Manuscript accepted on :07-July-2018
Published online on: 03-08-2018
Plagiarism Check: Yes
Reviewed by: Manuel Valdebran
Second Review by: Iman Tavassoly
Final Approval by: Mohamed Abdel-Daim
Tava Shelan Nagapan, Ahmad Rohi Ghazali, Dayang Fredalina Basri and Wenna Nallance Lim
and Wenna Nallance Lim
Biomedical Science Programme, School of Diagnostic and Applied Health Sciences, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, 50300 Kuala Lumpur, Malaysia.
Corresponding Author E-mail: rohi@ukm.edu.my
DOI : https://dx.doi.org/10.13005/bpj/1481
Abstract
Ultraviolet radiation (UVR) from sunlight is an environmental human carcinogen. Skin exposure to UVR would increase the oxidative stress, deoxyribonucleic acid (DNA) damage, melanogenesis and photocarcinogenesis. Therefore, development of photoprotective agent is necessary in order to reduce the cutaneous toxicity. The use natural active compounds like stilbenes and its derivatives have gained attention as photoprotection to skin due to its broad biological activities such as antioxidant, anti-inflammatory, anti melanogenesis and chemoprevention. This review article aims to analyse the existing literature on the photoprotective effect of stilbenes and its derivatives which include the resveratrol, pterostilbene, piceatannol and oxyresveratrol on in vitro and in vivo studies. This article describes the stilbenes and its derivatives protect and prevent UVR induced skin disorders via the reduction of oxidative stress, alleviation of DNA damage, inhibition of melanogenesis and anti photocarcinogenic effect.
Keywords
Chemoprevention; DNA Damage; Melanogenesis; Oxidative Stress; Stilbenes
Download this article as:| Copy the following to cite this article: Nagapan T. S, Ghazali A. R, Basri D. F, Lim W. N. Photoprotective Effect of Stilbenes and its Derivatives Against Ultraviolet Radiation-Induced Skin Disorders. Biomed Pharmacol J 2018;11(3). |
| Copy the following to cite this URL: Nagapan T. S, Ghazali A. R, Basri D. F, Lim W. N. Photoprotective Effect of Stilbenes and its Derivatives Against Ultraviolet Radiation-Induced Skin Disorders. Biomed Pharmacol J 2018;11(3). Available from: http://biomedpharmajournal.org/?p=21824 |
Introduction
The sunlight emits electromagnetic radiation that can be divided into three parts of the wavelength including ultraviolet (UV), visible light and infrared light. This ultraviolet radiation is a carcinogen to humans that involved in melanoma and non-melanoma skin cancer.1 UV radiation can be divided into three categories based on its wavelength which include ultraviolet A (UVA), ultraviolet B (UVB) and ultraviolet C (UVC). UVA (320 nm-400 nm) is capable of penetrating the epidermis and dermis of the human skin. Exposure to UVA has been shown to have a vital part in melanogenesis activity mostly by inducing the oxidative stress and impairment of antioxidant defense in melanocytes as well as melanoma cells.2 Besides, it also can indirectly induce DNA damage by forming reactive oxygen species (ROS).3 UVB can penetrate the epidermis of the skin. It is known to be more cytotoxic and genotoxic as compared to UVA. Prolong exposure to UVB may lead to photocarcinogenesis as well.4 Meanwhile UVC is fully absorbed by the stratosphere layer and blocked from reaching the earth surface. Thus, it is important to develop an appropriate photoprotective agent to protect and prevent the human skin from both UVA and UVB radiation harms.
Research studies on in vivo and in vitro showed that components from stilbenoid group proven to have a strong antioxidant defense and chemopreventive properties. Stilbenes can be regarded as 1,2-diphenylethylenes.5-7 They are class of plant polyphenols and have been proven that it has broad biological activities.8 The major component of natural stilbene are pterostilbene, resveratrol, piceatannol and oxyresveratrol. The research advances in anti- melanogenesis, antioxidant and anti-carcinogenic effect of natural stilbenes against UVR irradiation were reviewed in the present paper.
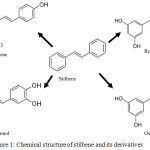 |
Figure 1: Chemical structure of stilbene and its derivatives.
|
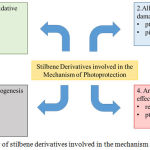 |
Figure 2: Overview of stilbene derivatives involved in the mechanism of photoprotection.
|
Reduction of Oxidative Stress
The skin has a strong antioxidant defense mechanism to prevent and protect the UV-induced oxidative stress. The oxidative stress induced by the ultraviolet radiation will be neutralised by the endogenous antioxidants such as superoxide dismutase (SOD), catalase and other molecules like glutathione (GSH) that reduce the formation of reactive oxygen species prior to the production of oxidative changes in tissue.9 The UVA and UVB has the ability to produce the superoxide ion through the activation of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and respiratory chain reaction. SOD catalyse dismutation of superoxide ion into oxygen molecule and hydrogen peroxide. Meanwhile, catalase breaks down the hydrogen peroxide into oxygen molecule and water molecule. The combination of SOD and catalase completely scavenges superoxide ion initiating ROS. However, prolong UV exposure can halt the capacity of cutaneous antioxidant that would lead to oxidative damage to the tissue. Hence, this would eventually results in skin disorders, photoaging and skin cancers.10
Resveratrol
Treatment of resveratrol on UVA induced human keratinocytes HaCaT cells are potentially protect from oxidative damage. Therefore, resveratrol able to increase the activity of antioxidant enzymes in which glutathione S-transferase (GST) has increased 1.4, 1.7, and 1.5 times in the UVA-irradiated group, treated with resveratrol before and after the irradiated groups respectively. Besides, resveratrol also increase the SOD level in treatment groups as compared to control group. This showed that resveratrol significantly improves the activity of antioxidant enzymes in the preventive groups.11 Besides, resveratrol also increased the antioxidant levels such as SOD and GSH-Px and reduced lipid peroxidation activity significantly in dose dependent manner. In another study, resveratrol attenuates the generation of reactive oxygen species in UVB irradiated human keratinocytes HaCat cells in a dose dependent manner. Furthermore, this study clearly showed that resveratrol partially inhibit the UVB induced cell death by decreasing the ROS generation and inhibition of caspase pathways.12 In vivo studies showed that pretreatment of SKH-1 hairless mice skin with resveratrol significantly inhibited the increased lipid peroxidation.13
Table 1: Summary of stilbene derivatives and its mechanism of photoprotection.
| Derivatives | Model/ cell line | Mechanism | References |
| Resveratrol | HaCat cell line
SKH hairless mice Guinea pig B16F10 melanoma cells Melan-A cells
HaCat cells SKH hairless mice p53+/-/SKH-1 mice A431 SCC cell line |
Reduction of oxidative stress
· ↑ GST, SOD · ↑ SOD, GSH-Px; ↓ lipid peroxidation · ↓ lipid peroxidation Inhibition of melanogenesis · ↓pigment index; ↓ tyrosinase, TRP-1, TRP-2, MITF protein expression · ↓ melanin level · ↓autophagy, thus ↓ pigmentation · ↓ melanoma cells proliferation; ↑ tyrosinase protein expression Anti-photocarcinogenic effect · ↓ ERK, p53, p38 activation; ↑ Bax/Bcl ratio · ↓ AKT, S6 activation in mTOR pathway · ↑ Beclin1 activation in apoptosis pathway · ↓ PCNA, MAPK; ↓cellular proliferation · ↓ cdk-2, cdk-4, cdk-6, cyclin D1, cyclin D2; ↓ cell cycle · ↑ E- cadherin · ↓ Tumor incidence · ↓ Survivin at protein & mRNA level, ↑ Smac/DIABLO · ↓ TGF- |
[10] [11] [12] [26] [26] [29] [42] [43] [44] [44] |
| Pterostilbene | HaCat cell line
HaCat cell line SKH hairless mice B16F10 melanoma cells SKH-1 hairless mice |
Reduction of oxidative stress
· ↓ intracellular ROS Alleviation of DNA damage · ↓ DNA comet tail · ↓ 8-OH-dG Inhibition of melanogenesis · ↓ tyrosinase Anti-photocarcinogenic effect · ↓ tumorigenesis |
[13] [13][14] [34] [14] |
| Piceatannol | HaCat cell line
NHEK cell line |
Reduction of oxidative stress
· ↑ GSH; ↓ intracellular ROS Alleviation of DNA damage · ↓ CPD production Inhibition of melanogenesis · ↓ tyrosinase, melanin level |
[15] [23] [35] |
| Oxyresveratrol | Murine melanoma B-16 cell line
Guinea pig |
Inhibition of melanogenesis
· ↓ tyrosinase · ↓ melanin index, tyrosinase, TRP-1, MITF |
[36] [38] |
Symbols: ↑ increase, ↓ decrease. Abbreviations: GSH glutathione; SOD superoxide dismutase; GSH-Px Glutathione peroxidase; TRP-1 tyrosinase related protein 1; TRP-2 tyrosinase related protein 2; MITF Microphthalmia-associated transcription factor; ERK extracellular-signal-regulated kinase; AKT Protein Kinase B; PCNA Proliferating cell nuclear antigen; MAPK Mitogen-activated protein kinase; cdk cyclin-dependent kinase; TGF-ꞵ Transforming growth factor beta; 8-OH-dG 8-hydroxy-2′ –deoxyguanosine; DNA Deoxyribonucleic acid; ROS reactive oxygen species; CPD Cyclobutane pyrimidine dimers.
Pterostilbene
Li and co-researchers discovered that pterostilbene able to reduce the intracellular ROS in human keratinocytes HaCat cells induced with UVB irradiation.14 These findings showed that pterostilbene decreased the oxidative damage by activating the endogenous antioxidant enzymes.15 In addition, upregulation of the antioxidant enzymes which include catalase, glutamate-cysteine ligase catalytic subunit, glutamate-cysteine ligase, modifier subunit, glutathione reductase, heme oxygenase 1, and NAD(P)H:quinone oxidoreductase has been confirmed that these target genes was induced by the activation of Nrf by pterostilbene through Phosphoinositide 3-kinase (PI3K) pathway.14
Piceatannol
Piceatannol is a phenolic stilbenoid which known to have a broad spectrum of biological activities.16 Maruki-Uchida and co-researchers proved that piceatannol has the ability to decrease the intracellular ROS level in a dose dependent manner in UVB irradiated human keratinocytes HaCat cells. The ROS level in the irradiated keratinocytes was decreased by 13% at 0.5µg/mL, 21% at 1µg/mL and 58% at 2µg/mL compared to the untreated cells. In addition to support this findings, piceatannol also increased the GSH level by 13% at 0.25µg/mL, 22% at 1µg/mL and 63% at 2µg/mL in human keratinocytes induced with UVB irradiation as compared to untreated cells.17
Alleviation of Dna Damage
The ultraviolet radiation that reaches the earth surface is comprised of approximately 95 % UVA and 5 % UVB. The ultraviolet radiation is an environmental human carcinogen.18 DNA damage induced by the UVR can be categorize into direct and indirect damage to the skin. Direct absorption of UV-B photons induces the formation of of three major classes of DNA lesion which include the cyclobutane pyrimidine dimers (CPDs), pyrimidine 6-4 pyrimidone photoproducts (6-4PPs) and the Dewar isomer.19 As in detail, direct DNA damage causes the cycloaddition between C5-C6 of two adjacent pyrimidine bases and changes them into CPD. Meanwhile, covalent bond formation of C6-C4 of two adjacent pyrimidine bases generates 6-4 PPs photoproduct, which further converts to its Dewar valence isomer upon UV excitation at 314 nm.20 Indirect DNA damage occurs when the photons from UVA release electron and energy transfer from cellular photosensitizer to oxygen molecules creating radical oxygen anion. This would eventually induces the oxidation of guanine moiety that would lead to structural rearrangement and 8-oxo-7, 8-dihyroguanine (8-oxo-G) and 8-oxo-7,8-dihydro-2’-deoxyguanosine (8-oxo-dG) formation. These alteration resulting in substitution or transition mutations in the epidermal cells that can lead to skin carcinogenesis.21 These components are the biochemical markers of UVA induced DNA damage. However, UVB is known to be the most mutagenic and genotoxic as compared to UVA2. This is because UVB being absorbed by the DNA efficiently and induce significant damage at lower doses.22,23
Pterostilbene
Pretreatment of pterostilbene to human keratinocyte HaCat cells after UVB treatment significantly reduced UVB-induced DNA comet tail as compared to the UV irradiated control using comet assay method. In addition, reduced DNA comet tail indicate decrease of DNA damage.14,23 Another study showed that pterostilbene also reduced the indirect DNA damage which can be identified using analysis of modified DNA base 8-hydroxy- 2-deoxyguanosine (8-OH-dG). Pterostilbene decreased the chronic UVB induced DNA damage in SKH female hairless mice and human keratinocyte HaCat cells as well.15
Piceatannol
Piceatannol has the ability to remove CPDs from DNA in UVB-irradiated normal human epidermal keratinocyte (NHEK). Exposure of NHEKs to 80 mJ/cm2 irradiation induced CPD formation immediately and this was used as a reference for DNA damage. CPD levels were measured after UVB exposure to determine DNA repair in irradiated cultures. Piceatannol at concentration of 2.0 µg/mL decreased CPD production by approximately 30% in UVB-exposed NHEKs compared with non-treated control cells. Therefore, DNA repair activity of UVB-damaged DNA in NHEKs might be mediated by piceatannol. Therefore, piceatannol has the potential to give protection against UVB-irradiated NHEK death and sun-damage in UVB-irradiated NHEK by enhancing pre-existing DNA repair mechanisms.25
Inhibition of Melanogenesis
Melanogenesis is in fact good for skin as it can help in protecting skin from UV exposure which can cause problems to skin for instance skin cancer, wrinkles, erythema and many more skin diseases by creating unwanted mechanisms in skin for example generation of reactive oxygen species (ROS) that commonly known as one of roots to carcinogenesis.26 Ironically, although melanin formation is good as it can be the best natural sun protection for human skin ever have, but the overproduction of melanin can cause many unwanted effects that can be appeared clearly on skin or can become a disease for chronic UV exposure that need serious medical intervention like skin cancer.27,28
Resveratrol
Tyrosinase becomes the main attention in melanogenesis related studies as tyrosinase is the key enzyme in regulating melanogenesis.29 More findings reported that resveratrol inhibit melanogenesis activity via different mechanisms. It is reported that biotransformed resveratrol by tyrosinase can inhibit the dopa oxidase activity of tyrosinase and able to become a competitive substrate to tyrosine and L-DOPA where these two substrates are the main substrates in human skin melanogenesis and recent in vivo study reported that resveratrol had decreased the expression of other regulator proteins in melanogenesis which are tyrosinase-related protein-1 (TRP-1), tyrosinase-related protein-2 (TRP-2) and Microphthalmia-associated transcription factor (MITF).27,28 The following study also reported that oxidised resveratrol by tyrosinase inhibit the tyrosinase activity itself via kcat type mechanism.30
Autophagy is a biological stress response in cells in which it removes waste proteins and organelles from the cytoplasm and also recycling their content via lysosomal pathway.31 Recent studies proven that autophagy involved in the biogenesis of melanin and degradation of the melanosome, able to reduce the production of melanin pigments.32 Hence, this destruction process to cells in the body also is one of anti-melanogenesis mechanism by resveratrol. Melan-A cells was induced with resveratrol for different doses for its effects on autophagy activity and it showed that resveratrol can strongly induce autophagy in melanocytes. In addition, autophagy is reported can regulate the effect of resveratrol on melanogenesis activity.33 Melanogenesis in melanocytes also can be regulated by α-melanocyte-stimulating hormone (α-MSH).34 Melanin level is reported low significantly by 28.2 ± 1.9% in in vitro study when B16F10 cells with stimulated α-MSH treated with resveratrol at 10µM together with no cytotoxicity found after 48 hours.28 Another similar study reported that when α-MSH stimulated B16 melanoma cells treated with resveratrol, the viability of melanoma cells as well as the cells invasion were significantly reduced. Signalling of α-MSH on B16 melanoma cells also reduced significantly by resveratrol.34 Microphthalmia-associated transcription factor (MITF) plays an important role in tyrosinase, tyrosinase-related protein-1 and tyrosinase-related protein-2 expression and these are the main enzymes in melanogenesis.34 Expression of MITF can be downregulated by resveratrol as what reported by Lee and co researchers.28 Contrary to the all previous findings above, recent study reported that tyrosinase activity increased to 1 up to 7 times by using concentration of resveratrol from 1 to 15 µM and this shows that resveratrol can promote melanogenesis in HT-144 melanoma cells instead of inhibiting it via MER/ERK pathway.37 Further assessments need to be done to clarify the role of resveratrol in regulating the melanogenesis.
Pterostilbene
Pterostilbene, a potential antimelanogenesis agent had been only reported in recent studies might be able to exert stronger antimelanogenesis activity than resveratrol’s, its natural analog. Melanogenesis was suppressed by 63% and intracellular tyrosinase activity was inhibited indirectly by 58% on B16F10 melanoma cells when treated with 10 µM of pterostilbene in much greater effects compared to resveratrol, resveratrol trimethyl ether and arbutin.38
Piceatannol
Another potent bioactive stilbene, piceatannol had been reported in study that with piceatannol doses 5 µM, 10 µM, 50 µM and 100 µM can reduce melanin level in α-MSH treated B16 cells from 77% down to 15 %. Activity of tyrosinase enzyme also measured with same doses of piceatannol indicates that tyrosinase activity was inhibited from 142% down to 41%. In addition, piceatannol was reported to exert a stronger inhibitory effect against tyrosinase with IC50 value of 1.5 µM compared to resveratrol with the value IC50 63.2 µM.39
Oxyresveratrol
Oxyresveratrol, a stilbenoid compound also had been studied in few studies and reported to have many therapeutic properties as effective as resveratrol and pterostilbene. Morus alba L. also known as white mulberry is one of richest source of oxyresveratrol and had been studied for commercial use.40 Oxyresveratrol was reported to have a potent suppressive effect when tested against dopa oxidase activity of mushroom tyrosinase in a dose-dependent manner. Inhibition on enzyme activity was exhibited from 25% to 84% with the concentration of oxyresveratrol from 0.3 µM to 5 µM and when the concentration of oxyresveratrol was used more than 10 µM, the inhibition on enzyme activity was more than 90%.39 Another similar study evaluated the suppressive effect of oxyresveratrol against mushroom tyrosinase activity with the concentration from 1 µM to 100 µM exhibited the inhibition effects from 31% to 97%. In addition, oxyresveratrol did not change the Km value of enzyme but reduced the Vmax value of mushroom tyrosinase activity (data not shown) and reported as a non competitive inhibitor of the mushroom tyrosinase enzyme.41 Topical application of oxyresveratrol on guinea pig skin can reduce the hyperpigmentation effect caused by UVB irradiation with index 572 with 2% treatment compared to other used which were compounds arbutin with index 719, mulberroside A with index 694 and oxyresveratrol-3-o-glucoside with index 585 whereas 5% treatment of oxyresveratrol exhibit index of melanin for 521 compared to other used compounds arbutin with index 684, mulberroside A with index 655 and oxyresveratrol-3-o-glucoside with index 570. Melanin synthesis inhibition by oxyresveratrol were also greater compared to arbutin, mulberroside A and oxyresveratrol-3-o-glucoside. In the same study, oxyresveratrol can inhibit the expression of tyrosinase, tyrosinase-related protein-1 and microphthalmia-associated transcription factor (MITF) for 10% greater than other compounds.42 However, in treated B16F10 cells with α-MSH at concentration 3 and 10 µM, oxyresveratrol could not inhibit melanin synthesis as effective as resveratrol and triacetyl resveratrol. Thus, human epidermal melanocytes stimulated with L-tyrosine was used to further evaluate the antimelanogenic activity of oxyresveratrol. Protein suppression of tyrosinase enzyme can be suppressed by oxyresveratrol but not as effective as resveratrol and triacetyl resveratrol.41 Recent in vitro study reported that low concentration of oxyresveratrol than 20 µM able to inhibit melanin synthesis potently on B16 cells whereas on melan-a cells, concentration used between 20 to 100 µM did not give significant difference in melanin content.44
Based on above studies, oxyresveratrol has a potential to be an antimelanogenic agent. To the best of our knowledge, data of oxyresveratrol based on in vivo study is much lesser than in vitro studies and thus, the anti-melanogenic activity of oxyresveratrol should be further explored be it in vivo study or in vitro study to evaluate the effectiveness of oxyresveratrol in exerting its anti-melanogenic activity in all aspects.
Anti-Photocarcinogenic Effect
Carcinogenesis induced by the ultraviolet radiation is a multistage process which include the initiation, promotion and progression that would lead to the mutation resulting in activation of oncogenes or silencing of tumor suppressor gene. Thus, disturbing the normal growth behaviour of keratinocytes and melanocytes In addition, it is also depends on the applied dosage applied.45
Resveratrol
Vitale and co researchers reported that 100 mM resveratrol extensively reduced Extracellular Signal-regulated Kinase (ERK) activation after 24 hour pretreatment. In addition, both 2 and 24 hours of resveratrol pretreatment with concentrations 25 mM and 100 mM have induced p38 and p53 activation above UVB induced levels. Furthermore, Bax/Bcl2 ratio essentially increased in cells pretreated for 24 hours with 25 mM resveratrol prior UVB irradiation. These findings showed that resveratrol has the protective effect in response to UVB irradiation towards the early activation of apoptosis triggers such as ERK, p38, p53, and Bax/Bcl2 ratio. On the other hand, both concentrations and times of pretreatment of resveratrol which are 25 mM, 100 mM and 2 hours and 24 hours respectively reduced UVB-induced phosphorylation of the two proteins which include AKT and S6 that in involved in mTOR pathway. This suggest that resveratrol able to trigger apoptosis and also autophagy as well. Vitale and co researchers confirmed that resveratrol increased beclin-1 expression as well at both concentrations of 25 mM, 100 mM. Expression of Beclin1 which is an essential autophagy effector with an important role in apoptosis pathway. Imaging autophagosomes in vivo by using the fluorescent dye monodansylcadaverine (MDC) was done to further confirm that resveratrol pretreatment does not prevent, but enhances the formation of autophagosomes that would lead to autophagy activation. All these findings indicate that resveratrol enhances UVB-induced HaCaT cells death via both apoptosis and autophagy which is essential to develop this polyphenol as photochemopreventive agent.46
Topical application of resveratrol (10mmol/0.2 ml acetone/mouse) on dorsal skin significantly inhibited the UVB-mediated increase in proliferating cell nuclear antigen (PCNA) protein levels. PCNA is responsible markers for cellular proliferation. Furthermore, topical application of resveratrol prior to UVB irradiation also inhibits the Mitogen-activated protein kinase (MAPK) which is responsible for cellular proliferation, Other than that, western blot analysis also showed that downregulation of expression of cdk-2, cdk-4, cdk-6, cyclin D1and cyclin D2 protein levels in epidermis. These findings strongly suggest resveratrol able to inhibits the progression of cell cycle. As a whole, resveratrol has the ability to prevent the UVB induced skin cancer.47
Kim and co researchers have reported that oral administration of resveratrol can slow down the UV induced skin tumorigenesis in highly tumor susceptible p53 SKH-1 hairless mice. In addition, resveratrol treatment reduced the average number of skin tumors by 43% at week 22 as compared to control group. Expression of epithelial cadherin (E-cadherin), a surface receptor, that has an essential role in the formation of adherence junctions and it is often mutated or lost in cancer cells. Skin and squamous carcinoma cells (SCC) excised from the resveratrol-treated mice showed increased immunohistochemical distribution of E-cadherin compared with UV-irradiated and non-treated skin and SCCs. They further discovered that resveratrol downregulates TGF-b2/Smad-dependent and -independent signaling pathways in A431 cells. Resveratrol -mediated suppression of in vitro invasiveness is rescued by Akt-dependent TGF-b2 upregulation. Resveratrol at 50–100 mm diminished the invasiveness of A431 cells by 75–90% (P<0.002) compared with untreated cells.
Resveratrol highly significant inhibition in the tumor incidence as examined by Kaplan-Meier Analysis. In resveratrol-treated group, majority of the lesions were identified to be actinic keratoses with or without inflammation. An appreciable overexpression in the levels of Survivin in skin tumors at protein- and mRNA- levels, whereas the treatment of skin with resveratrol resulted in a downmodulation in protein and mRNA levels of Survivin. Resveratrol may protect the skin from UVB-mediated damages, including skin cancer development via modulating the expression and function of Survivin. Studies have suggested that a direct interaction between Survivin and Smac/DIABLO is essential for the antiapoptotic activity of Survivin. As shown by immunoblot analysis, the data demonstrated that the levels of Smac/DIABLO are significantly down-regulated in UVB-induced tumors, whereas the treatment of skin with resveratrol reversed this response.48
Pterostilbene
Pretreatment of the female SKH-1 hairless mice skin with pterostilbene resulted in an immediate reduction in UVB-induced skin tumorigenesis in which 90 % of tumor free-mice at the end of treatment on the 40th week. This mechanism can be associated with maintenance of skin antioxidant defense and also inhibition of UVB induced oxidative damages.15
Conclusion
In summary, prolong exposure of skin to UVR radiation would cause hyperpigmentation and skin cancer. Natural active compound such as natural stilbene and its derivatives has the potential to develop as photoprotective agent. The present review therefore shows that these stilbene derivatives exert important role in photoprotection mechanism via reduction of oxidative stress, alleviation of DNA damage, inhibition of melanogenesis and photocarcinogenesis. As for the future studies, most of the available literature on the activity of stilbene derivatives especially piceatannol refers to the in vitro properties. It is, therefore, important to apply both in vitro and in vivo or ex vivo studies.
Acknowledgement
This project was funded by Universiti Kebangsaan Malaysia under research code grant (GUP-2016-081).
References
- Mancebo S.E, Wang S.Q. Skin cancer: Role of ultraviolet radiation in carcinogenesis. Reviews on Environmental Health. 2014;29(3):265–273.
CrossRef - Anna B, Blazej Z, Jacqueline G, Andrew C.J, Jeffrey R, Andrzej S. Mechanism of UV-related carcinogenesis and its contribution to nevi/melanoma. Expert Review of Dermatology. 2007;2(4):451–469 .
CrossRef - Fisher G.J, Kang S, Varani J, Bata-Csorgo Z, Wan Y, Datta S, Voorhees J.J. Mechanisms of Photoaging and Chronological Skin Aging. Archives of Dermatology. 2002;138:1462-1470.
CrossRef - Holick M.F. Biological effects of sunlight, ultraviolet radiation, visible light, infrared radiation and Vitamin D for health. Anticancer Research. 2016;36(3):1345– 1356.
- Wolter F, Clausnitzer A, Akoglu B, Stein R. Piceatannol, a Natural Analog of Resveratrol, Inhibits Progression through the S Phase of the Cell Cycle in Colorectal Cancer Cell Lines. The Journal of Nutrition. 2002;132(2):298-302.
CrossRef - Pan M.H, Gao J.H, Lai C.S, Wang Y.J, Chen W.M, Lo C.Y, Wang M, Dushenkov S, Ho C.T. Antitumor Activity of 3,5,40-Trimethoxystilbene in COLO 205 Cells and Xenografts in SCID Mice. Molecular Carcinogenesis. 2008;47:184–196.
CrossRef - Hasiah A.H, Ghazali A.R, Weber J.F.F, Velu S, Thomas N.F,Hussain I.S.H. Cytotoxic and antioxidant effects of methoxylated stilbene analogues on HepG2 hepatoma and Chang liver cells: Implications for structure activity relationship. Human and Experimental Toxicology. 2011;30(2):138–144.
CrossRef - Shen T, Wang X.N, Lou H.X. Natural stilbenes: an overview. Natural Product Reports. 2009;26:916–935.
CrossRef - Moyal D. Prevention of ultraviolet-induced skin pigmentation. Photodermatology, Photoimmunology & Photomedicine. 2004;20:243–247.
CrossRef - Nichols J.A, Katiyar S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, anti-oxidant and DNA repair mechanisms. Archives of Dermatological Research. 2010;302(2):71-83.
CrossRef - Liu Y, Chan F, Sun H, Yan J, Fan D, Zhao D, An J, Zhou D. Resveratrol protects human keratinocytes HaCaT cells from UVA-induced oxidative stress damage by downregulating Keap1 expression. European Journal of Pharmacology. 2011;650:130–137.
CrossRef - Park K, Lee J. Protective effects of resveratrol on UVB-irradiated HaCaT cells through attenuation of the caspase pathway. Oncology Reports. 2008;19:413-417.
CrossRef - Afaq F, Adhami V.M, Ahmad N. Prevention of short-term ultraviolet B radiation-mediated damages by resveratrol in SKH-1 hairless mice. Toxicology and Applied Pharmacology. 2003;186:28–37.
CrossRef - Li H, Jianga N, Liang B, Liu Q, Zhanga E, Penga L, Denga H, Li R, Li Z, Zhu H. Pterostilbene protects against UVB-induced photo-damage through a phosphatidylinositol-3-kinase-dependent Nrf2/ARE pathway in human keratinocyte. Redox Report. 2017;22(6):501-507.
CrossRef - Sirerol J.A, Feddi F, Mena S, Rodriguez M.L, Sirera P, Aupí M, Pérez S. Asensi M, Ortega A, Estrela J.M. Topical treatment with pterostilbene, a natural phytoalexin, effectively protects hairless mice against UVB radiation-induced skin damage and carcinogenesis. Free Radical Biology and Medicine. 2015;85:1–11.
CrossRef - Sato D, Shimizu N, Shimizu Y, Akagi M, Eshita Y, Ozaki S, Nakajima N, Ishihara K, Masuoka N, Hamada H, Shimoda K, Kubota N. 2014. Bioscience, Biotechnology, and Biochemistry. 2014;78(7):1123–1128.
CrossRef - Maruki-Uchida H, Kurita I, Sugiyama K, Sai M, Maed K, Itoa T. The Protective Effects of Piceatannol from Passion Fruit (Passiflora edulis) Seeds in UVB-Irradiated Keratinocytes. Biological and Pharmaceutical Bulletin. 2013;36(5):845–849.
CrossRef - Cadet J, Douk T. Formation of UV-induced DNA damage contributing to skin cancer development. Photochemical & Photobiological Sciences. 2018.
CrossRef - Rastogi R.P, Kumar R.A, Tyagi M.B, Sinha R.P. Molecular Mechanisms of Ultraviolet Radiation-Induced DNA Damage and Repair. Journal of Nucleic Acids. 2010.
CrossRef - Rochette P.J, Therrien J.P, Drouin R, et al. UVA-induced cyclobutane pyrimidine dimers form predominantly at thymine-thymine dipyrimidines and correlate with the mutation spectrum in rodent cells. Nucleic Acids Research. 2003;31(11):2786–2794.
CrossRef - Pérez-Sáncheza A, Barrajón-Catalána E, Herranz-Lópeza M, Castilloc J, Micol V. Lemon balm extract (Melissa officinalis, L.) promotes melanogenesis and prevents UVB-induced oxidative stress and DNA damage in a skin cell model. Journal of Dermatological Science. 2016.
CrossRef - Bennett D.C. Ultraviolet wavebands and melanoma initiation. Pigment Cell & Melanoma Research. 2008;21:520–524.
CrossRef - Ravanat J.L, Douki T, Cadet J. Direct and indirect effects of UV radiation on DNA and its components. Journal of Photochemistry and Photobiology B: Biology. 2001;63:88–102.
CrossRef - Zainol M, Stoute J, Almeida G.M, Rapp A, Bowman K.J, Jones G.D.D. ECVAG. Introducing a true internal standard for the Comet assay to minimize intra- and inter-experiment variability in measures of DNA damage and repair. Nucleic Acids Research. 2009;37(22):e150.
CrossRef - Ikeoka S, Nakahara T, Iwahashi H, Mizushina Y. The Establishment of an Assay to Measure DNA Polymerase-Catalyzed Repair of UVB-Induced DNA Damage in Skin Cells and Screening of DNA Polymerase Enhancers from Medicinal Plants.







