Manuscript accepted on :16-Mar-2019
Published online on: 09-04-2019
Plagiarism Check: Yes
Reviewed by: Dr. Amarendranath Choudhury
Second Review by: Muthulakshmi
Fatmawaty1, Ni G. M. Anggreni1, Naufal Fadhil1 and Vivitri D. Prasasty*2
1Department of Medical Chemistry, Faculty of Medicine, University of Indonesia, Jalan Salemba Raya 6, Jakarta 10430, Indonesia.
2Faculty of Biotechnology, Atma Jaya Catholic University of Indonesia, Jalan Jenderal Sudirman 51, Jakarta 12930, Indonesia.
Corresponding Author E-mail: vivitri.dewi@atmajaya.ac.id
DOI : https://dx.doi.org/10.13005/bpj/1686
Abstract
Piper crocatum and Persea americana Mill leaves are commonly used in traditional medicinal remedies, such as antidiabetes, antitumors and Alzheimer treatment. However, the extensive use of plant extracts over worldwide becomes significant concerns including its safety, efficacy, and quality. Therefore, the accurate scientific evaluation has become a precondition for acceptance of herbal health claims. The aim of this study was to determine the antioxidant activities of Piper crocatum Ruiz & Pav and Persea americana Mill leaf extracts. Isolation of antioxidant fractions were conducted using organic solvent extraction techniques. Antioxidant assays were conducted by using in vitro and in vivo methods involving DPPH and MDA methods. In this study, in vitro assays of Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts showed the best activity in water fraction. Moreover, in vivo assays of both plant leaf extracts showed the best dose at 8 mg. Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts have been successfully determined in antioxidant actions in vitro and in vivo. Persea americana Mill in water and ethanol solvents exhibit strong antioxidant properties. Meanwile, Piper crocatum Ruiz & Pav exhibit moderate activity in water and weak antioxidant activity in ethanol. Both plant leaves showed that 8 mg dose was better than the dose of 4 mg and 16 mg in vivo.
Keywords
Antioxidant; In Vitro; In Vivo; Piper Crocatum; Persea Americana
Download this article as:| Copy the following to cite this article: Fatmawaty F, Anggreni N. G. M, Fadhil N, Prasasty V. D. Potential In Vitro and In Vivo Antioxidant Activities from Piper Crocatum and Persea Americana Leaf Extracts. Biomed Pharmacol J 2019;12(2). |
| Copy the following to cite this URL: Fatmawaty F, Anggreni N. G. M, Fadhil N, Prasasty V. D. Potential In Vitro and In Vivo Antioxidant Activities from Piper Crocatum and Persea Americana Leaf Extracts. Biomed Pharmacol J 2019;12(2). Available from: https://bit.ly/2Un37qV |
Introduction
Many medicinal plants family member from Indonesia known as potential candidate for natural antioxidant, such as mengkudu (Moringa citrifolia),1 rambutan (Nephelium lappaceum sp.),2 betel (Piper betle)3,4 and avocado (Persea americana Mill).5 Red betel is commonly found in tropical area and its leaves are conventionally used by the surrounding natives as medical treatment to overcome several diseases such as diabetes, inflammation6 and wound healing.3 Beside its unique odor from its essential content, red betel leaves also possess several functional active compounds including flavonoid, alkaloid, saponins, and tannins.7 Many recent studies have reported that active compounds of medicinal plants exhibit hypoglycemic activity and antioxidant activity8-10 whereas comparable to which shown by Butylated hydroxytoluene (BHT) and vitamin E.11
Several studies have been carried out to evaluate antioxidant activities of avocado fruit and its part. Antioxidant activity of avocado peel extract had been evalutaed using radical scavenging assay including Ferric Reducing Antioxidant Power (FRAP) and Oxygen Radical Absorbance Capacity (ORAC).12 Daiuto et al. also evaluated antioxidant activities of seed and peel parts of avocado.13 Avocado leaves were enriched with phenolic bioactive compounds which potential as a natural antioxidants14 and positively contains alkaloids, flavonoids, saponins, tannins and steroids under methanolic solution to extract avocado leaves.15 Avocado leaves have been empirically used as a diuretic, analgesic, anti-inflammatory, hypertensive, hypoglycemic, diarrhea, sore throat and hemorrhage cure.16,17 Meanwhile, avocado fruit is nutritious as a preservative and antioxidant.18,19 Avocado flesh can be used as an anti-hyperlipidemia and has the potential to reduce the risk of metabolic síndrome.20-22
The potentials use of red betel (Piper crocatum Ruiz & Pav) and avocado (Persea americana Mill) leaf extracts as natural source of antioxidant must be completed by the understanding of its safety and possible side effects.23 Moreover, we also carried out antioxidant activity assays of red betel and avocado leaf extracts to shed light on the safety of its potential application as natural source of antioxidant without any further negative side effects. We have employed 1,1-diphenyl-2-picrylhidrazyl (DPPH) in so called cytotoxicity assay. The specimens’ resistance on DPPH will provide the insight of their capability to overcome free radicals without necessarily distinguish the radical’s type.24 The aim of this research was to determine the antioxidant activities of Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts in vitro and in vivo by using DPPH and MDA methods in mice models.
Method
Preparation of Extract Simple
Red betel and avocado leaves were obtained from Aromatic Research Center (Balitro) in Bogor. Samples were cleaned, dried and ground into fine powder. To obtain the extract, the maceration process is done by soaking the fine powder as much as 200 g in 500 mL of solvents. Four different solvents were used in this study, including ethanol, ethyl acetate, hexane and water. The sample solution was stirred every two hours at room temperature to extract the bioactive compounds. After two days, the soaking result was filtered. The residue was again soaked in a fresh volume of solvents and the soaking process was repeated three times until clear filtrates were obtained. The resulting extracts were then evaporated using rotary evaporator at 35°C, 50 rpm. The concentrated extracts were dried by oven at 40°C until reached constant dry weight.
Phytochemical Analysis
Phytochemical analysis was evaluated qualitatively to determine its bioactive compounds in plant leaf extracts of Persea americana Mill and Piper crocatum Ruiz & Pav leaf at various solvent fractions. Phytochemical analyses were caried out including glycoside, saponins, flavonoids, alkaloids, triterpenoid, steroids, essential oils, and tannins.25
In Vitro Antioxidant Assay with DPPH Method
In this study, antioxidants were determined by DPPH method. Vitamin C was used as standard solution with concentrations of 2.5, 5.0, 10, and 20 μg/mL. Moreover, vitamin C were reacted with 0.5 ml DPPH (1 mM in methanol solvent). The solution was homogenized by vortex and was allowed to complete at room temperature for 30 min., then the absorbance of solution was measured using spectrophotometer at 515 nm. The results obtained were given as percentage inhibition. Furthermore, all extract was done following the same method above, at various concentration of 10, 50, 100, 200 μg/mL, respectively. The IC50 value represented as the concentration required for 50% inhibition of DPPH was calculated using the graph of inhibition percentage versus the extract concentration in mg/g (w/w), by following equation below.
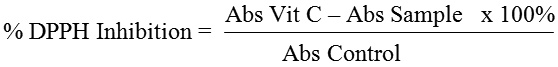
The intensity of IC50 of antioxidant activity may vary in different medicinal plant as categorized on Table 1.
Table 1: Category of antioxidant activity strength in vitro against DPPH.
| Intensity of IC50 | Value (μg/mL) |
| Very active | <50 |
| Active | 50-100 |
| Medium | 101-250 |
| Weak | 250-500 |
| Inactive | >500 |
In Vivo Antioxidant Assay with Malondialdehyde (MDA) Method
Plasma MDA levels measured by means of a thibabituric acid (TBA) reaction in mice groups before and after treatment. The in vivo test was began by weighing a mouse (190 g – 200 g) and labeled. The extract was administered to the mice daily for 10 days with the dose of 4 mg/200 g body weight (BW) for the first group, 8 mg/200 g BW for the second group, and 16 mg/200g BW for the third group, and a dose of vitamin C was 2.08 mg/200 g BW for positive control group and 1 ml of water for negative control group. On the tenth day, mice were given maximum physical activity that was in the form of swimming for 20 minutes. Observation was done on all mice activities in water. Furthermore, 2 mL blood sample was taken from each mouse through its tail after centrifugation to separate the blood plasma. Measurement of MDA levels in blood plasma was done by reacting 250 μl of blood plasma with 100 μl of 8.1% Sodium Duodecyl Sulfate (SDS) and 750 μl of 0.5 M HCl, 750 μl TBA and 125 μl aquabidest. All substance was vortexed subsequently become homogeneous. The solution was then heated at 90°C for 15 min., then cooled for 10 min. After cooling in solution, 2.5 ml n-Butanol and 500 μl aquabidest was added. The absorbance of MDA levels in plasma were measured by fluorometer at 520 nm excitation and 550 nm emission.
Table 2: In vivo treatment of antioxidant activity with plant extract.
| Mice group | Fraction | Dose (mg sample/g mice body wight) |
| A1 | Persea americana Mill | 4 mg/200 g |
| A2 | Persea americana Mill | 8 mg/200 g |
| A3 | Persea americana Mill | 16 mg/200 g |
| B1 | Piper crocatum Ruiz & Pav | 4 mg/200 g |
| B2 | Piper crocatum Ruiz & Pav | 8 mg/200 g |
| B3 | Piper crocatum Ruiz & Pav | 16 mg/200 g |
| D | Vitamin C (positive control) | 2.08 mg/200 g |
| E | Water (negative control) | – |
Sample size use in this research was following Federer’s formula:
(k-1) (n-1) > 15
(8-1) (n-1) > 15
n = 3
k = number of group
n = number of mice in group
Results
Phytochemical Analysis and In Vitro Antioxidant Activities
The results of phytochemistry analysis of Persea americana Mill Piper crocatum Ruiz and Puf are shown in Table 3. The in vitro antioxidant activity of Percea americana Mill and Piper crocatum Ruiz & Pav are showed in Table 4 and Table 5, respectively. Potential antioxidant activity of Persea americana Mill leaves extract was tested by comparison with antioxidant activity of vitamin C. The parameter used for antioxidant activity against DPPH radical was IC50 which indicated that concentration of compound is required to reduce DPPH radical by 50%. The smaller the value of IC50, the more effective the function of the assayed extracts as antioxidant agents.
Table 3: Phytochemistry analysis ethanol extract and ethyl acetat of Percea americana Mill and Piper crocatum Ruiz and Puf.
| Phytochemical
Constituent |
Percea americana Mill extract | Piper crocatum Ruiz and Puf extract | ||||||
| ethanol | water | ethyl acetate | hexane | ethanol | water | ethyl acetate | hexane | |
| Glycosides | + | + | + | – | + | + | + | – |
| Saponin | + | + | + | – | + | + | – | – |
| Flavonoid | + | + | + | – | + | + | – | – |
| Alkaloid | + | + | + | – | + | + | + | – |
| Triterpenoid/Steroid | + | – | + | – | + | – | – | – |
| Essential oil | + | + | + | – | + | + | + | – |
| Tannin | + | + | + | – | + | + | – | – |
Table 4: In vitro antioxidant activities of leaf extracts of Persea americana Mill in various extraction fractions.
| Extract Fraction | Concentration (μg/mL) | % Inhibition | IC50 ((μ/mL) |
| Vitamin C (Positive control) | 5 | 42.5 | 7.03 |
| Ethanol | 50 | 66.7 | 35.90 |
| Ethyl Acetate | 50 | 28.9 | 157.30 |
| Hexane | 50 | 8.0 | 440.80 |
| Water | 50 | 91.6 | 29.70 |
Table 5: In vitro antioxidant activities of leaf extracts of Piper crocatum Ruiz & Pav in various extraction fractions.
| Extract Fraction | Concentration (μg/mL) | % Inhibition | IC50 ((μ/mL) |
| Vitamin C (Positive control) | 5 | 42.5 | 7.03 |
| Ethanol | 100 | 33 | 185.29 |
| Ethyl Acetate | 10 | 0.8 | 202.78 |
| Hexane | 10 | 0.1 | 552.2 |
| Water | 10 | 6.2 | 81.24 |
The results of in vivo assay of Persea americana Mill leaf extracts showed MDA levels before treatment was higher than MDA levels after treatment at 4 mg extract concentration (Fig 1). Meanwhile, Piper crocatum Ruiz & Pav leaf extracts also showed similar MDA levels before treatment was higher than MDA levels after treatment at 4 mg extract concentration (Fig 2).
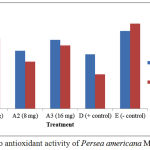 |
Figure 1: In vivo antioxidant activity of Persea americana Mill leaf extracts.
|
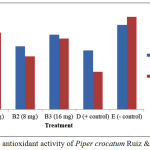 |
Figure 2: In vivo antioxidant activity of Piper crocatum Ruiz & Pav leaf extracts.
|
Discussions
For both studies of in vitro and in vivo assays, we used water as a negative control and vitamin C as a positive control. These methods were carried out to evaluate the antioxidant potential of Persea americana Mill and Piper crocatum Ruiz & Pav extracts against free radicals. In vitro antioxidant assays are very beneficial, cost-effective and time saving to investigate the antioxidant potential of Persea americana Mill and Piper crocatum Ruiz & Pav extracts before getting the extract to the in vivo mouse model for the antioxidant activity by free radical scavenging. Vitamin C as positive control is a water-soluble vitamin with the mechanism works as an antioxidant by stopping the propagation stage (chain-breaking antioxidant) and provides rapid electron transfer inhibiting lipid peroxidation, thus making the potent antioxidant of vitamin C.24 The differences of IC50 value of Persea americana Mill extract in water, ethanol, ethyl acetate and hexane are caused by the solvent polarity. Water and ethanol are polar solvents, ethyl acetate is semi polar and hexane is non-polar solvents.26,27 Identification of major and minor compounds in Persea americana Mill extract, ethanol, water and ethyl acetate were found to be most effective in the extraction of different compounds. Meanwhile Piper crocatum Ruiz & Pav extract pattern most effective in ethanol and water, but it was moderate in ethyl acetate. However, both Persea americana Mill and Piper crocatum Ruiz & Pav extracts were found none of constituent compounds in hexane.
Aqueous fraction of Persea americana Mill leaf extract can neutralize free radicals maximally, with IC50 value was 29.7, but not as good as antioxidant vitamin C (IC50 = 7.03). This is due to the content of vitamin C is pure antioxidant compound that can potentially neutralize the free radicals better than plant extracts. The aqueous fraction of Piper crocatum Ruiz & Pav leaf extracts also showed the the best IC50 value compared to other solvent extracts. However, it still higher than vitamin C as positive control (IC50 = 7.03). Both aqueous plant extracts contained bioactive compounds such as flavonoid, alkoloid polyphenols, glucosides and terpenoids. These compounds contribute to antioxidant activities, but they are not supposed to be pure compounds and still bound to one another with the glycoside group.28
In vivo assay of Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts, there were decreases of MDA levels in all groups, except negative control as shown on Figure 1 and Figure 2, respectively. This is because both leaf test extracts potentially as exogenous antioxidants, so it can suppress the free radicals that are formed, after the mice are given the physical burden of swimming for 15 minutes. In the positive control of decreased MDA levels was much better than with MDA level reduction at concentrations of 4 mg, 8 mg and 16 mg groups. The increased plasma MDA levels were elevated in the negative control group after the mice treatment through swimming, suggesting that the endogenous antioxidants in the body of mice were unable to neutralize free radicals. Endogenous and physiological reactive oxygen species (ROS) are largely generated within mitochondria as by-products of respiratory electron transport chain of normal cellular metabolism. Excessive exposure to ROS may interfere the redox homeostasis causes the cell vulnerable to ROS, and ultimately can induce cell damage.
Conclusion
Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts have been successfully determined as antioxidant agents in vitro and in vivo. Persea americana Mill in water and ethanol solvents exhibit strong antioxidant properties. Meanwhile, Piper crocatum Ruiz & Pav exhibit moderate activity in water and weak antioxidant activity in ethanol. Both plant leaves showed that 8 mg dose was better than the dose of 4 mg and 16 mg, in vivo. The antioxidant systems of exogenous antioxidants could repress the ROS level by regulating the genes expression and related metabolic networks to maintain the redox balance and support cellular component for stress adaption. Thus, the antioxidant remedies using Persea americana Mill and Piper crocatum Ruiz & Pav leaf extracts offer a promising strategy to prevent and treat the diseases caused by the excessive ROS exposure.
Acknowledgements
We would like to thank Mr. Winarno and staff in Animal Facility of Faculty of Medicine for providing in vivo mice study. This research was funded by DRPM University of Indonesia.
Conflict of Interest
There is no conflict of interest.
References
- Krishnaiah D., Bono A., Sarbatly R., Anisuzzaman S. Antioxidant activity and total phenolic content of an isolated Morinda citrifolia L. methanolic extract from Poly-ethersulphone (PES) membrane separator. J King Saud Univ Eng Sci. 2015;27:63-67.
CrossRef - Palanisamy U., Cheng H. M., Masilamani T., Subramaniam T., Ling L. T., Radhakrishnan A. K. Rind of the rambutan, Nephelium lappaceum, a potential source of natural antioxidants. Food Chem. 2008;109:54-63.
CrossRef - Dasgupta N., De B. Antioxidant activity of Piper betle L. leaf extract in vitro. Food Chem. 2004;88:219-224.
CrossRef - Rathee J. S., Patro B. S., Mula S., Gamre S., Chattopadhyay S. Antioxidant activity of Piper betel leaf extract and its constituents. J Agric Food Chem. 2006;54:9046-9054.
CrossRef - Rodríguez-Carpena J. G., Morcuende D., Andrade M. J., Kylli P., Estévez M. Avocado (Persea americana Mill.) phenolics, in vitro antioxidant and antimicrobial activities and inhibition of lipid and protein oxidation in porcine patties. J Agric Food Chem. 2011;59:5625-5635.
CrossRef - Anugrahwati M., Purwaningsih T.,Manggalarini J., Alnavis N., Wulandari D., Pranowo H. Extraction of Ethanolic Extract of Red Betel Leaves and Its Cytotoxicity Test on HeLa Cells. Procedia Eng. 2016;148:1402-1407.
CrossRef - Irawan C. GC-MS composition of leaf extract of Piper cf. arcuatum blume and their antioxidant activity and toxicity studies. J Pharmacogn Phytochem. 2017;6:461-468.
- Hassan S. K., El-Sammad N. M., Mousa A. M., Mohammed M. H., Farrag A. R. H., Hashim A. N. E. Hypoglycemic and antioxidant activities of Caesalpinia ferrea Martius leaf extract in streptozotocin-induced diabetic rats. Asian Pac J Trop Biomed. 2015;5:462-471.
CrossRef - Arya A., Nyamathulla S., Noordin M. I., Mohd M. A. Antioxidant and hypoglycemic activities of leaf extracts of three popular terminalia species. E-J Chem. 2012;9:883-892.
CrossRef - Pamunuwa G., Karunaratne D., Waisundara V. Y. Antidiabetic properties, bioactive constituents, and other therapeutic effects of Scoparia dulcis. J Evid Based Complementary Altern Med. 2016.
CrossRef - Hope N., Bellare A. A comparison of the efficacy of various antioxidants on the oxidative stability of irradiated polyethylene. Clin Orthop Relat Res. 2015;473:936-941.
CrossRef - Calderon-Oliver M., Escalona-Buendia H. B., Medina-Campos O. N., Pedraza-Chaverri J., Pedroza-Islas R., Ponce-Alquicira E. Optimization of the antioxidant and antimicrobial response of the combined effect of nisin and avocado byproducts. LWT-Food Sci Technol. 2016;65:46-52.
CrossRef - Daiuto E. R., Tremocoldi M. A., de Alencar S. M., Vieites R. L., Minarelli P. H. Chemical composition and antioxidant activity of the pulp, peel and by products of avocado ‘Hass. Rev Bras Frutic. 2014;36:417-424.
CrossRef - Yamassaki F. T., Campestrini L. H., Zawadzki-Baggio S. F., Maurer J. B. B. Avocado leaves: Influence of drying process, thermal incubation and storage conditions on preservation of polyphenolic compounds and antioxidant activity. Int J Food Prop. 2017;20:1-14.
CrossRef - Rahman N., Dewi N. U., Bohari. Phytochemical and Antioxidant Activity of Avocado Leaf Extract (Persea americana Mill.). Asian J Sci Res. 2018;11:1-7.
- Brai B. I. C., Odetola A. A., Agomo P. U. Effects of Persea americana leaf extracts on body weight and liver lipids in rats fed hyperlipidaemic diet. Afr J Biotechnol. 2007;6.
- Jesus D., Oliveira J. R., Oliveira F. E., Higa K. C., Junqueira J. C., Jorge A. O., Back-Brito G. N., Oliveira L. D. Persea americana glycolic extract: In vitro study of antimicrobial activity against Candida albicans biofilm and cytotoxicity evaluation. Sci World J. 2015.
- Comerford K. B., Ayoob K. T., Murray R. D., Atkinson S. A. The role of avocados in complementary and transitional feeding. Nutrients. 2016;8:316.
CrossRef - Ayala‐Zavala J. F., Rosas‐Domínguez C., Vega‐Vega V., González‐Aguilar G. A. Antioxidant Enrichment and Antimicrobial Protection of Fresh‐Cut Fruits Using Their Own Byproducts: Looking for Integral Exploitation. J of Food Sci. 2010;75.
- Dreher M. L., Davenport A. J. Hass avocado composition and potential health effects. Crit Rev Food Sci Nutr. 2013;53:738-750.
CrossRef - Devalaraja S., Jain S., Yadav H. Exotic fruits as therapeutic complements for diabetes, obesity and metabolic syndrome. Food Res Int. 2011;44:1856-1865.
CrossRef - Banos G., Pérez-Torres I., El Hafidi M. Medicinal agents in the metabolic syndrome. Cardiovascular & Hematological Agents. Med Chem. 2008;6:237-252.
CrossRef - Palombo E. A. Traditional medicinal plant extracts and natural products with activity against oral bacteria: potential application in the prevention and treatment of oral diseases. J Evid Based Complementary Altern Med. 2011.
CrossRef - Boutennoun H., Boussouf L., Rawashdeh A., Al-Qaoud K., Abdelhafez S., Kebieche M. In vitro cytotoxic and antioxidant activities of phenolic components of Algerian Achillea odorata leaves. Arab J Chem. 2017;10:403-409.
CrossRef - Ciulei I. Methodology for Analysis of Vegetable Drugs. Practical Manual on the Industrial Utilisation of Medicinal and Aromatic Plants Center Building. Romania. 1982;1-82.
- Aaby K., Ekeberg D., Skrede G. Characterization of phenolic compounds in strawberry (Fragaria×ananassa) fruits by different HPLC detectors and contribution of individual compounds to total antioxidant capacity. J Agric Food Chem. 2007;55:4395-4406.
CrossRef - Abarca-Vargas R., Peña Malacara C. F., Petricevich V. L. Characterization of Chemical Compounds with Antioxidant and Cytotoxic Activities in Bougainvillea x buttiana Holttum and Standl,(var. Rose) Extracts. Antioxidants. 2016;51.
- Maestri D. M., Nepote V., Lamarque A. L., Zygadlo J. A. Natural products as antioxidants. Phytochem: Adv Res. 2006;37:105-135.







