Manuscript accepted on :October 04, 2017
Published online on: --
Aslan Alekseevich Teuvov1 , Arthur Muzkharbievich Baziev2
, Arthur Muzkharbievich Baziev2 , Zarema Nuriydinovna Lovpache1
, Zarema Nuriydinovna Lovpache1 , Irina Sergeevna Teunikova1
, Irina Sergeevna Teunikova1 and Sergey Mikhailovich Chudopal2
and Sergey Mikhailovich Chudopal2
1Department of General Surgery, Medical Faculty, H.M. Berbekov Kabardino-Balkaria State University, Nalchik, Russia.
2Department of Nervous Diseases, Neurology and Psychiatry, Medical Faculty, H.M. Berbekov Kabardino-Balkaria State University, Nalchik, Russia.
Corresponding Author E-mail: ateuvov@bk.ru
DOI : https://dx.doi.org/10.13005/bpj/1306
Abstract
Diabetes mellitus (DM) is often complicated by the development of purulent-necrotic lesions of the lower limbs, leading not only to early disability due to limb amputation, but also shortening the life span. The Diabetic Foot Syndrome (DFS) is a pathological state of feet in diabetes mellitus, which occurs against the background of impaired peripheral nerves, vessels, skin, soft tissues, bones and joints and is accompanied by destructive and purulent necrotic processes. DFS is observed in various forms in 20-80% of patients with diabetes and increases the risk of gangrene of the lower extremities by 20 times. And amputation of the lower limbs is performed to these patients by 15-20 times more often than in people without diabetes. The objective of the study was to evaluate the effectiveness of applying ozonated solutions in the comprehensive treatment of diabetic foot syndrome. Methods of research: all patients are divided into 2 groups. The main clinical trial group included 17 patients with DM, who were admitted in the period from January to December 2016 and had DFS developed (3-5 severity class according to F. Wagner’s classification). Clinical, hematological, bacteriological, cytological studies were performed at admission before the beginning of treatment and on the 1-3-5-7-10th postoperative day. Data from medical records of 20 patients admitted in the same period of 2015 were the subjects of the study as a control group. The control group received traditional treatment. In the main group, traditional treatment was supplemented with sessions of topical and general ozone therapy. The treatment results of 37 patients with diabetes having DFS developed were analyzed; the outcomes and prospects for the development of ozone therapy of this pathology in purulent surgery were evaluated. In the main group a rapid transition from inflammatory-degenerative processes to regenerative ones and fast complete elimination of microbial flora were observed. On the 3rd-5th day, the patients of the main group showed normalization of Erythrocyte Sedimentation Rate (ESR) parameters and total leukocyte count, decrease in the content of stab and segmented leucocytes, increase in the number of lymphocytes and monocytes, a decrease in the number of bed-days as compared to the control group. Our studies have demonstrated that supplementing the comprehensive treatment of patients having acute purulent surgical diseases with intravenous and topical ozone-containing medications exerted a pronounced therapeutic and economic effect.
Keywords
Diabetic Foot Syndrome; Ozone Therapy; Purulent Surgery
Download this article as:| Copy the following to cite this article: Teuvov A. A, Baziev A. M, Lovpache Z. N, Teunikova I. S, Chudopal S. M. Ozone Therapy in the Comprehensive Treatment of Diabetic Foot Syndrome. Biomed Pharmacol J 2017;10(4). |
| Copy the following to cite this URL: Teuvov A. A, Baziev A. M, Lovpache Z. N, Teunikova I. S, Chudopal S. M. Ozone Therapy in the Comprehensive Treatment of Diabetic Foot Syndrome. Biomed Pharmacol J 2017;10(4). Available from: http://biomedpharmajournal.org/?p=17341 |
Introduction
Diabetes mellitus (DM) is often complicated by the development of purulent necrotic lesions of the lower extremities, leading not only to early disability due to limb amputation, but also shortening the life span.1,2
Diabetic Foot Syndrome (DFS) is a pathological state of feet in diabetes mellitus, which occurs against the background of impairment to the peripheral nerves, vessels, skin, soft tissues, bones and joints and is accompanied by destructive and purulent necrotic processes. DFS is observed in various forms in 20-80% of DM patients and increases the risk of gangrene of the lower extremities by 20 times. Amputation of the lower limbs is performed to these patients by 15-20 times more often than in people without diabetes. In most cases, amputations are made because of ischemia and a deep infection or a combination of both.3,4 DFS is more frequent in case of inadequate treatment of diabetes. It occurs in two clinical forms: neuropathic and neuroischemic.5
With the neuropathic DFS the peripheral nervous system is affected with a violation of sensitivity. The main blood flow does not suffer.
With a neuroischemic form, neuropathy is less pronounced, but decompensated, critical ischemia always dominates. It is caused by polylocal occlusions and stenoses mainly of distal arteries. With the development of irreversible ischemia, moist gangrene or uncontrolled dry necrosis is observed.
The oxidative stress is the most important link in the pathogenesis of diabetes; this stress is induced by tissue damage and cell death leading to increased production of free radicals and inhibition of antioxidant protection.4
The objective of the research is to evaluate the effectiveness of applying ozonized solutions in the complex treatment of Diabetic Foot Syndrome.
The tasks of the Research are as follows
To study the effect of ozonized solutions on the course of the nearest postoperative period in patients with Diabetic Foot Syndrome;
To determine the effect of ozonized solutions on the endotoxicosis indices and the level of microbial bodies in smears-prints;
To identify the economic effect of including ozonized solutions in the treatment of patients with DFS
Materials and Methods
Medical supervision involved 37 patients with diabetes, who had DFS developed (of the 3rd-5th DFS severity class according to F. Wagner’s classification6). All patients are divided into 2 groups. The first, control group (n = 20) were patients who received traditional treatment. In the second, main group (n = 17) traditional treatment was supplemented with sessions of local and general ozone therapy.
The patients of both groups matched by sex, age, primary and concomitant diseases and developed complications.
The main group of the clinical trial included patients who were admitted in the period from January to December 2016. Clinical, hematological, bacteriological, cytological studies were performed at admission before the beginning of treatment and on the 1-3-5-7-10th postoperative day. Data from medical records of 20 patients admitted in the same period of 2015 were the subjects of the study as a control group.
The comprehensive treatment was carried out by several specialists (a surgeon, an endocrinologist, and an intensivist) and included conservative therapy and surgical correction.
Conservative therapy began with correction and/or compensation of diabetes mellitus with obligatory transfer to insulin therapy, antibacterial and symptomatic therapy, measures to reduce the critical ischemia manifestations.
The surgical treatment was based on the principle of tissue conservation and maximum preservation of the length of the foot. Wide lancing of purulent necrotic foci, removal of necrotic and purulence-impregnated tissues and adequate drainage were carried out. However, single surgical intervention did not always allow achieving radical surgical treatment, repeated surgical treatment with necrectomy was required.
The volume of surgical interventions, taking into account the severity of DFS, is given in Table 1.
Table 1: Volume of surgical treatment of DFS
| DFS severity class according to F. Wagner
|
Extent of operations |
Total |
|||||||
| Phlegmon lancing | Astragalar amputation | Foot amputation (resection) | Lower extremity amputation | ||||||
| Main group | Control group | Main group | Control group | Main group | Control group | Main group | Control group | ||
| III severity class | 19 | 15 | 1 | 2 | 37 | ||||
| IV severity class | 4 | 5 | 2 | 1 | 12 | ||||
| V severity class | 6 | 5 | 11 | ||||||
| Total | 19 | 15 | 4 | 5 | 2 | 7 | 8 | 60 | |
In the main group, the traditional treatment complex was supplemented by sessions of local and general ozone therapy.
For the general ozone therapy, 200 ml of ozone-saturated 0.9% sodium chloride solution was injected intravenously daily with ozone concentration being 800-1000 mcg/L. The duration of the treatment course depended on the general condition of the patients, the wound process progress, the dynamics of the biochemical, clinical and immunological parameters of the body.
In the postoperative period local ozone therapy included daily dressings of wounds and the introduction of ozonized saline with concentrations of 3.3-4 mg/L in drains.
The timing of the regenerative processes in a purulent wound and the number of microbial bodies in smears-imprints were evaluated as the main outcome of the study.
The endotoxicosis rates and the number of bed-days were evaluated as additional outcomes of the study in the main group as compared to the control one.
The intoxication severity was judged by the parameters of the average molecular weights (AMW) and the leukocytal intoxication index (LII) according to Kalf-Kalif.
Cytological examination of the material was carried out by applying a defatted sterile slide to the wound surface cleaned from the exudate. Wound imprints were fixed with 96% ethyl alcohol and stained by Romanovsky-Giemsa procedure. To obtain a more authentic view of the wound process dynamics, the cellular composition was expressed as a percentage, the quantitative characteristics of the basic cellular elements were reduced to a common cytogram taking into account microflora, leukocytes (lymphocytes, monocytes, macrophages, and neutrophils).
When carrying out hematological studies, the following parameters of peripheral blood were studied: the dynamics of the total leukocyte count, leukogram, ESR.
The statistical analysis was performed using the StatGraphics 5.0 program.
Results
Age and sex composition of patients is given in Table 2.
Table 2: Age and sex characterization of patients
| Age | Main group
(n=17) |
Control group
(n=20) |
Total | ||
| males | females | males | females | ||
| 21-30 | 1 | – | – | – | 1 |
| 31-40 | – | 1 | – | – | 1 |
| 41-50 | 1 | – | 1 | – | 2 |
| 51-60 | 1 | – | 2 | 2 | 5 |
| 61-70 | 3 | 5 | 4 | 2 | 14 |
| > 70 | – | 5 | 3 | 6 | 14 |
| Total | 6 | 11 | 10 | 10 | 37 |
Diabetes mellitus, type II, severe form, decompensation stage was diagnosed in 80% of patients.
According to the form of purulent-necrotic lesion of the limb, our patients were distributed as follows:
Foot phlegmon and abscess (the 3rd severity class according to Wagner) – 23;
Gangrene of one or more toes (the 4th severity class) – 7;
Disseminated foot gangrene, with pronounced intoxication, requiring amputation of the lower limb in an emergency order (the 5th severity class) – 9. of these, one patient has both feet simultaneously affected.
After a traditional surgical intervention (opening and drainage of abscesses, amputations of toes, feet or limbs7,8) and local visual examination of wounds of soft tissues of patients was performed daily during dressings. Before the treatment, purulent discharge, pronounced fibrinous plaque on the wound bottom, edema and hyperemia of the surrounding tissues were noted. In the process of ozone treatment, medicinal manipulations in the wound region did not cause any painful sensations in patients by 3rd-5th day. When analyzing the treatment results, it was found that under the influence of ozone therapy, there was significant reduction in pain, more intensive decrease in swelling and hyperemia of the tissues, early clearance of wounds compared with the control group.
On the first day after the operation the analysis of hematological parameters revealed that the patients of the study groups had high ESR, a leukogram showed high neutrophil count with a shift to the left, and the total lymphocyte count was decreased. On the 3rd-5th day, the patients of the main group showed normalization of ESR parameters and total leukocyte count, decrease in the content of stab and segmented leucocytes, increase in the number of lymphocytes and monocytes, a decrease in the number of bed-days as compared to the control group.
According to our data, St.Aureus (55.8%), St.epidermidis (28.4%), and Ps.aurugenosea (12.2%) were causative agents of the purulent process, and the associations of microorganisms (St.aureus + St.epidermidis) were inoculated in 42.8%. In the main group, against the background of the ozone application, the growth of pathogens was absent in the wounds already on the 5th day.
In the cytograms before the treatment, the cellular elements of the smears were presented predominantly (90%) by neutrophilic leukocytes, mostly with manifested degeneration and rare cases of macrophages (1.4%); fibroblasts were not detected. Morphologically, necrotic detritus was noted.
The dynamics of cytological and bacteriological changes is presented in Table 3.
As Table 3 demonstrates, there was a rapid transition from inflammatory-degenerative processes to regenerative one and fast, complete elimination of microbial flora in the main group, while in the control group the cytograms dynamics and the infestation of wounds by microorganisms testified to the sluggish flow of regeneration processes.
Table 3: Characterization of cytological and bacteriological changes in the wound
| Cellular composition of smears | Main group
(n=17) |
Control group
(n=15) |
||||||
| 1 day | 3 days | 5 days | 7 days | 1 day | 3 days | 5days | 7days | |
| Neutrophils | 90% | 58% | 36% | 20% | 91% | 70% | 62% | 55% |
| Macrophages | 1.4% | 5% | 18% | 32% | 1.2% | 1.2% | 10% | 25% |
| Fibroblasts | – | 3% | 15% | 30% | – | – | 5% | 15% |
| Microorganisms | 107 | 107 | – | – | 107 | 107 | 106 | 105 |
The dynamics of endotoxicosis indices (AMW, LII) in both groups is shown in Figures 1 and 2, which illustrate that faster detoxification occurred under the influence of general and local application of ozone.
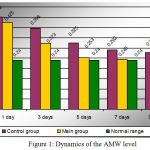 |
Figure 1: Dynamics of the AMW level
|
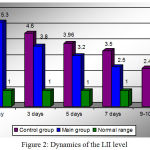 |
Figure 2: Dynamics of the LII level
|
Figure 3 shows the ratio of bed-days in patients of both groups, which indicates that the application of ozone therapy enabled to reduce the number of bed-days by an average of 7 days for the main group of patients.
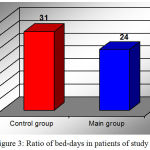 |
Figure 3: Ratio of bed-days in patients of study groups
|
One patient died, whose postoperative period was complicated by the development of pulmonary artery thromboembolia.
Discussion
The clinical picture improved earlier in the main group of patients under the influence of ozone therapy. Restoration of endotoxicosis results and cessation of the wound microflora growth occurred on the 5th day, whereas in the control group the recovery period took 10 days or more.
As is known, medical ozone increases the permeability of cellular membranes for glucose, which is achieved by stimulating the pentose phosphate shunt and aerobic glycolysis which are suppressed in diabetes, and this helps reduce hyperglycemia by better glucose entering the tissue. Glucose is oxidized to the final products, and as a result, the basic function of carbohydrates – the energy function – is restored. The energy hunger of tissues is removed. Catabolic processes are oppressed, LPO is normalized.4,9,10
Another important circumstance is that, under the action of ozone, glucose metabolism in erythrocytes is more active, hemoglobin releases oxygen to tissues better, oxygen supply is increased, and hypoxia is eliminated, which plays almost a key role in the treatment process. Ozone therapy contributes to prevention of the development and progression of processes associated with low entry of glucose into the cell, such as neuropathies, arthropathies, angiopathies.9,10
It is necessary to take into account the immune-modulating effect of ozone, especially for the insulin-dependent form of diabetes, which is characterized by the liability to chronic infections and pustular lesions against the background of suppressed immunity.
The therapy is based on the intravenous administration of ozonized normal saline (ONS) in a volume of 200 ml with ozone concentration at the output of 1000 mg/L, which are conducted every other day in the amount of 10-14 procedures. Infusions can be replaced with rectal ozone introductions according to the same schedule as the ONS, in an amount of 500 ml with the ozone dose of 1000 mcg.
Additional ozone therapy procedures are prescribed depending on the type of diabetes mellitus and the available complications. With the signs of secondary immunodeficiency, minor autohemotherapy (MiAHT) with ozone is performed in 6-8 sessions 2-3 times a week; with diabetes mellitus of the second type, ozone reflexotherapy is additionally used according to the common formula.10
In this paper, the authors investigated the persistent characteristics of strains isolated in the patients’ wound culture. The purulent process was caused by St.aureus (55.8%), St.epidermidis (28.4%), and Ps.aeruginosa (12.2%), and associations of microorganisms (St.aureus + St.epidermidis) were revealed in 42.8% of wound culture. The main group showed a rapid transition from inflammatory-degenerative processes to regenerative ones and fast, complete elimination of microbial flora. On the 3rd-5th day, normalization of ESR parameters and total leukocyte count, decrease in the content of stab and segmented leucocytes, increase in the number of lymphocytes and monocytes were observed in the main group in comparison with the control group, which coincides with the literature data.11,12,13,14,15,16
Factors that could significantly affect the findings of the study were not identified. In the course of the treatment, minor autohemotherapy was used to prevent postinjection complications and reflexology in the absence of special equipment.
Conclusion
Thus, our studies have shown that supplementing the comprehensive treatment of patients having acute purulent surgical diseases in the main group with intravenous and topical application of ozone-containing medications resulted in:
Earlier improvement in the general condition of patients,
Decrease in endotoxicosis indices, acceleration of regenerative processes in the purulent wounds, decrease in the number of microbial bodies in smears-prints,
Reduction of bed-days.
It should be noted that ozone therapy methods have a pronounced curative effect in DFS, they are easy to use, available to any surgical hospital and are economically viable.
Acknowledgement
The authors express gratitude to the whole medical staff of Surgery Department №1 (the branch manager – Dzagalov M.M.) and of the Angiosurgery Department (the branch manager – Batov A.Z.) of the SBHI “Republican Clinical Hospital” of the Ministry of Healthcare of Kabardino-Balkarian Republic for help in fulfilling the research.
Conflict of Interest
No conflict of interest to declare.
The Sources of Funding
The work is done completely at the expense of the author.
References
- Malyar N.M, Freisinger E, Meyborg M, Lüders F and Lawall H. Amputations and mortality in in-hospital treated patients with peripheral artery disease and diabetic foot syndrome. Journal of Diabetes and its Complications. 2016;30(6):1117-1122.
CrossRef - Pscherer S, Dippel F.W, Lauterbach S and Kostev K. Amputation rate and risk factors in type 2 patients with diabetic foot syndrome under real-life conditions in Germany. Primary Care Diabetes. 2012;6(3):241-246.
CrossRef - Obolenskiy V.N, Leval P.S, Ermolova D.A and Protsko V.G. Diabetic foot syndrome: Our approach to the treatment of the patients. Wound Medicine. 2014;4:30-36.
CrossRef - Bocci V, Zanardi I, Huijberts M.S.P and Travagli V. Diabetes and chronic oxidative stress. A perspective based on the possible usefulness of ozone therapy. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2011;5(1):45-49.
CrossRef - Mitish V.A, Eroshkin I.A, Galstyan G.R, Doronina L, Paskhalova Y, Eroshenko A. Purulent-necrotic lesions of neuroischemic form of diabetic foot syndrome. New possibilities of complex surgical treatment. Endocrine surgery. 2008;2(1):15-19.
CrossRef - Obolenskiy V.N, Protsko V.G and Komelyagina E.Y. Classification of diabetic foot, revisited. Wound Medicine. 2017;18:1-7.
CrossRef - Sohrabi S and Russell D. Diabetic foot and foot debridement technique. Surgery (Oxford). 2017;35(9):500-504.
CrossRef - Miller R.J. Neuropathic Minimally Invasive Surgeries (NEMESIS): Percutaneous Diabetic Foot Surgery and Reconstruction. Foot and Ankle Clinics. 2016;21(3):595-627.
CrossRef - Kulikov G.A, Turova E.A and Kiseleva O.M. The Effectiveness of different methods of ozone therapy in vascular complications of diabetes mellitus. Problems of balneology, physiotherapy and exercise therapy. 2002;5:17-20.
- Maslennikov O.V and Kontorshchikova K.N. Practical ozone therapy. Allowance. Vector-Tis, Nizhny Novgorod. 2003.
- Noor S, Khan R.U and Ahmad J. Understanding Diabetic Foot Infection and its Management. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2017;11(2):149-156.
CrossRef - Charles P.G.P, Uçkay I, Kressmann B, Emonet S and Lipsky B.A. The role of anaerobes in diabetic foot infections. Anaerobe. 2015;34:8-13.
CrossRef - Uçkay I, Aragón-Sánchez J, Lew D and Lipsky B.A. Diabetic foot infections: what have we learned in the last 30 years? International Journal of Infectious Diseases. 2015;40:81-91.
CrossRef - Peters E.J. G and Lipsky B. A. Diagnosis and Management of Infection in the Diabetic Foot. Medical Clinics of North America. 2013;97(5):911-946.
CrossRef - Karmaker M, Sanyal S.K, Sultana M and Hossain M.A. Association of bacteria in diabetic and non-diabetic foot An investigation in patients from Bangladesh. Journal of Infection and Public Health. 2016;9(3): 267-277.
CrossRef - Salvia L.M.T, Clark A.T and Karchmer A.W. Diabetic Foot Infection. Hospital Medicine Clinics. 2012;1(2):185-198.
CrossRef







