensory type had EDSS(SD) 1.35(1.71) subsequently, visual 2.53(2.32), cerebellar 3.19(1.43) and motor 5.34(1.77) with Pvalue=0.003 . Patients with cerebellar symptoms had higher rate of relapse(>1 relapse per year) (66.7%), followed by motor(65%), visual(27.9%) and sensory symptoms(18.4%) ( Pvalue=.045).
Table 1: Frequencies of multiple sclerosis in different age-onset groups and gender
| Age | Male
N(%) |
Female
N(%) |
total |
| <16 | 1(2.2%) | 1(1.2%) | 2(1.7%) |
| 16-20 | 2(4.4%) | 6(7.8%) | 8(6.6%) |
| 21-25 | 8(17.8%) | 12(15.6%) | 20 (16.4%) |
| 26-30 | 11(24.4%) | 22(28.6%) | 33(27%) |
| 31-35
36-40
≥40 |
16(36.6%)
3(6.7%) 4(7.9%) |
26(33.8%)
8(10.4%) 2(2.6%) |
42(34.4%)
11(9%) 6(4,9%) |
| total | 45(100%) | 77(100%)) | 122(100%) |
Table 2: Comparison of the clinical pattern and disability according by sex
| Male
N(%) |
Female
N(%) |
Pvalue | |
| Age: mean (SD) in years
|
37.1(8.1) | 35.6(7.4) | 0.69 |
| Age at onset: mean (SD) in years
|
30.4(6.5) | 29.6(6) | 0.8 |
| Marital status | 0.73 | ||
| married
|
30(66.7%) | 49(63.6%) | |
| single
|
13(28.9%) | 22(28.7%) | |
| widowed/divorced | 2(4.4%) | 6(7.7%) | |
| EDSS (SD) | 3.59(2.53) | 3.06(2.34) | 0.03 |
| Duration of disease (SD) | 6.11(4) | 6.2(4.9) | 0.35 |
| Type of disease (SD) | 0.67 | ||
| RRMS | 25(55.6%) | 48(62.3%) | |
| SPMS | 17(37.8%) | 27(35.1%) | |
| PPMS | 3(6.6%) | 2(2.6%) | |
| Type of first attack | 0.25 | ||
| visual | 18(40%) | 25(32.5%) | |
| cerebellar | 7(15.5%) | 14(18.1%) | |
| sensory | 9(20%) | 29(37.7%) | |
| motor | 11(24.5%) | 9(11.7%) | |
| Relapse per year | 0.29 | ||
| ≤ 1 | 28(62.2%) | 55(71.4%) | |
| ˃ 1 | 17(37.8%) | 22(28.6%) |
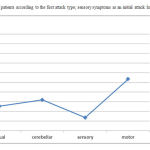 |
Figure 1: EDSS of patients according to the first attack type; sensory symptoms as an initial attack had the lowest EDSS |
Discussion
Previously, Iran was considered as a low risk area for MS14,15; however, recently several studies in different part of Iran showed significant increase in the prevalence of multiple sclerosis and regarded as the medium risk area16-21. MS is predominant among women compared to men6,22,23. According to former studies in different parts of Iran, female preponderance has been observed among Iranian MS patients with female/male ratio varied from 1.2 to 3.619,24-27. In agreement with these results, present study showed the female to male ratio of 1.7. Moreover, the results of our study revealed that the RRMS was the dominant pattern which is in consistent with previous reports20,21,23. Also, we found that SPMS more common than PPMS and frequency of men suffering from progressive disease higher than women, which is similar to Rezaali et al reports28. However, Montalban et al and Kalanie et al23,29 have shown a higher frequency of PPMS compared to SPMS, and prevailing frequency of women with progressive disease in comparison with men. In this study, the age at onset of MS in women was lower than men, approximately 1 year which is statistically insignificant (P=0.8). In addition, 86.9% of the patients were 20-40 years old with the peak incidence age at onset 31-35 and only 1.6% of the patients were early onset MS(<16 years old). According to the present study, we estimated that MS mostly occurs in adults, and early and late onset age groups account for negligible percentage of patients. This is in accordance to the results of several earlier studies19,20,30-33. In agreement with the previous data from epidemiological surveys in Tehran and Qom20,28, we demonstrated in this study that prevalence of MS and EDSS were higher among married, that statistically insignificant(P=0.73 and 0.133 respectively); Additionally we did not know whether the disease happened before marriage or after. There are few studies concerning the role of marriage status in MS34. The average disability of our patients (EDSS) was 3.4± 2.45, with higher rate of disability among men than in women (3.59±2.53 for male, 3.06±2.34 for female, P= .03) and the average duration of illness was 6.17±4.5year (6.20±4.9 for female and 6.11±4 for male, P=0.35) which is similar to the Ebrahimi et al.35, who have mentioned greater disability among men and no statistically significant differences between men and women with regards to the duration of disease. It seems that men suffered from more disability, and the rate of progression of MS in men was faster than in women. Tremlett and his colleagues suggested a 38% faster progression in males, and that younger age at disease onset predicts a slower rate of progression36.This is in concordance with our findings that women tend to be afflicted earlier than men and rate of progression of disease was slower and older age at onset of disease was associated with higher EDSS(P=.004). We illustrated that visual and sensory symptoms were the most frequent initial presentation of multiple sclerosis (66.6%), followed by cerebellar (17.2%) and motor deficit (16.4%), as multiple epidemiological investigations have shown similar results22,27,35. As Lublin et al and Hammond et al38,39, mentioned that the advancement of disability depends on individual patient, the gap between attacks, the type of MS, symptoms and signs of the first attack, we showed that higher frequency of attacks per year and motor and cerebellar symptoms as first attack were related with greater EDSS and rapid rate of progression(P=.01 and .003 respectively). Moreover, patients with cerebellar and motor symptoms had higher rate of relapse per year (P=0.045).
In conclusion, we found that multiple sclerosis was more frequent within the ages 31-35 years and there was a linear relationship between age at onset and rate of progression. Also, type of first attack, frequency of relapses and gender had prognostic significance.
There was no difference between epidemiologic and demographic features of multiple sclerosis in military personnel and their families and general population, and it seemed that military status had no considerable effect on the frequency of multiple sclerosis and rate of disability.
References
- Frohman EM, Rache MK, Raine CS. Multiple sclerosis-the plaque and its pathogenesis. N Engl J Med 2006; 354: 942-55.
- Alonso A, Hernan MA. Temporal trends in the incidence of multiple sclerosis: a systematic review. Neurology 2008; 71:129-35.
- Willer CJ, Ebers GC. Susceptibility to multiple sclerosis: interplay between genes and environment. Curr Opin Neurol 2000; 13(3): 241-7.
- Sotgiu S, Pugliatti M, Fois ML, Arru G, Sanna A, Sotgiu MA, et al. Genes, environment, and susceptibility to multiple sclerosis. Neurobiol Dis 2004; 17(2): 131-43.
- Pugliatti M, Sotgiu S, Rosati G. The worldwide prevalence of multiple sclerosis. Clin Neurol Neurosurg 2002; 104:182-91.
- Rosati G. The prevalence of multiple sclerosis in the world: an update. Neurol Sci.2001; 22(2):117-39.
- Ramagopalan SV, Sadovinck AD. Epidemiology of multiple sclerosis. Neurol Clin 2011; 29(2): 207-17.
- Kurtzke JF. A reassessment of the distribution of multiple sclerosis. Part one. Acta Neurol Scand. 1975; 51(2):110-36.
- Kurtzke JF. On the prevalence of multiple sclerosis in Serbia. Neuroepidemiology 2011; 37(2): 107-8.
- Benamer HT, Ahmed ES, Al-Din AS, GrossetDG: Frequency and clinical patterns of multiple sclerosis in Arab countries: a systematic review. J Neurol 2009; 278: 1–4.
- Pakpoor J, Ramagopalan S. Multiple sclerosis and the Iranian revolution: reply to the comment by Magzhi et al. Neuroepidemiology 2012; 40: 70.
- Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, et al. Diagnostic criteria for multiple sclerosis : 2010 revision to the McDonald criteria. Ann Neurol 2011; 69(2): 292-302.
- Renoux C, Vukusic S, Mikaeloff Y, Edan G, Clanet M, Dubois B, et al. Natural history of multiple sclerosis with childhood onset. N Engl J Med 2007; 356(25): 2603-13.
- Kurtzke JF. Geographic distribution of multiple sclerosis: An update with special reference to Europe and the Mediterranean region. Acta Neurol Scand 1980; 62(2): 65-80.
- Poser CM. The epidemiology of multiple sclerosis: a general overview. Ann Neurol 1994; 36(Suppl 2): S180-S193.
- Moghtaderi A, Rakhshanizadeh F, Shahraki-Ibrahimi S. Incidence and prevalence of multiple sclerosis in southeastern Iran. Clin Neurol Neurosurg 2013; 115(3): 304-8.
- Hashemilar M, Savadi-Ouskui D, Farhoudi M, Ayromlou H, Asadollahi A. Multiple sclerosis in East Azerbaijan, North West Iran. Neurology Asia 2011; 16(2): 127-31.
- Jajvandian R, Ali Babai A, Torabzadeh S, Rakhshi N, Nikravesh A. Prevalence of multiple sclerosis in North Khorasan province, northern Iran. Proceeding of the 5th Joint triennial congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis; 2011 Oct 20; Amsterdam, The Netherlands.
- Saadatnia M, Etemadifar M, Maghzi AH. Multiple sclerosis in Isfahan, Iran. Int Rev Neurobiol 2007; 79:357-75.
- Sahraian MA, Khorramnia S, Ebrahim MM, Moinfar Z, Lotfi J, Pakdaman H. Multiple sclerosis in Iran: a demographic study of 8,000 patients and changes over time. Eur Neurol 2010; 64(6): 331-6.
- Etemadifar M, Janghorbani M, Shayegannejad V, Ashtari F. Prevalence of multiple sclerosis in Isfahan, Iran. Neuroepidemiology 2006; 27:39–44.
- Moreira MA, Felipe E, Mendes MF, Tilbery CP. Multiple sclerosis: descriptive study of its clinical forms in 302 cases. Arq Neuropsiquiatr 2000; 58(2B): 460-6.
- Kalanie H, Gharagozli K, Kalanie AR. Multiple sclerosis: report on 200 cases from Iran. Mult Scler 2003; 9(1): 36-8.
- Abedini M, Habibi Saravi R, Zarvani A, Farahmand M. Epidemiology of multiple sclerosis in Iran, Mazandaran province, 2007.Mazandaran Journal of Medical Science 2008; 66:82-7.
- Nabavi SM, Pour farzam Sh, Ghasemi H. Clinical Course and prognosis of 203 patients with MS in Shahid Mostafa Khomeini Hospital, Tehran 2002. Tehran University Medical Journal2006; 64:90-7.
- Ale-Yasin H, Sarai A, Alaeddini F, Ansarian E, Lotfi J, Sanati MH. Multiple sclerosis: A study of 318 cases. www.ams.ac.ir/aim/0251/aim/025124.htm. [Accessed 2010/10/07].
- YousefiPour GA, Rasekhi AR. Multiple sclerosis: A risk factor analysis in Iran. www.ams.ac.ir/aim/0253/0253191.htm [Accessed 2010/10/07]
- Rezaali S, Khalilnezhad A, Naser Moghadasi A, Chaibakhsh S, Sahraian MA. Epidemiology of multiple sclerosis in Qom: demographic study in Iran. Ir J Neurol 2013; 12(4): 136-43.
- Montalban X, Rio J. Primary progressive multiple sclerosis. Neurol Sci 2001; 22(Suppl 2): S41-S48.
- Cole GF, Stuart CA. A long perspective on childhood multiple sclerosis. Dev Med Child Neurol 1995; 37(8): 661-6.
- Ghezzi A, Deplano V, Faroni J, Grasso MG, Liguori M, Marrosu G, et al. Multiple sclerosis in childhood: clinical features of 149 cases. Mult Scler 1997; 3(1): 43-6.
- Etemadifar M, Nasr Esfahani AH, Khodabandehlou R, Maghzi AH. Childhood-onset multiple sclerosis: Report of 82 patients from Isfahan, Iran. Arch Iranian Med 2007; 10:152-6.
- Thannhauser JE, Mah JK, Met LM. Adherence of adolescents to multiple sclerosis disease-modifying therapy. Peiatr Neurol 2009; 41:119-23.
- Tepavcevic DK, Pekmezovic T, Drulovic J. Quality of life assessment in patients with multiple sclerosis. Vojnosanit Pregl 2009; 66(8): 645-50.
- Ebrahimi HA, Sedighi B. Prevalence of multiple sclerosis and environmental factors in Kerman province, Iran. Neurology Asia 2013; 18(4): 385-9.
- Tremlett H, Paty D, Devonshire V. Disability progression in multiple sclerosis is slower than previously reported. Neurology 2006; 66(2):172-7.
- Sharafaddinzadeh N, Moghtaderi A, Majdinasab N, Dahmardeh M, Kashipazha D, Shalbafan B: The influence of ethnicity on the characteristics of multiple sclerosis: a local population study between Persians and Arabs. Clin Neurol Neurosurg 2013; 115: 1271–5.
- Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis.Neurology 1996; 46:907-9.
- Hammond SR, McLeod JG, Macaskill P, et al. Multiple sclerosis in Australia: prognostic factors. J Clin Neurosci 2000; 7:16-9.







