Manuscript accepted on :September 13, 2017
Published online on: --
Plagiarism Check: Yes
Utkarsh Mangal
Consultant Orthodontist, 21/63 Bhrigu Path, Mansarovar Jaipur-302020, Rajasthan India.
DOI : https://dx.doi.org/10.13005/bpj/1253
Abstract
With increasing awareness of orthodontic treatment in all age groups of society, emphasis on enhancing the rate of tooth movement has increased. This article discusses the existing methods of accelerating tooth movement and the role of platelet rich plasma. Also briefly describes the methods of use of platelet rich plasma and its effects on orthodontic tooth movement.
Keywords
growth factor Platelet rich plasma (PRP); tooth movement; orthodontics;
Download this article as:| Copy the following to cite this article: Mangal U, Influence of Platelet Rich Plasma on Orthodontic Tooth Movement: A Review. Biomed Pharmacol J 2017;10(3). |
| Copy the following to cite this URL: Mangal U, Influence of Platelet Rich Plasma on Orthodontic Tooth Movement: A Review. Biomed Pharmacol J 2017;10(3). Available from: http://biomedpharmajournal.org/?p=16686 |
Introduction
Since the inception of the practice of orthodontics, one of the domain in the research has been the tooth movement and associated biological reaction. Research has been done to study various approaches to achieve tooth movement with most physiological manner but with maximum pace. The increasing demand for orthodontic treatment in the adult patients has given impetus to finding methods to achieve accelerated orthodontic tooth movement. This has led to research in different modalities including chemical, surgical and mechano-surgical methods. Many of these procedures make use of the regional acceleratory phenomenon (frost 1983), based on the principle that when the bone is irritated surgically, an inflammation cascade is initiated which caused increased osteoclastogenesis, hence causing faster tooth movement. Majority of the procedures involve an insult to the bony tissue. This has alternatively promoted the need for non-invasive to less invasive procedures, leading to increased research in field of acceleration of orthodontic tooth movement by use of untrasonic vibrations, photobiomodulation, low level laser therapy, and use of pharmaceutical approach.
All of the above approaches, though, proven to be effective, vary largely in the degree of effectiveness. The invasive techniques involving more bone removal such as conventional corticotomy have been significantly more effective than the non-surgical procedures or the less invasive procedure like micro-osteoperforations or peizopuncture1
In comparison to the non-invasive approaches, all surgical procedures cause insults to the bone. The mechanical simulation triggers a higher osteoclastic activity leading to resorption of the alveolar bone causing decrease of the alveolar bone density, and loss of alveolar bone of the target teeth. This, phenomenon, however, is absent in non-invasive procedures and not long lasting in minimal insult techniques like piezocision,2 piezopuncture3,4 and micro-osteoperforation.5
Therefore, for achieving equally effective biological response from the minimally invasive procedure, biochemical adjuncts may be used. This involves use of cytokines, such as prostaglandin and hormones like relaxin. However, the use of supplementary hormones or other allogenic products can cause undue systemic effects.
Platelet rich plasma (PRP) is another approach in tissue regeneration which has been widely used in various surgical fields including dental surgical procedures. Recently, PRP has become a valuable adjunct to promote healing in many procedures in dental and oral surgery including ablative surgical procedures, and surgical repair of the alveolar cleft and periodontal plastic surgery, as well as procedures relating to the placement of osseointegrated implants. “In such procedures, the adhesive nature of PRP facilitates the easier handling of graft material, with more predictable flap adaptation and hemostasis, and a more predictable seal than is the case with primary closure alone” 6–13
PRP: Definition and Biological Composition
PRP was introduced in dental literature, in 1998, by Robert Marx,6 as an adjunct in the mandibular reconstructive procedure, enhancing the radiographic maturation rate of the graft alone.
Platelet-rich plasma (PRP) is an autologous concentration of human platelets in a small volume of plasma. It comprises of the concentration of platelets and the seven-fundamental growth factor which are actively secreted by platelets to initiate wound healing.
These Growth Factors include:14:
Three isomers of platelet-derived GF (PDGFaa, PDGFbb, and PDGFab), 2 of the numerous transforming GFs-b (TGFb1 and TGFb2),
Vascular endothelial GF (VEGF), and Epidermal GF (EGF)
The small volume of plasma encompassing the platelet concentration contains three cell adhesion proteins viz. fibrin, Fibronectin, and vitronectin, which are essential for osteoconduction and forming a matrix for bone, connective tissue linkage, and epithelial migration., all of which are critical in the process of healing6
Synthesis of PRP
The use of PRP in dental procedures, focusing on wound healing and reconstruction, has largely been in the gel form, which in contrast to conventional medical procedure, is prepared as gelled admix of PRP with CaCl2 and thrombin.
This combination causes a booster action of the PRP with instantaneous release of growth factors alike a bolus dose. However, in orthodontic treatment, a long duration of action with constant and slow release of such growth factors is ideally desirable, to provide prolong
The process of fabrication of PRP involves the use of machine centrifugation process, which should be sterile and precisely suited to platelet separation from the RBC and their sequestration in high concentration without lysing the platelets or damaging them losing their secretory potential.
The autologous PRP should be prepared under aseptic processing procedures.
The following procedure, as described by Eric Lou et al, can be used to prepare injectable PRP solution.
A volume of 60 ml of whole blood is drawn from the medial cubital vein of a patient using three 30 ml syringes that each contained 3 ml of 10% sodium citrate solution as an anticoagulant.
Heparin is not recommended for using as the anticoagulant due to its systemic effects and inducing alveolar bone resorption.
One ml of the blood is used for checking the platelet counts.
The remaining 59 ml of whole blood is first centrifuged at 1000 rpm for 12 min at room temperature.
The blood is then separated into its three basic components as the RBCs at the bottom, the buffy coat (platelets) in the middle, and the poor platelet plasma (PPP) at the top.
The RBCs are discarded, and the remaining buffy coat and PPP are collected and centrifuged again at 3000 rpm for 8 min.
After the second centrifugation, the PPP is removed until 4 ml remained, and then the remaining PPP is mixed with the buffy coat to become PRP.
One ml of the PRP is analyzed for its platelet count.
Mechanism of Action of PRP
The action of PRP is derived by the degranulation of cellular alfa-granules, consisting of growth factors and cytokines. These biochemical messengers generate during the clotting process while the coagulation occurs.6
It initiates with secretion of the growth factors, within first hour of the process of clotting, with majority of the messengers derived in 1-2 hours. Hence, PRP must be synthesized in an anticoagulated state and used within ten mins of clot initiation. The anticoagulated state PRP remains viable for up to 8 hours in a sterile environment.6
The growth factors in the granules within the platelets are in an inactive state and made soluble by initiation of the clotting mechanism.
Following an initial burst of PRP-GFs, the synthesis and secretion of the additional GFs by the platelets continue for the remaining lifespan of 5-7 days. After this, the inflammatory macrophages, continue to stimulate healing by secretion of similar growth factors. Therefore, the number of platelets in the blood clot within the graft, wound, or adherent to a flap sets the rate of wound healing. PRP being a rich source of platelets serves as the supplement source, during the physiological course of healing, and provides an increased concentration of GF. This promotes the cellular activity and boosts the healing procedure.
For the success of the PRP, it is also essential that autologous sources be used, and pre-synthesised homologous should be avoided. This ensures HLA compatibility at the site of application.
The process of release of cytokines and their interaction with the cells can be appreciated in the figure 1.
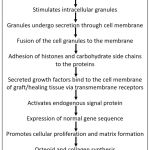 |
Figure 1: The biochemical pathway in PRP action
|
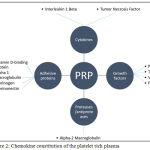 |
Figure 2: Chemokine constitution of the platelet rich plasma
|
Healing capacity of PRP on bone tissue is attributed to the anti-inflammatory effect of the PRP contents. However, the effect is multifactorial.
Table 1: Chemokines released derived from platelets via the granular release.
| Growth Factor | Effect |
| PDGF | chemo-attractant and stimulator of cell proliferation. (osteoprogenitor) |
| TGF | Promotes production of matrix proteins |
| EGF | Stimulates epithelial and mesenchymal cell proliferation and differentiation |
| VEGF | Mediator of angiogenesis |
Besides from GFs (Table 1.) the PRP also contains cytokines (ILs and TNFs), adhesive proteins, proteases, antiproteases, and leukocytes.
Orthodontic tooth movement can be described as an inflammatory process, and therefore the presence of ILs and TNFs contributes in the acceleration of the tooth movement.
Now, due to the multiple and overlapping effects of the constituents of the PRP, the effect on the bone tissue is considered multifactorial.
Therefore, the effectiveness of PRP is influenced by-
Concentration of the platelets
Cellular composition of PRP
Balance between anabolic and catabolic activities (Pro and anti-inflammatory cellular activity)
According to Hesham et al.,15 the action of PRP can be considered as an anti-inflammatory, by directly influencing the monocytic secretory activity.
Promotes production of RANTES (regulated on activation, normal t-cell expressed and secreted),
Blocks MCP-1 (monocyte chemotactic protein) release from monocytes,
Increases the concentration of LXA4, suggesting that PRP facilitates healing by controlling the local inflammatory response.
Efficacy of PRP: Soft Tissue Versus Hard Tissue
Though PRP does not contain any Bone Morphogenic Protein, the efficacy of PRP in the enhancement of the healing procedure of the bone is due to the interaction of the growth factors released which promote proliferation of adult mesenchymal stem cells. These also augment the rate of graft healing by improving osteoconduction and enhance bone formation.16
Clinical Usefulness of PRP
The relative ease of acquiring the PRP and its role as an autologous graft has simplified the clinical application. However, the usefulness and efficacy continue to be a topic of research.
The Dosage and Effects of Submucosal Injection of Platelet Rich Plasma
The duration of action of PRP injection is 5-6 months with a maximum acceleration of tooth movement observed in second to the fourth month after injection.
The regimen suggested by Liou et al.17 is as mentioned in table 2.
Table 2: The regiment for use of PRP injections by Liou et al12
| Frequency of injection | Time of injection | Purpose |
| Single | Beginning of treatment | Alignment and leveling. |
| Two | Beginning and six months after the first injection. | Anterior Retraction |
| Two | Beginning and six months after the first injection. | Protraction of posterior teeth |
Optimal PRP fold for a higher than the two-fold acceleration of orthodontic tooth movement and no pressure side alveolar bone loss is 11.0–12.5 as mentioned by Liou et al., by which it can be prepared by dilution of PRP with PPP.
In vitro study by Choi12 (JOMS 2004) concluded that the rate of bone cell proliferation reduced with increasing the concentration (>5%) of PRP
However, in an animal study done by Gulec et al,18 it was observed that the PRP effect is dose dependent with the effect of the high concentration of PRP lasts for longer, and enhances the rate of tooth movement, as much as up to 1.7 times.
Studies by Marx R6,14 have reported a positive effect on bone regeneration and enhanced graft uptake. This has also been supported in in-vivo study by Mazor et al.19
However, the additional benefit has been refuted in studies by Aghaloo et al20 and Froum et al.21
The reason behind the different results and clinical outcomes can be attributed to the existing differences in the protocols for obtaining the PRP.
True PRP is an autologous concentration of platelets in plasma, obtained via centrifugation, resulting in sequestration of the platelets with viable growth factor secretory potential.
For this purpose, an efficient protocol of preparation is essential. According to Marx R (2004), the use of FDA approved centrifugation units, and autologous blood is critical to obtain effective outcomes with use of PRP.
Safety of PRP and Possibility of Infection
The safety of PRP largely depends on the source of the blood used to synthesize the concentrate. With autologous source the chances of adverse reaction and are negligible. In a sterile protocol, the chance of transmissibility of the blood-borne infections is also avoided.
As the PRP concentrate of the platelets is essentially like the natural clotting, the concentrate does not promote bacterial proliferation.
Also, the PRP derived GF are only trans-membranous, they are not mutagenic and hence only stimulate natural healing process and have no role in tumour formation
Conclusion
PRP has the potential and capability to promote periodontal regeneration through various mechanisms. The effect of PRP in localized acceleration of tooth movement is dependent on the concentration used. However, the method of synthesis is critical to the success of PRP based acceleration of tooth movement. The use of injectable PRP at a different stage of orthodontic treatment can improve the quality of the treatment outcome by influencing the bone quality and enhancing the rate of tooth movement.
Acknowledgement
I would like to acknowledge Dr Siddarth Shetty, my teacher, for his guidance in completion of the review paper.
Conflict of Interest
None
Funding Source
None
References
- Kalemaj Z., DebernardI C. L., Buti J. Efficacy of surgical and non-surgical interventions on accelerating orthodontic tooth movement: a systematic review. Eur J Oral Implantol. 2015;8(1):9-24.
- Dibart S., Sebaoun J. D., Surmenian J. Piezocision: a minimally invasive, periodontally accelerated orthodontic tooth movement procedure. Compend Contin Educ Dent. 30(6):342-344
- Sebaoun J. D. M., Surmenian J., Dibart S. Accelerated orthodontic treatment with piezocision: a mini-invasive alternative to conventional corticotomies. L’Orthodontie Française. 2011;82(4):311-319.
CrossRef - Kim Y. S., Kim S. J., Yoon H. J., et al. Effect of piezopuncture on tooth movement and bone remodeling in dogs. Am J Orthod Dentofac Orthop. 2013;144(1):23-31..
CrossRef - Alikhani M., Raptis M., Zoldan B., et al. Effect of micro-osteoperforations on the rate of tooth movement. Am J Orthod Dentofacial Orthop. 2013;144(5):639-648.
CrossRef - Marx R. E., Carlson E. R., Eichstaedt R. M., Schimmele S. R., Strauss J. E., Georgeff K. R. Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(6):638-646.
CrossRef - Fennis J. P. M., Stoelinga P. J. W., Jansen J. A. Reconstruction of the mandible with an autogenous irradiated cortical scaffold, autogenous corticocancellous bone-graft and autogenous platelet-rich-plasma an animal experiment. Int J Oral Maxillofac Surg. 2005;34(2):158-166.
CrossRef - Del Corso M., Vervelle A., Simonpieri A., et al. Current knowledge and perspectives for the use of platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) in oral and maxillofacial surgery part 1: Periodontal and dentoalveolar surgery. Curr Pharm Biotechnol. 2012;13(7):1207-1230.
CrossRef - Plachokova A. S., Nikolidakis D., Mulder J., Jansen J. A., Creugers N. H. J. Effect of platelet-rich plasma on bone regeneration in dentistry: a systematic review. Clin Oral Implants Res. 2008;19(6):539-545.
CrossRef - Bae J. H., Kim Y. K., Myung S. K. Effects of Platelet-Rich Plasma on Sinus Bone Graft: Meta-Analysis. J Periodontol. 2011;82(5):660-667.
CrossRef - Holly D., Mracna J. The use of platelet rich plasma with guided tissue regeneration in defects caused by periodontal diseases. Bratislava Med J. 2009;110(10):669-671.
- Choi B. H., Zhu S. J., Kim B. Y., Huh J. Y., Lee S. H., Jung J. H. Effect of platelet-rich plasma (PRP) concentration on the viability and proliferation of alveolar bone cells: An in vitro study. Int J Oral Maxillofac Surg. 2005;34(4):420-424.
CrossRef - Arora N. S., Ramanayake T., Ren Y. F., Romanos G. E. Platelet-Rich Plasma in Sinus Augmentation Procedures: A Systematic Literature Review: Part II. Implant Dent. 2010;19(2):145-157.
CrossRef - Marx R. E. Platelet-Rich Plasma: Evidence to Support Its Use. J Oral Maxillofac Surg. 2004;62(4):489-496.
CrossRef - El-Sharkawy H., Kantarci A., Deady J., et al. Platelet-Rich Plasma: Growth Factors and Pro- and Anti-Inflammatory Properties. J Periodontol. 2007;78(4):661-669.
CrossRef - Slater M., Patava J., Kingham K., Mason R. S. Involvement of platelets in stimulating osteogenic activity. J Orthop Res. 1995;13(5):655-663.
CrossRef - Liou E. J. W. The development of submucosal injection of platelet rich plasma for accelerating orthodontic tooth movement and preserving pressure side alveolar bone. APOS Trends Orthod. 2016;6:5-11.
CrossRef - Güleç A., Bakkalbaşı B. Ç., Cumbul A., Uslu Ü., Alev B., Yarat A. Effects of local platelet-rich plasma injection on the rate of orthodontic tooth movement in a rat model: A histomorphometric study. Am J Orthod Dentofac Orthop. 2017;151(1):92-104.
CrossRef - Mazor Z., Peleg M., Garg A. K., Luboshitz J. Platelet-rich plasma for bone graft enhancement in sinus floor augmentation with simultaneous implant placement: patient series study. Implant Dent. 2004;13(1):65-72.
CrossRef - Aghaloo T. L., Moy P. K., Freymiller E. G. Evaluation of platelet-rich plasma in combination with anorganic bovine bone in the rabbit cranium a pilot study. Int J Oral Maxillofac Implants. 19(1):59-65.
- Froum S. J., Wallace S. S., Tarnow D. P., Cho S. C. Effect of platelet-rich plasma on bone growth and osseointegration in human maxillary sinus grafts: three bilateral case reports. Int J Periodontics Restorative Dent. 2002;22(1):45-53.







