Manuscript accepted on :May 29, 2017
Published online on: --
Plagiarism Check: Yes
Mohammed Arish1, Seyed Mohammad Mirhosseini2, Elahe Rajaei3, Asma Arish4, Seyed Davood Mirhosseini5 and Alireza Dashipour6
1FRCS Edin., Alzahra eye hospital, Zahedan University of Medical Sciences, Zahedan, IR Iran.
2Alzahra eye hospital, Zahedan University of Medical Sciences, Zahedan, IR Iran.
3Alzahra eye hospital, Zahedan University of Medical Sciences, Zahedan, IR Iran.
4Insurance social hospital, Zahedan, IR Iran.
5Department of Pharmacy School, Shahid Beheshti University of Medical Science, Tehran, IR Iran.
6Department of Nutrition and Food Science, School of medicine, Zahedan University of Medical Sciences, Zahedan, IR Iran.
Corresponding Author E-mail: dr.mirhosseini.62@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/1172
Abstract
According to differences between Primary Open Angle Gluacoma and Normal Tension Glaucoma (in pathogenesis, genetic, signs and symptoms) we tried to compare the Optical Coherence Tomography (OCT) appearance of the prelaminar and Lamina cribrosal thickness in these two disorders. So in this study we analyzed the anterior portion of the Optic Nerve Head in patients of primary open angle glaucoma (POAG) and normal tension glaucoma (NTG). 77 patients were analyzed between March 2013 till December 2014 according to the inclusion and exclusion criteria of this study; All patients aged between 30 to 65 years old. however, thirty-three (42.9%) patients (17 males and 16 female) of POAG and Forty-four (57.1%) patients of NTG group (22 males and 22 female). All of them underwent Enhanced Depth Image(EDI)-OCT (Carl ziess, sirus, 5000, German) on the first examination. Two observers measured the Optic Nerve Head Depth (ONHD), Anterior Surface of the Lamina Cribrosa depth (ASLCD) and Lamina Cribrosa Thickness (LCT) twice in each eye from the obtained EDI-OCT images manually and also measurement automatically was done by OCT relative program, finally Prelaminar Cribrosa Depth (PLCD) achieved by calculating the difference between ASLCD and ONHD mathematically. Independent sample T-test was used to compare means of PLCD, ONHD and LCT between the two groups (POAG & NTG). The results showed statistically significant variation between the two groups. Although mean of LCT was thicker in NTG group, PLCD and ONHD means were thinner. These findings support the notion that the LC is located more posteriorly in POAG group as compared with NTG group. And also LCT was significantly thicker in NTG group as compared with POAG group, so these two indices maybe considered as a point of differentiation between the POAG and NTG.
Keywords
Optical; Coherence; Tomography; According
Download this article as:| Copy the following to cite this article: Arish M, Mirhosseini S. M, Rajaei E, Arish A, Mirhosseini S. D, Dashipour A. Comparison of the Prelaminar and Lamina Cribrosal Thickness in Patients With Primary open Angle Glaucoma and Normal Tension Glaucoma by Optic Coherence Tomography (Oct). Biomed Pharmacol J 2017;10(2). |
| Copy the following to cite this URL: Arish M, Mirhosseini S. M, Rajaei E, Arish A, Mirhosseini S. D, Dashipour A. Comparison of the Prelaminar and Lamina Cribrosal Thickness in Patients With Primary open Angle Glaucoma and Normal Tension Glaucoma by Optic Coherence Tomography (Oct). Biomed Pharmacol J 2017;10(2). Available from: http://biomedpharmajournal.org/?p=15319 |
Introduction
Glaucoma is one of the common causes of irreversible blindness worldwide leaving a great burden on medical organization for detection, treatment and rehabilitation of this disease (1,2,3,4), especially primary open-angle glaucoma (POAG) which represents a significant public health problem (1,2). Although glaucoma related blindness is irreversible as no treatment can revived dead optic nerves but prevention is possible in almost all cases.(2)
Normal tension glaucoma (NTG) is known to be a special type of glaucoma which is still a matter of dispute in its etiologic, diagnostic, therapeutic aspects and its terminology. The criteria defining NTG have been highly variable though in general, individuals with Optic nerve head(ONH) damage and visual field(VF) defects compared with open angle glaucoma and with IOP which has never been documented above than 21 mmHg is regarded as NTG. (5)
A lot of studies have been carried out, that indicated difference of POAG vs NTG such as:
Optic nerve head: localized affection in NTG vs POAG. (6,7,8)
VF defect: overall VFD less affected in NTG and both central and paracentral defects were seen vs PAOG. The characteristic VF defect is an arcuate type and the central VF defect is the last portion to be affected. (8,9,10,11,12)
Genetic: different gene expression in lymphocytes in NTG vs PAOG. (13,14)
Blood flow: NTG associated with a transient episode of vascular insufficiency ONH and a variety of cardiovascular and hematologic abnormalities (increasing blood and plasma viscosity and hypercoagulability states). (15,16,17)
Central nervous system (CNS) functional Magnetic Resonance Imaging (fMRI): fMRI post yellow/blue stimuli, the fMRI activity decreasing was found to be greater than that in black/white stimulation in POAG. No similar effect was observed in NTG. (18)
Intracranial pressure (ICP): ICP is lower in NTG vs PAOG and non-glaucomatous control subject. This low ICP leaving to an abnormality high trans lamina Cribrosa pressure difference. (19,20)
The lamina Cribrosa (LC) is the continuation of the peripapillary scleral flange, which forms the anterior roof of the orbital cerebro-spinal fluid (CSF) space, which is the continuation of the inner layer of the posterior sclera. The LC is a porous connective tissue structure, which is regarded as a major site of irreversible damage to the retinal ganglion cell axons in glaucoma (21, 22).
In POAG, the mechanism of optic neuropathy is predominantly associated with the IOP. In NTG, although IOP is still an important factor, other pressure-independent factors such as an increased frequency of migraine headaches, Raynaud’s phenomenon, and sleep apnea have been observed, suggesting a vascular role in the nerve damage of patients with NTG (23,24).
The description of the anatomical structure of the LC as it appears in enucleated fixed specimens, one should note that the Lamina Cribrosa in vivo has a continuous movement. Due to pulse-synchronous changes in the Trans lamina Cribrosa pressure difference (TLPD), the LC moves back and forth in a sagittal direction in a synchronous frequency with the pulse (25,26,27).
The physiological importance of the Laminma Cribrosa movements remains unclear. One may hypothesize that the movements are helpful to allow the orthograde axoplasmic flow to enter the eye and the retrograde axoplasmic flow to leave it (28).
According to these data, in our study we tried to show that the OCT appearance of anterior portion of ONH is a site of differentiation between POAG and NTG.
This is largely due to Optical Coherence Tomography (OCT) images of the LC are therefore less informative (because of light-attenuation artifacts, such as vessel shadowing and fading of the signal with increasing depth) (29) than those acquired using gold standard ex vivo techniques, such as conventional histology (30), three-dimensional (3D) histomorphometry (31), and second harmonic generation imaging (32).
Although challenges remain in accurately delineating the posterior boundary of the LC and visualizing structures obscured by the retinal vasculature, Spectral Domain (SD)-OCT affords reliable measurement of the ONHD and anterior lamina Cribrosa surface, commonly defined by the perpendicular distances from a line joining the ends of Bruch’s Membrane Opening (BMO), or from the BMO plane fit in 3 dimensions, to the ONH surface and the anterior LC surface, respectively (34-41 and 45, 46).
Strouthidis et al (33) demonstrated a significant increase in anterior lamina cribrosa surface over a mean follow-up of 2.8 months in 9 Rhesus macaques induced with experimental glaucoma and proposed that ONH imaging and measurements would be useful in longitudinal follow-up of patients with glaucoma (33).
Clinical studies have demonstrated anterior displacement of the ONH and anterior LC surfaces after trabeculectomy (39-41) and posterior displacement of the ONH surface during acute (IOP) elevation (45).
Study of the ONH structures, including the LC, is relevant to understanding the mechanisms of retinal ganglion cell degeneration in glaucoma and devising new diagnostic and therapeutic strategies.
So according to above mentioned references about the major role which is played by LC, and of course controversy remains about whether NTG represents a
distinct disease entity or whether it is simply considered to be POAG with IOP within the average range (lower than 21 mmHg), in this study we tried to analyzed the anterior portion of the ONH to compare it in primary open angle glaucoma (POAG), and normal tension glaucoma (NTG), and we do emphasis on demonstrating that which part of the anterior ONH portion is significantly different from the others in these two groups and maybe considered as a differential point between POAG and NTG.
Methods
This prospective study enrolled newly diagnosed glaucoma patients. The inclusion criteria of POAG were as follows:
An IOP level greater than 21 mmHg in an eye with no antiglaucoma medications,
Characteristic glaucomatous ONH damage,
Glaucomatous visual field defect,
Open iridocorneal angle and normal angle in gonioscopic examination.
The inclusion criteria of NTG were as the follows:
An IOP lever below 21 mmHg in an eye with no antiglaucoma medications,
Characteristic glaucomatous ONH damage,
Related visual field defect (paracenteral or scotoma),
No neuro-radiological evidence of optic nerve damage,
Open iridocorneal angle, and normal gonioscopic examination.
Patients were excluded if the spherical equivalent of refractive error was out of the range above -6 and +6 diopters, or if an ocular disease other than glaucoma was present. This study was performed in ALZAHRA eye hospital, Zahedan university of medical science between March 2013 till December 2014.
Thirty-three eyes of Thirty-three patients (17 males and 16 females) with POAG and Forty-four eyes of Forty-four patients (22 males and 22 females) with NTG, which met the above criteria, aged between 30 to 65 years old were enrolled.
First, each person underwent a full ophthalmic assessment. Included slit-lamp biomicroscopy, achievement of the Best-Corrected Visual Acuity, diurnal IOP by Goldman tonometer, and central corneal thickness, dilated examination of the ONH and fundus, and visual field examination. Visual fields were achieved with SITA Standard profile central 30/2 (Humphrey Visual Field Analyzer II; Carl Zeiss-Meditec ).
Optic Nerve Head Depth (ONHD), Pre-Lamina Cribrosa Depth (PLCD) and Lamina Cribrosa Thickness (LCT) measurement:
All subjects underwent Enhanced Depth Image (EDI) Spectral Domain (SD) -OCT (Carl Ziess, Cirus, 5000, German) on the first visit. Two observers measured the ONHD, Anterior Surface of Lamina Cribrosa (ASLCD) and LCT twice in each eye from the obtained EDI-OCT images both manually and automatically by relative OCT program. The measurement was performed by a glaucoma specialist. To evaluate the interobserver reproducibility of our measuring method, the interclass correlation coefficient was calculated. The interobserver interclass correlation coefficient for measuring the prelaminar tissue thickness was 0.95 and all of them compared with automatically results and rechecked to be the same. All of parameters were measured perpendicularly from the reference line that connected the Bruch’s Membrane Openings (BMO). the visibility of the lamina Cribrosa configuration on all of the scan images was restored by adaptive compensation according to protocols published elsewhere (42,43).
ONHD measured from Bruch’s membrane to anterior surface of ONH. ASLCD was defined as Bruch’s membrane and the anterior surface of the lamina Cribrosa distance. The difference of these two measurements was regarded as the Prelaminar Cribrosa Depth (PLCD) in each eye, first perpendicular line was drawn at the center and second and third at 100μm nasally and temporally from a reference line connecting both ends of Bruch’s membrane opening (BMO). The prelamina Cribrosa depth (PLCD) equals the difference of ASLCD and ONHD (Fig. 1a, b,).
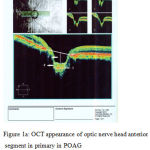 |
Figure 1a: OCT appearance of optic nerve head anterior segment in primary in POAG No.1 arrow shows ONHD, No.2 arrow shows PLCD and No.3 arrow shows LCT
|
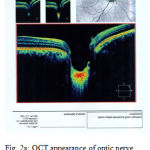 |
Figure 2a: OCT appearance of optic nerve head anterior segment in NTG The amount of ONHD, PLCD, LCT was calculated as the same as Fig. 1a
|
We defined the thickness of the lamina Cribrosa as the distance between the anterior and posterior borders of the highly reflective region in the horizontal EDI-OCT cross-sections of the ONH The measurement of LC thickness was performed as centrally as possible where there was minimal effect of vascular shadows (44).
Ethical Considerations
All patients underwent examination followed the tenets of the Declaration of Helsinki which was approved by the Zahedan University of Medical Sciences Research Ethics Committee.
Statistical Analysis
Independent sample T- test were used for analytic statistics. The Statistical analysis was performed for windows (v. 12.0.0; SPSS Inc., Chicago, IL) ,and P-Values < 0.05 were considered statistically significant.
Results
EDI SD-OCT images were achieved in 77 patients, thirty-three (42.9%) patients in
POAG group, Forty-four (57.1%) patients in NTG group, according to the inclusion and exclusion criteria; So no statistically significant differences were seen in age, sex, and also spherical equivalent, central corneal thickness between the POAG and NTG groups. All OCT and HRT parameters, including the RNFL average thickness and vertical cup/disc ratio, were similar between these two groups.
Table 1: ONHD, PLCD and LCT indices
| groups | N | Mean | Std. Deviation | t | df | p | |
| ONHD | NTG | 44 | 629.4 | 53.59 | -2.2 | 75 | 0.026 |
| POAG | 33 | 655.51 | 44.9 | ||||
| PLCD | NTG | 44 | 70.15 | 7 | -35.32 | 75 | |
| POAG | 33 | 172.03 | 17.36 | <0.0001 | |||
| LCT | NTG | 44 | 256.59 | 16.96 | 19.42 | 75 | <0.0001 |
| POAG | 33 | 168.39 | 22.91 |
Independent sample T-test was used to compare means of PLCD, ONHD and LCT between the two groups (POAG & NTG). The results showed that these parameters are variables between the two groups. Although mean of LCT was thicker in NTG group, PLCD means were thinner. (Table: ONHD, PLCD and LCT indices)
The mean LCT in POAG patients was 168.39 μm. However, in the patients with NTG, LCT was 256.59 μm, which is thicker than that in the POAG groups in this study (p-value < 0.0001). (Table: ONHD, PLCD and LCT indices).
There were statistically significant differences between ONHD in these two groups (p-value=0.026). 629.40 μm in NTG group vs 655.51 μm in POAG group. (Table: ONHD, PLCD and LCT indices)
We considered ASLCD as a more obvious index from Bruch’s membrane to insertion of lamina Cribrosa (two highest reflective lines) to achieve an exact measurement of PLCD by subtraction it from ONHD.
So, the mean PLCD (difference between ASLCD and ONHD) in POAG patients was 172.03 μm. However, in the patients with NTG, PLCD was 70.15 μm, which is thinner than that in the POAG group (p-value < 0.0001).
Discussion
In this prospective study we have shown that the LCT is thinner in POAG patients than in NTG group; however, PLCD was thicker in POAG patients than in NTG group.
According to the different OCT appearance of the anterior ONH portion in these two groups (POAG & NTG), these findings maybe support the notion that the posteriorly located lamina Cribrosa insertion and its thickness are more important components of glaucomatous optic nerve damage , As well as the Wang N et al study (46) and also Clinical studies which have demonstrated anterior displacement of the ONH and anterior LC surfaces after trabeculectomy (39-41) and posterior displacement of the ONH surface during acute (IOP) elevation (45).
So according to our study the LCT and PLCD may be considered as a point of differentiation between the POAG and NTG.
A previous study by Agoumi et al. [9] compared the prelaminar tissue between patients with glaucoma and controls in vivo after an acute IOP elevation. In their study, although their analysis focused on prelaminar tissue displacement, the prelaminar tissue thickness after an acute IOP elevation of approximately 13 mmHg in the patients with glaucoma decreased by 7.3 μm, whereas in age-matched normal controls, the thickness decreased by 20.6 μm. They explained that this decreased compliance in patients with glaucoma may have been due to tissue remodeling of the prelamina, which resulted in increased stiffness (47-49).
So we suggest that more studies to be carried out to get a clear role of the LC in pathogenesis of glaucoma, as if these changes which have been observed in the OCT of these patients might be a primary cause of the cupping or maybe secondary to IOP rising
And also IOP rising in the POAG is it the cause of LC changes or whether the displacement of LC and its thickness have led to elevation of IOP?
This study has limitations. First, the anterior and especially the posterior surface of LC was not always visible in all patients. However, we have taken the point of the consideration to avoid its bias. These parameters are likely to be invisible in eyes with a deeply located lamina Cribrosa. Consistent with this notion, ASLCD and PLCD was thicker in eyes in which the LC position was invisible. In addition, the ASLCD position was more often invisible in the POAG patients. This would lead to under estimation of the average ASLCD for the entire POAG patients. Thus, any potential bias derived from this limitation would actually support our finding. We consider that the true difference between the POAG patients versus NTG groups is greater than was revealed by the present study.
Acknowledgment
We do appreciate the sincere advices and encouragement by Dear Professor C. Hoyt in all steps for preparing this article.
The authors of this article also are thankful to Mrs. S. Sarani & Mrs. Poudineh as medical technician of ALzahra eye hospital for spending a lot of their time to get very nice and clear OCT pictures of our patients.
References
- Epstein DL, Allingham RR, Schuman JS, editors. Chandler & grants glaucoma. 4thed. New York: Lippincott Williams & wilkins; 1997.
- Shields MB. Textbook of the glaucoma. 4th ed. New York: Lippincott Williams & wilkins; 1998.
- Simmons ST. Glaucoma. In: Skuta GL, Cantor LB, Weiss JS, editors. Basic & clinical science course. San Fransisco: American academy of ophthalmology;2007.
- Diekmann H, Fischer D. Glaucoma and optic nerve repair. Cell Tissue Res. 2013 Aug;353(2):327-37.
CrossRef - Essentials in ophthalmology, Glaucoma edited by F. Grehn, R. Stamper, R.A.Hitchings, chapter 10. (page 147-155)
- Häntzschel J, Terai N, Furashova O, Pillunat K, Pillunat LE. Ophthalmologica. 2013 Dec 7
- Suh MH, Kim SH, Park KH, Yu HG, Huh JW, Kim DM.
- Hayamizu F, Yamazaki Y, Nakagami T, Mizuki K. Clin Ophthalmol. 2013; 7:807-13. doi: 10.2147/OPTH.S42468. Epub 2013 May 3.
CrossRef - Koch EC, Arend KO, Bienert M, Remky A, Plange N. ScientificWorldJournal. 2013 Oct 24; 2013:726912. doi: 10.1155/2013/726912. eCollection 2013.
CrossRef - Lee J, Kong M, Kim J, Kee C. J Glaucoma. 2013 Jun 6.
- Huang P, Shi Y, Wang X, Liu M, Zhang C.J Glaucoma. 2013 Apr 29.
- Sakata R, Aihara M, Murata H, Mayama C, Tomidokoro A, Iwase A, Araie M J Glaucoma. 2013 Mar;22(3):250-4. doi: 10.1097/IJG.0b013e31823298fb
CrossRef - Kato T, Meguro A, Nomura E, Uemoto R, Nomura N, Ota M, Kashiwagi K, Mabuchi F, Iijima H, Kawase K, Yamamoto T, Nakamura M, Negi A, Sagara T, Nishida T, Inatani M, Tanihara H, Aihara M, Araie M, Fukuchi T, Abe H, Higashide T, Sugiyama K, Kanamoto T, Kiuchi Y, Iwase A, Chin S, Ohno S, Inoko H, Mizuki N.
- Fraenkl SA, Golubnitschaja O, Yeghiazaryan K, Orgül S, Flammer J. Eur J Ophthalmol. 2013 May 31;23(6):841-849. doi: 10.5301/ejo.5000306.
CrossRef - Willekens K, Abegão Pinto L, Vandewalle E, Marques-Neves C, Stalmans I. Graefes Arch Clin Exp Ophthalmol. 2014 Mar;252(3):477-83. doi: 10.1007/s00417-013-2533-y. Epub 2013 Nov 27
CrossRef - Mamikonian VR, Galoian NS, Sheremet NL, Kazarian EE, Kharlap SI, Shmeleva-Demir OA, Andzhelova DV, Tatevosian AA. Vestn Oftalmol. 2013 Jul-Aug;129(4):3-8. Russian
- Shiga Y, Omodaka K, Kunikata H, Ryu M, Yokoyama Y, Tsuda S, Asano T, Maekawa S, Maruyama K, Nakazawa T.
- Lestak J, Tintera J, Svata Z, Ettler L, Rozsival P. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2013 Jun 4. doi: 10.5507/bp.2013.038
CrossRef - Wang N, Yang D, Jonas JB. J Glaucoma. 2013 Jun-Jul;22 Suppl 5: S11-2. doi: 10.1097/IJG.0b013e31829349a2. Review. Erratum in: J Glaucoma. 2013 Sep;22(7):597. Yang, Diya [added] CrossRef
- Wostyn P, De Groot V, Van Dam D, Audenaert K, De Deyn PP. Am J Ophthalmol. 2013 Jul;156(1):5-14. e2. doi: 10.1016/j.ajo.2013.03.003. Epub 2013 Apr 19
CrossRef - Ren, R., Wang, N., Li, B., Li, L., Gao, F., Xu, X., Jonas, J.B., 2009. Lamina cribrosa and peripapillary sclera histomorphometry in normal and advanced glaucomatous Chinese eyes with normal and elongated axial length. Investig. Ophthalmol. Vis.Sci. 50, 2175e2184.
- Quigley HA, Addicks EM, Green WR, Maumenee AE. Optic nerve damage in human glaucoma. II. The site of injury and susceptibility to damage. Arch Ophthalmol. 1981; 99:635–649.
CrossRef - Shields MB. Normal-tension glaucoma: is it different from primary open-angle glaucoma? Curr Opin Ophthalmol. 2008; 19:85–88. doi: 10.1097/ICU.0b013e3282f3919b. pmid:18301279
CrossRef - Park HY, Jeon SH, Park CK. Enhanced depth imaging detects lamina cribrosa thickness differences in normal tension glaucoma and primary open-angle glaucoma. Ophthalmology. 2012; 119:10–20. doi: 10.1016/j.ophtha.2011.07.033. pmid:22015382
CrossRef - Han, Y., McCulley, T.J., Horton, J.C., 2008. No correlation between intraocular pressure and intracranial pressure. Ann. Neurol. 64, 221e224.
- Harder, B., Jonas, J.B., 2007. Frequency of spontaneous pulsations of the centrai retinal vein in normal eyes. Br. J. Ophthamol. 91, 401e40
- Jonas, J.B., Wang, N., Yang, D., 2012a. Retinal vein pulsation is in phase with intracranial pressure and not intraocular pressure. Investig. Ophthalmol. Vis. Sci. 53, 6045.
CrossRef - Quigley, H.A., Anderson, D.R., 1976. The dynamics and location of axonal transport blockade by acute intraocular pressure elevation in primate optic nerve. Investig. Ophthalmol. Vis. Sci. 15, 606e616.
- Girard MJ, Strouthidis NG, Ethier CR, Mari JM. Shadow removal and contrast enhancement in optical coherence tomography images of the human optic nerve head. Invest Ophthalmol Vis Sci. 2011; 52:7738–7748.
CrossRef - Ren R, Li B, Gao F, et al. Central corneal thickness, lamina cribrosa and peripapillary scleral histomorphometry in nonglaucomatous Chinese eyes. Graefes Arch Clin Exp Ophthalmol. 2010; 248:1579–1585.
CrossRef - Yang H, Downs JC, Sigal IA, Roberts MD, Thompson H,Burgoyne CF. Deformation of the normal monkey optic nerve head connective tissue following acute IOP elevation within 3-D histomorphometric reconstructions. Invest Ophthalmol Vis Sci. 2009; 50:5785–5799.
CrossRef - Sigal IA, Grimm JL, Jan NJ, Reid K, Minckler DS, Brown DJ. Eyespecific IOP induced displacements and deformations of human lamina cribrosa. Invest Ophthalmol Vis Sci. 2014;55: 1–15.
CrossRef - Strouthidis NG, Grimm J, Williams GA, Cull GA, Wilson DJ, Burgoyne CF. A comparison of optic nerve head morphology viewed by spectral domain optical coherence tomography and by serial histology. Invest Ophthalmol Vis Sci. 51:1464 1474.
CrossRef - Mari JM, Strouthidis NG, Park SC, Girard MJ. Enhancement of lamina cribrosa visibility in optical coherence tomography images using adaptive compensation. Invest Ophthalmol Vis Sci. 2013; 54:2238–2247.
CrossRef - Hodapp E, Parrish RK II, Anderson DR. Clinical Decisions in Glaucoma. St Louis: Mosby; 1993.
- Copete S, Flores-Moreno I, Montero JA, Duker JS, Ruiz-Moreno JM. Direct comparison of spectral-domain and swept-source OCT in the measurement of choroidal thickness in normal eyes. Br J Ophthalmol. 2014; 98:334–338.
CrossRef - Lee EJ, Kim TW, Kim M, Girard MJ, Mari JM, Weinreb RN. Recent structural alteration of the peripheral lamina cribrosa near the location of disc hemorrhage in glaucoma. Invest Ophthalmol Vis Sci. 2014; 55:2805–2815.
CrossRef - Schneider CA, Rasband WS, Eliceiri KW. NIH image to ImageJ: 25 years of image analysis. Nat Methods. 2012; 9:671– 675.
CrossRef - Lee EJ, Kim TW, Weinreb RN, Park KH, Kim SH, Kim DM. Visualization of the lamina cribrosa using enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol. 2011; 152:87–95, e81.
- Miki A, Ikuno Y, Jo Y, Nishida K. Comparison of enhanced depth imaging and high penetration optical coherence tomography for imaging deep optic nerve head and parapapillary structures. Clin Ophthalmol (Auckland, NZ). 2013; 7:1995– 2001.
CrossRef - Yang H, Qi J, Hardin C, et al. Spectral-domain optical coherence tomography enhanced depth imaging of the normal and glaucomatous nonhuman primate optic nerve head. Invest Ophthalmol Vis Sci. 2012; 53:394–405.
CrossRef - Girard MJ, Strouthidis NG, Ethier CR, Mari JM. Shadow removal and contrast enhancement in optical coherence tomography images of the human optic nerve head. Invest Ophthalmol Vis Sci. 2011; 52: 7738–7748.
CrossRef - Mari JM, Strouthidis NG, Park SC, Girard MJ. Enhancement of lamina Cribrosa visibility in optical coherence tomography images using adaptive compensation. Invest Ophthalmol Vis Sci. 2013; 54: 2238–2247.
CrossRef - Dai Woo Kim, et all. prelamina and lamina Cribrosa in Glaucoma patients with unilateral visual field loss. Investigative ophthalmology and visual science April 2016, Vol.57, 1662-1670. Doi: 10.1167/iovs. 15-
- Park HY, Shin HY, Park CK. Imaging the posterior segment of the eye using swept-source optical coherence tomography in myopic glaucoma eyes: comparison with enhanced-depth imaging. Am J Ophthalmol. 2014;157:550–557.
CrossRef - Wang N, Xie X, Yang D, Xian J, Li Y, et al. (2012) Orbital cerebrospinal fluid space in glaucoma: The Beijing intracranial and intraocular pressure (iCOP) study. Ophthalmology 119:2065–2073 e2061. doi: 10.1016/j.ophtha.2012.03.054.
CrossRef - Hernandez MR. Ultrastructural immunocytochemical analysis of elastin in the human lamina cribrosa. Changes in elastic fibers in primary open-angle glaucoma. Invest Ophthalmol Vis Sci. 1992; 33:2891–2903. pmid:1526740.
- Hernandez MR, Andrzejewska WM, Neufeld AH. Changes in the extracellular matrix of the human optic nerve head in primary open-angle glaucoma. Am J Ophthalmol. 1990; 109:180–188. pmid:2405683.
- Hernandez MR, Ye H. Glaucoma: changes in extracellular matrix in the optic nerve head. Ann Med. 1993; 25:309–315. pmid:8217094.







