Manuscript accepted on :24-Nov-2018
Published online on: 03-12-2018
Plagiarism Check: Yes
Reviewed by: MD. Sarwar Hossain
Second Review by: Muhammad Shahzad Aslamb
Final Approval by: Dr. Ayush Dogra
Puneet Sudan1  and Jitender Singh2
and Jitender Singh2
1Chandigarh College of Pharmacy-Landran (Mohali)-Punjab, India.
2UIPS, Chandigarh University-Gharuan (Mohali)-Punjab, India.
Corresponding Author E-mail: cgc.ccp.ps@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/1599
Abstract
The aqueous extracts of Azadirachta indica, Trigonella foenum graecum, Allium sativum, Cinnamomum tamala, Capsicum annum were used to test their antifungal properties against the keratinophilic fungus Microsporum gypseum. Plant aqueous extracts were taken at two different concentrations of 5% and 10%. The present study revealed that Allium sativum and Capsicum annum at 10% conc. had more potent antifungal potential as compared to all the other extracts followed by Azadirachta indica, Trigonella foenum graecum and Cinnamomum tamala.
Keywords
Azadirachta Indica; Allium Sativum; Antifungal; Capsicum Annum; Cinnamomum Tamala; Trigonella Foenum Graecum
Download this article as:| Copy the following to cite this article: Sudan P, Singh J. Antifungal Potential of Aqueous Extract of Some Traditional Plants on Microsporum Gypseum. Biomed Pharmacol J 2018;11(4). |
| Copy the following to cite this URL: Sudan P, Singh J. Antifungal Potential of Aqueous Extract of Some Traditional Plants on Microsporum Gypseum. Biomed Pharmacol J 2018;11(4). Available from: http://biomedpharmajournal.org/?p=24523 |
Introduction
Plants are considered to be vital and potent source of phytochemical such as phenols, flavonoids, and essential oils etc which usually act as antimicrobials. From last few decades we have witnessed the development of many different synthetic drugs; however, due to the increase in the bacterial resistance towards the existing chemotherapeutics and many side effects, attention has been drawn towards the search of new and potent antimicrobials of herbal origin.1 The main advantage of using plant extracts is that, the crude extracts contain a mixture of compounds viz. phenols, acids, esters, aldehydes etc., for which it is difficult to develop resistance by bacteria as compared to the synthetic antibiotics that contain a single compound.2 The WHO has taken an initiative towards the exploration and development of plant based health care products and making them available to maximum population for their betterment.3 In the past several attempts were made to screen various plants and their active chemical constituents for their antifungal activity against many pathogenic fungi. Mahadevan reported that several plants contain free formed chemicals, potent phytoconstituents capable of inhibiting the growth and germination of dreadful pathogenic fungi. Therefore, in recent years various researchers have shown concern towards the screening of many herbal plants for their fungi toxic properties. Thus, these herbal plants can be considered as potential candidates for new antifungal drugs and many herbal formulations.4
It has been revealed that water and ethanolic extracts of leaves of Azadirachta indica, Jatropha curcas, Jatropha gossypifolia, Cassia alata, Aloe vera and Anacardium occidentale were assessed in vitro for their efficacy in treating ringworm infections. At the concentration of 2, 5 mg per litre the aqueous extract of aloe vera was found to be effective against T. mentagrophytes and T. rubrum. Cassia alata at the concentration of 5 mg per litre was active against T. mentagrophytes and at 2 mg per litre active against T. rubrum. Jatropha gossypifolia aqueous extract was effective against T. rubrum at 2 mg per litre itself. The ethanolic extract of leaves of Aloe vera, Anacardium occidentale, Azadirachta indica, Jatropha curcas, and Jatropha gossypifolia showed very high activity against T. mentagrophytes and T. rubrum at the concentration of 2,5,10 mg per litre. This is evident from the above that ethanol has the ability to extract the phytochemical.5
Aqueous extracts of Pergularia tomentosa and Mitracarpus scaber at the concentration of 10, 20, 40, 80 and 160 mg/ml were active on Trichophyton mentagrophytes and Trichophyton rubrum. These plants showed activity against Microsporum audouini and Microsporum gypseum only at the concentration of 80mg/ml and 160mg/ml. The chloroform and hexane extract of Pergularia tomentosa showed activity against Trichophyton rubrum, Trichophyton mentagrophytes, and Microsporum gypseum at low concentration of 10mg/ml.6 Aqueous extracts of Allium sativum, Nigella sativa and Lawsonia inermis were also studied for antifungal activity. All the three plants inhibited the growth of dermatophytes. However, the aqueous extract of Allium sativum was found to be most effective.7 Thus the phytocomponents and beneficial uses of plant resources appear to be an indigenous, non-toxic source to control many harmful diseases. At present research on higher plants comes out with promising results and reported that the herbal plant extracts possess antimicrobial activity which helps in controlling many diseases of plants, animals and even humans. In the present study, the effect aqueous extracts at two different concentrations (5% and 10%) of Azadirachta indica, Trigonella foenum graecum, Allium sativum, Cinnamomum tamala, Capsicum annum were used to test their antifungal properties against the keratinophilic fungus Microsporum gypseum.
Material and Methods
In the present study, common herbal plants with easy accessibility like Azadirachta indica, Trigonella foenum graecum, Allium sativum, Cinnamomum tamala, Capsicum annum have been used to evaluate their antifungal potential against the keratinophilic fungus Microsporum gypseum. Plants were obtained from local sources, identified and herbarium sheets are submitted at Pharmacognosy Department, Chandigarh College of Pharmacy-Landran (Mohali).Fungal strain Microsporum gypseum with MTCC no. 2829 was procured from IMTECH Chandigarh. To carry out crude extraction, ten grams of washed, shade dried plant material was crushed with the help of mortar and pestle along with 10 ml of distilled water. The crude material was then filtered through double layered muslin cloth and filter paper. The filtrate thus obtained was used for evaluation of antifungal studies.
Evaluation of Anti Fungal Activity
The effect of plant extracts on mycelial growth was studied in, in-vitro condition on Sabouraud Glucose Agar medium (SGA). The medium supplemented with desired concentrations of plant extracts was poured in petriplates. These petriplates were inoculated with 5mm diameter mycelial disc taken from the margins of 8-10 day old colony raised on SGA. SGA without plant extract served as control. Three replicates of each concentration were maintained. The inoculated plates were incubated at 28±20C for seven days. The diameter of the colony was measured on 3rd, 5th and 7th days.
Results
Aqueous extracts of leaves and bulbs of Azadirachta indica, Trigonella foenum graecum, Allium sativum, Cinnamomum tamala and Capsicum annum at two different concentrations (5% and 10%) were evaluated for their antifungal properties against Microsporum gypseum. The results were recorded on 3rd, 5th and 7th days and are presented in the Tables (1-4) and figures (1, 2).
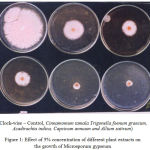 |
Figure 1: Effect of 5% concentration of different plant extracts on the growth of Microsporum gypseum.
|
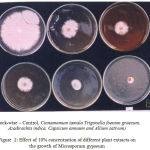 |
Figure 2: Effect of 10% concentration of different plant extracts on the growth of Microsporum gypseum.
|
Table -1 gives an account of the effect of different plant extracts on the growth of Microsporum gypseum recorded on 3rd day. It has been observed that of all the extracts used in the present study, maximum fungal growth inhibition was achieved by the aqueous extract from the bulbs of Allium sativum and no fungal growth was recorded at 10% concentration and a minimum growth was recorded at 5% concentration. This was followed by Capsicum annum, Azadirachta indica, Trigonella foenum graecum and Cinnamomum tamala. It has been observed that with the increase in concentration of the extract the inhibitory effect became more noticeable.
Table 1: Effect of 5% and 10% plant extracts on the growth of Microsporum gypseum in cm (3rd day).
| S No | Extracts | Conc-5% | Conc-10% |
| 1 | Control | 2.86 | 2.86 |
| 2 | Cinnamomum tamala | 2.10 | 1.68 |
| 3 | Trigonella foenum graecum | 1.78 | 1.35 |
| 4 | Azadirachta indica | 1.69 | 1.35 |
| 5 | Capsicum annuum | 1.31 | 0.75 |
| 6 | Allium sativum | 0.65 | – |
The effect of different plant extracts on the growth of Microsporum gypseum noted on 5th day is summarized in the Table -2. As was observed on 3rd day maximum fungal growth inhibition was achieved by the aqueous extract of Allium sativum since a minimum amount of fungal growth was observed at 5% and 10% concentrations. This was followed by Capsicum annum, Azadirachta indica, Trigonella foenum graecum and Cinnamomum tamala. It has been observed that effect of the aqueous extracts of Allium sativum and Capsicum annum on the growth Microsporum gypseum were more or less similar with a little difference.
Table 2: Effect of 5% and 10% plant extracts on the growth of Microsporum gypseum in cm (5th day).
| S No | Extracts | Conc-5% | Conc-10% |
| 1 | Control | 4.2 | 4.2 |
| 2 | Cinnamomum tamala | 3.0 | 2.2 |
| 3 | Trigonella foenum graecum | 2.5 | 1.9 |
| 4 | Azadirachta indica | 2.2 | 1.84 |
| 5 | Capsicum annuum | 1.6 | 1.10 |
| 6 | Allium sativum | 1.1 | 0.71 |
Table – 3 gives an account of the effect of different plant extracts on the growth of Microsporum gypseum recorded on 7th day. The results obtained were similar to those observed on 3rd and 5th days. The effect of aqueous extracts of Allium sativum and Capsicum annum was more pronounced of all the extracts followed by Azadirachta indica, Trigonella foenum graecum and Cinnamomum tamala.
Table 3: Effect of 5 % and 10% plant extracts on the growth of Microsporum gypseum in cm (7th day).
| S No | Extracts | Conc-5% | Conc-10% |
| 1 | Control | 3.5 | 3.5 |
| 2 | Cinnamomum tamala | 2.75 | 1.9 |
| 3 | Trigonella foenum graecum | 2.45 | 1.69 |
| 4 | Azadirachta indica | 1.80 | 1.6 |
| 5 | Capsicum annuum | 1.40 | 0.9 |
| 6 | Allium sativum | 1.76 | 0.61 |
Discussion
The present study revealed that amongst the five extracts tested, the extract of Allium sativum was found to be most effective almost completely checking the mycelial growth at 10% concentration showing 83.09% inhibition, followed by Capsicum annum, Azadirachta indica, Trigonella foenum graecum and Cinnamomum tamala. The results revealed that all the plant extracts were inhibitory to the mycelial growth. As the concentration of extracts increased in the medium, maximum growth inhibition of the test fungus was recorded.
It was reported that Allium sativum contains allyl compounds. Allicin (diallyl disulphide) is the active principle of Allium sativum. Besides allicin, other active compounds reported in Allium sativum are allisatin I, allisatin II, garlicim and garlic phytoncide etc.8
It has been proved that minimal inhibitory concentration of Allium sativum on Microsporum gypseum, M. canis, Trichophyton mentagrophytes and Trichophyton rubrum, and reported varying degree of reaction of these extracts towards different organisms.9
It has been studied that the effect of raw material, from Neem tree, neem oil and neem leaves extract on fungi pathogenic to man. Dried plant parts of neem aqueous extracts and eluotropic solvent were tested in agar diffusion test. Different inhibitory effects on Trichophyton rubrum, T. violaceum, T. mentagrophytes, Epidermophyton floccosum, Microsporum canis, Candida albicans, Fusarium spp. and Scopulariopsis brevicaulis was observed.10 The methanol and ethanolic extract of Azadirachta indica against Candida albicans, Aspergillus niger Aspergillus fumigates and Microsporum gypseum was found growth inhibitory, as the zone of inhibition were observed.11 Ethanolic extract of capsicum annum was proved to be potent antifungal drug against Listeria monocytogenes, Escherichia coli, Candida albicans, Cryptococcus neoformans, Aspergillus flavus.12 Acetone extract of capsicum annum was proved to be potent antifungal drug against Staphylococcus aureus, Klebsiella pneumonia, Streptococcus pyrogens, E.coli, Salmonella typhi, Aspergillus flavus, Candida albicans.13 It also has been observed in this study suggests that the methanol extract of Trigonella foenum L. revealed a significant scope to develop a novel broad spectrum of antibacterial herbal formulation.14 Furthermore, the fungi toxicity of the extract remained unchanged even at high temperature, on autoclaving and during storage. This clearly indicates that herbal plants are untapped vital reservoirs of various valuable chemicals. These antipathogenic, antimicrobial chemicals are widely distributed in higher plants belonging to diverse families, genera and species. These may be distributed throughout the plant or may be in certain parts of a plant or in its special tissues. A wide range of plants are still unexplored for their antimicrobial activity, medicine and agriculture. It needs to be demonstrated that the plants with strong antifungal activity may be effectively and beneficially exploited in the control of keratinophilic infections. Hence, the present study becomes important aspect as the plant extracts employed in the present study have exhibited potent antifungal properties.
Acknowledgments
The author(s) received no specific funding for this work.
Conflict of Interests
There is no conflict of interest.
Funding Source
This research was supported by Shri Guru Ram Dass Educational Society-Mohali (Punjab).The practical work was carried out at Chandigarh College of Pharmacy-Landran, Mohali-Punjab.
References
- Gislene G. F. N., Juliana L., Paulo C. F and Giuliana L. S. Antibacterial activity of plant extracts and phytochemical on antibiotic resistant bacteria. Brazilian Journal of Microbiology. 2000;31: 247–256.
- Kiranmayee R., Bhuvaneswari C.,Lakshmi M. N and Archana G. Antibacterial activity of Alpinia galanga (L) Willd crude extracts. Appl. Biochem. Biotechnol. 2010;162:871–884.
CrossRef - Goud P. S. P., Murthy K. S. R., Pillaiah T and Babu G. V. A. K. Screening for antimicrobial and antifungal activity of some medicinal plants of Nallamala in Andhra Pradesh. India. J Econ Taxon Bot. 2005;29:704–708.
- Mahadevan A.,Agarwal G. P and Bilgrami K. S. (eds) Recent studies in the disease resistance of plants. In: Physiology of Parasitism Symposium. Today and Tomorrows Printers and publishers, New Delhi. 1979;78.
- Adejumo T. O and Bamidel B. S. Control of dermatophyte causing agen (Trichophyton mentagrophytes and Trichophyton rubrum) using six medicinal plants. Journal of medicinal plants research. 2009;3(II):906-13.
- Shinkafi S. A., Mange S. B. Isolation of dermatophytes and screening of selected medicinal plants used in the treatment of dermatophytoses. International research journal of microbiology. 2011; 2(1):040-048.
- Kader H. A. A., Seddek S. R and A. A., El-Shanawany A. A. Invitro study of the effect of some medicinal plants on the growth of some dermatophytes Assiut veterinary. Medical Journal. 1995;34:36-42.
- Cavallito V. J., Buck J. S and Luter C. M. Allicin the antibacterial principle of Allium sativum II and determination of chemical structure. J. Amer.Chem.Soc. 1974;66:1940.
- Guevara B. Q., Solevilla R. C., Mantaring N. M., Remulla R. A and Santos P.C. Antifungal properties of Allium sativum grown in Phillipines. Acta Manilana. 1983;22:1-14.
- Khan M., Wassilew S. W and Schumutterer H. Natural pesticides from the neem tree (Azadirachta indica A. Juss) and other tropical plants In: Ascher K. R. S. (ed.) Proceedings of the 3rd international Neem Conference, Nairobi, Kenya. 1986;645- 650.
- Patil R. C., Kulkarni C. P and Pandey A. Antifungal and phytochemical properties of Tinospora cordifolia, Azadirachta indica and Ocimum sanctum leaves extract. Journal of Medicinal Plants Studies. 2017;5(5):23-26.
- Anikwe L. U., Onoja U. S., Onyeke C. C., Nweze E. I. Antimicrobial activities of four varieties of Capsicum annuum fruits cultivated in Southeast Nigeria against multidrug-resistant and susceptible organisms. Journal of basic Pharmacology and Toxicology. 2017;1(2):21-26.
- Gayathri N., Gopalakrishnan M and Sekar T. Phytochemical screening and antimicrobial activity of Capsicum chinense Jacq. International Journal of Advances in Pharmaceutics. 2016;5(1):12-20.
- Kumari S. O., Rao B. N and Gajula G. R. Phytochemical analysis and anti-microbial activity of Trigonella foenum gracum (Methi seeds). Journal of Medicinal Plants Studies. 2016;4(4):278-281.







