Manuscript accepted on :January 16, 2017
Published online on: --
Plagiarism Check: Yes
I. I. Ilyas1, N. T. Kartinah1, T. Andriani1, R. A. Goernarjo1, D. N. Kahanjak2 and H-J Freisleben3
1Department of Medical Physiology, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia.
2Department of Physiology, Faculty of Medicine, University of Palangka raya, Palangkaraya, Indonesia.
3Medical Research unit, Faculty of medicine, university Indonesia, Jalan Salemba Raya 6, Jakarta, 10430, Indonesia.
Corresponding Author E-mail: @ermitailyas@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/1076
Abstract
Overtraining is associated with impaired regeneration of body cells. One of the causes of overtraining syndrome is increased production of reactive oxygen species, causes decreased levels of antioxidants. Hibiscus sabdariffa Linn. contains antioxidants, especially anthocyanin. This study aims to investigate whether H. sabdariffa can prevent overtraining syndrome through decrease in MDA levels and increase in plasma GPx activity. The research subjects were 20 male rats divided randomly into 5 groups: C (control); C-Hib (400 mg/kg/day of H. sabdariffa); A-Ex (moderate aerobic exercise); OT (overtraining exercise); OT-Hib (overtraining exercise with H. sabdariffa 400 mg/kg/day). We examined GH, IGF-1, levels of free IGFBP-3, IGFBP-3 expression, MDA levels, and plasma GPx activity. We found IGFBP-3 gene expression in OT and OT-Hib higher than in groups C, C-Hib and A-Ex. The differences between the groups were not statistically significant (p> 0.05). MDA levels in the OT-Hib group were significantly lower than in group OT (p = 0.019). The mean level of GPx in OT-Hib was higher than in group OT, but the difference was statistically not significant. There were strong negative correlations between GPx activities and both, MDA levels and IGFBP-3 gene expression. In conclusion, we found that H. sabdariffa decreases MDA levels and increases GPx activity in our overtraining model and can thus prevent overtraining syndrome.
Keywords
Physical exercise; overtraining syndrome; oxidative stress; Hibiscus sabdariffa Linn; IGFBP-3 gene expression
Download this article as:| Copy the following to cite this article: Ilyas I. I, Kartinah N. T, Andriani T, Goernarjo R. A, Kahanjak D. N, Freisleben H. J. Effects of Hibiscus sabdariffa Linn. on Malondialdehyde and Glutathione Peroxidase in an Overtraining Rat Model characterized by Growth Hormone, Insulin-like Growth Factor-1, and Insulin-like Growth Factor Binding Protein-3. Biomed Pharmacol J 2017;10(1). |
| Copy the following to cite this URL: Ilyas I. I, Kartinah N. T, Andriani T, Goernarjo R. A, Kahanjak D. N, Freisleben H. J. Effects of Hibiscus sabdariffa Linn. on Malondialdehyde and Glutathione Peroxidase in an Overtraining Rat Model characterized by Growth Hormone, Insulin-like Growth Factor-1, and Insulin-like Growth Factor Binding Protein-3. Biomed Pharmacol J 2017;10(1). Available from: http://biomedpharmajournal.org/?p=13364 |
Introduction
Long-term training of athletes, especially in the preparation phase for competitions can lead to overtraining syndrome due to the volume, duration and intensity of exercise exceeding the athlete’s capability. The overtraining syndrome is characterized by reduction of performance due to disturbances in physical condition, immunity, and emotion1-3. The occurring physical disturbance settles for a certain period of time and cannot be overcome during this time despite being given adequate rest. This indicates an interruption in the recovery phase and longer time requirement to restore the body3-4. The recovery phase is needed to repair damaged tissues and to regenerate them through the processes of anabolic protein metabolism, replication, and differentiation of skeletal muscle cells. The mechanisms of the body response for tissue regeneration are mediated by the insulin-like growth factor-1 (IGF-1). IGF-1 is produced in the liver through stimulation by growth hormone (GH) and in the plasma, it binds to its carrier protein, the insulin-like growth factor-binding protein-3 (IGFBP-3)2. As a response to adequate physical exercise the body should react by increasing the production of GH, IGF-1 and IGFBP-33,5,6.
In a previous study unbound IGFBP-3 in the plasma (free plasma IGFBP-3) decreased in over-trained subjects6. The decrease of IGFBP-3 was considered an indicator or marker of overtraining syndrome7,8. However, the mechanism of the decrease of free IGFBP 3 in the plasma is not yet known. Interference between the actions of GH, IGF-1 and IGFBP-3 is estimated to occur in athletes with overtraining syndrome and the processes of anabolic protein metabolism and skeletal muscle cell replication and differentiation are considered to become impaired3.
Based on these considerations, questions arose how physical exercise and overtraining affect the levels of IGFBP-3 in the plasma and whether the levels of IGFBP-3 are influenced by the levels of IGF-1 and GH. Therefore, this study was conducted to determine the effect of aerobic exercise in the overtraining rat model on the levels of IGF-1, GH, IGFBP-3 and the interference with IGFBP-3 expression.
It was suggested that overtraining syndrome is associated with oxidative stress9. Excessive reactive oxygen species (ROS) can occur because of the increased oxygen consumption during severe aerobic exercise. Therefore, we investigated whether decreased IGFBP-3 in the overtraining syndrome is caused by impaired IGFBP-3 gene expression and how it is influenced by Hibiscus sabdariffa.
Most studies examined oxidative stress caused by physical exercise through lipid peroxidation, commonly determined by the production of its by-product, malondialdehyde (MDA)10-13. To overcome damages by free radicals, the body is equipped with endogenous antioxidants, both enzymatic, e.g., superoxide dismutase (SOD), catalase, and glutathione peroxidase (GPx) and non-enzymatic, e.g., glutathione (GSH)14. Examples of exogenous non-enzymatic antioxidants are various vitamins and so-called secondary plant components like anthocyanin, flavonoids, and many others12. This study investigates the effect of exogenous antioxidants from H. sabdariffa to prevent the overtraining syndrome in rats, especially the impact on IGFBP-3 gene expression and on the plasma level of free IGFBP-3. The plant known as Red Tea or Rosela is a family member of Malvaceae, native to Asia or Africa15. In traditional medicine, calyx extract has been used for various indications; we were especially interested in its powerful antioxidant properties in the overtraining rat model16.
Materials and Methods
This research has been approved by the Ethical Committee of Medical Faculty, Universitas Indonesia / Cipto Mangunkusumo Hospital (No. 289 / H2.F1 / ETIK / 2013).
Experimental Animals
This study was an experimental study using 30 male Wistar strain Rattus norvegicus (200 -250 g) provided by BPOM RI (Indonesian Food and Drug Administration). Before and during treatment, the rats were maintained healthy, fed a standard diet with free access to drinking water ad libitum. Cages were kept clean and air conditioned (ambient temperature 23±1°C) with a light-dark cycle of 12 hours. The animals were maintained properly according to the code of conduct of the animal handling commission in use of experimental animals. The animals were acclimatized for 3 weeks and to get them familiarized with the research conditions, two weeks for the environment and the third week for the training program. The size of the cages was 40cm x 50cm x 40cm and each cage contained five animals. Cages had a wooden base and were equipped with containers for food and water. Physical exercise devices used in this study were animal treadmills type L-6000 with six lanes (tracks) that can be used simultaneously. Duration, speed, and the slope of the tracks could be adjusted according to the desired training procedure. At the end of each track a metal plate was connected to electric current of 2 mA to deliver an electric shock to the animal in case the animal stopped walking (or running) or did not follow the direction or velocity of the track. In the experiments the velocity of the track was set according to the aerobic exercise program or to the aerobic overtraining program established by Hohl17. To introduce the training program in the acclimatization phase and to minimalize the stress in subsequent experiments the animal treadmill was set to low speed (~12m / min) without a slope. Duration of training was less than 10 minutes for each session in one day.
Animals were divided randomly into 5 groups: 1) control group (C), 2) control group with administration of H.sabdariffa at a dose of 400 mg/kg/ day (C-Hib), 3) group of rats with mild aerobic exercise (A-Ex), 4) group of rats with aerobic overtraining program (OT), 5) group of rats with aerobic overtraining program and administration of H. sabdariffa at a dose of 400 mg/kg/ day (OT-Hib). For the treatment given to the mild aerobic exercise group (A-Ex) the treadmill was running at a speed of 12m/min, for 10 min. The exercise was performed twice a week for 11 weeks17.
After completion of the 11 weeks training program, blood samples were taken 3 days later in order to eliminate the acute effect of aerobic physical exercise9. Rats were sacrificed with the application of ether and blood samples taken by cardiac puncture. The levels of serum GH, IGF-1 and IGFBP-3 were measured using ELISA Kits for rat growth hormone (GH ELISA Kit – Cusabio®), for rat insulin-like growth factor 1 (IGF-1 ELISA Kit – Cusabio®), and for rat insulin-like growth factor binding protein 3 (IGFBP-3 ELISA Kit – Cusabio®).
The treatment given to the aerobic exercise overtraining (OT and OT-Hib) group was treadmill running with a gradual increase of duration and speed over the 11 weeks of the program. H.sadariffa extract was obtained from the Herbal Research Centre in Bogor, Indonesia and administered through a syringe cannula orally into the stomach, every day for 11 weeks, except on Saturdays and Sundays for the C-Hib and oT-Hib groups. Our dosage of 400 mg/kg/day is based on an investigation of the influence of H. sabdariffa in rats with diabetes mellitus referring to the optimal dose of 400mg/kg/day18.
Measurement of Parameters
Blood sampling performed on day 3 post-treatment was done for measuring, GH, IGF-1, IGFBP-3, gene expression of IGFBP-3, MDA, and GPx. Rats were sacrificed with ether, then blood samples were taken by cardiac puncture. Measurement of the levels of GH, and IGF-1, was performed by ELISA technique as mentioned above. MDA content was measured by means of a TBARS (TCA Method) Assay Kit No. 100870 (Cayman Chem, Ann Arbor, MI, USA) based on the method of Yagi19. GPx content was measured using RS 505 Backpack kit from Randox Laboratories Limited20. The measurements were done in the Department of Biochemistry of Medical Faculty, Universitas Indonesia. In addition, the liver tissue was taken to examine the gene expression of IGFBP-3, was done in the Research Laboratory of Department of Oral Biology, Faculty of Dentist, Universitas Indonesia. Liver tissue was soaked with Ringer’s solution and then stored at -80°C. Qualitative RT PCR (qPCR) method was used to examine the gene expression. Measurement of gene expression was performed in the following stages: extraction of RNA, preparation of cDNA, RT-PCR.
RNA Extraction
Liver homogenate was produced by adding 1 mL of Trizol® Reagent to 50 mg of liver tissue. Then the samples were incubated at room temperature and continued to be centrifuged at a speed of 12,000 x g for 15 minutes at 4°C. In the separation phase, the liquid part was taken from the sample. In the RNA precipitation stage, the sample was incorporated with 0.5 mL of isopropanol 100% for each 1 mL of the Trizol® Reagent, then incubated for 10 minutes and centrifuged with a speed of 12,000 x g for 10 min at 4°C. Furthermore, the RNA was washed by removing the supernatant. Subsequently, the RNA pellet was washed with 1 mL of ethanol 75% for each 1 mL of Trizol® Reagent. Then it was briefly vortexed before being centrifuged at a speed of 7500 x g for 5 min at 4°C. For RNA resuspension about 20-50 mL RNase-free water was used to be mixed into the tube and then the tube was incubated in a water bath for 10-15 minutes.
DNA Preparation
Mixing iScript 5x reaction mix, iScript reverse transcriptase, nuclease-free water and RNA template (100 fg to 1 pg total RNA) were mixed to reach a total volume of 20 mL. The mixture was incubated at 25°C for 5 minutes, at 42°C for 30 minutes, and at 85oC for 5 minutes.
Primer Design
Primers used in the quantitative RT PCR can be seen in Table 1.
Table 1: Primer pairs used in Real Time PCR quantification.
| Gene Bank
ACC No |
Gene
Name |
PS Forward 5` to 3` | PS Reverse 3` to 5` | Amplificon
size |
AT | QT |
| NM 012588 | IGFBP 3 | CGC TAC AAA GTT GAC TAT GAG | CGT CTT TCC CCT TGG T | 292 | 60 | 79 |
| NM 001101 | B Actin | CTA CGT CGC CCT GGA CTT CGA GC | GAT GGA GCC GCC GAT CCA CAC GG | 385 | 60 | 79 |
Abbreviations: GenBank Accession numbers, PS=Primer sequence, AT=annealing temperature, QT=quantification temperature, bp=base pairs, IGFBP-3=insulin like-growth factor binding protein-3.
RT-PCR
Entered into each well of cDNA were 1 mL of Fp (B-actin or IGFBP 3 depending on the well), 1 mL of Rp (B-actin or IGFBP 3 depending on the well), 1 mL of Syber Green Reagent 5 mL, and 2ml of dH2O, so each well contained a total volume of 10 mL. Gene amplification was accomplished as follows: polymerase activation stage, 95°C for 190 seconds; the stage of denaturation, 95°C for 15 seconds; annealing (AT 10 seconds), elongation (72°C, 20 seconds), quantification (QT 5 seconds).
Statistical Analysis
Data were analyzed using SPSS 17; analysis was performed with one way ANOVA followed by post-hoc LSD. The level of statistical significance was set at p < 0.05. Correlations were calculated using Pearson’s analysis.
Results
Levels of Growth Hormone, IGF-1, and IGFBP-3 and IGFBP-3 Gene Expression
In Table 2, GH levels were not significantly different between all groups (p > 0.05), although they tended to be lower in C-Hib and OT. On the other hand, GH level in OT-Hib was highest in all groups. Levels of IGF-1 were not significantly different between all groups (p>0.05), although IGF-1 was higher in the OT-Hib, OT and C-Hib groups than in C and A-Ex. Levels of IGFBP-3 were significantly different (p<0.05) between OT-Hib and both, groups C and OT. IGFBP-3 gene expression was not significantly different between all groups (p>0.05); it was higher in OT and OT-Hib groups than in the other groups (C, C-Hib, and A-Ex), where their values were almost alike.
MDA Levels
MDA levels differ significantly between groups A-Ex (3.3 ± 0.9 nmol/mL), C (1.5 ± 0.6 nmol/mL), and C-Hib (0.6 ± 0.2 nmol/mL; Fig.1, *a), between OT (3.6 ± 0.8 nmol/mL) and C, C-Hib, OT-Hib (2.4 ± 0.9 nmol/mL; Fig.1, *b) and between OT-Hib and C, C-Hib (*c,d), all differences with p<0.05. Highest values were found in group OT, followed by A-Ex. In group OT-Hib MDA levels were lower than in the two former groups (Fig. 1).
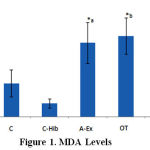 |
Figure 1: MDA Levels |
MDA levels in control (C), control-H.sabdariffa (C-Hib), mild aerobic exercise (A-Ex), overtraining exercise (OT) and overtraining exercise-H.sabdariffa (OT-Hib) groups. Mean values, error bars indicate standard deviations. (Note: test results performed with α = 0.05).
GPx Activities
Lowest GPx activity was found in the OT group (1.4 ± 0.1 U/L), which was significantly different from C-Hib (1.8 ± 0.1 U/L) and A-Ex (1.7 ± 0.1 U/L; Fig.2,*c,d). OT-Hib group (1.5 ± 0.3 U/L) shows the same value as controls and is significantly lower than group C-Hib (1.8 ± 0.1 U/L; Fig. 2,*a), whereas A-Ex significantly differs from controls (1.5 ± 0.1 U/L; Fig.2,*b); all these differences with p<0.05 (Fig. 2).
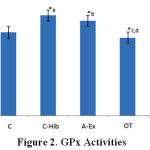 |
Figure 2: GPx Activities |
GPx activity in control (C), control-H.sabdariffa (C-Hib), mild aerobic exercise (A-Ex), overtraining exercise (OT), and overtraining exercise-H.sabdariffa (OT-Hib) groups. Mean values, error bars indicate standard deviations. (Note: test results performed with α = 0.05).
Correlations
Correlations were calculated between all parameters measured, between physiological parameters (GH, IGF-1, IGFBP-3) correlations were expected, because they are not considered independent (e.g., strong correlation of 0.784 between IGF-1 and IGFBP-3 expression). More important were correlations of MDA levels and GPx activities, e.g., between levels of MDA and IGFBP3, and between IGFBP3 expression and GPx activities. There were strong negative (inverse) correlations between GPx activities and both, MDA levels (r = -0.527) and IGFBP3 expression (r = -0.625, p = 0.006). No further correlations could be found; after our preliminary experiments, we had suspected strong negative correlation between the levels of MDA and IGFBP3; however, this result could not be confirmed in our main experiments.
Discussion
Overtraining is experienced as a decrease in performance and physiological functions within weeks up to years caused by psychological and physiological factors, although the exact mechanisms are not yet fully understood1-3,21-26. In humans, conditions are more common in populations who have high levels of physical activity such as athletes in training programs or during training camps to achieve maximum performance. Research on overtraining syndrome using human subjects has obstacles, not only because of ethical concerns, but also because of differing designs and incomparable protocols. Therefore, researchers use male Wistar rats as test subjects in an overtraining animal model17. This model applies standard physical overtraining exercise protocols and allows for analysis of biomarkers from body tissues and comparison of results between studies.
At the time of starting the treatment phase in our experiments, the age of the rats was eleven weeks and at the end of the treatment phase 22 weeks. The age of 11-22 weeks in rats is equivalent to 17-28 years of age in man27.
Growth hormone (GH) is secreted from the pituitary glands (hypophysis) under regulation of the hypothalamic GHRH4,28. Physical exercise is one of the factors that increase the secretion of GHRH and thus, GH level4,29,30.
The significance of these results strongly depends on the time of measurements after physical exercise29. GH increased immediately post-training as an acute response and returned to normal within 6 hours after exercise because of the negative feedback mechanism of the axis GH/IGF14. We conducted the GH plasma assay in response to chronic stimulus so that there were no significant differences; however, there was a tendency to decreased GH in the OT group29.
The release of GH by anterior pituitary gland into circulation stimulates the liver to produce IGF-15,6,29,30. Apart from liver, GH also goes into other tissues such as muscles and bones stimulating the formation of IGF-1 via these networks5,6, thus constituting the main factor that increases IGF-1 secretion after aerobic exercise29-31. Two weeks of aerobic exercise increased IGF-1 levels in circulation31. However, the increase was temporary only in the acute phase and returned to the original levels within 10-15 minutes after training30.
The secretion of IGFBP-3 is also mediated by the activation of the GH/IGF axis, i.e., the levels of IGF-1 and IGFBP-3 exert positive correlations with GH secretion4. Thus, elevated levels of IGF-I and IGFBP-3 found in physical exercise are due to increased secretion of GH from pituitary gland, which then stimulates IGF-I release from the liver and other tissues26-31.
As shown in Table 2, GH in the OT group was lower than in the C, A-Ex and OT-Hib groups. In the A-Ex group GH was slightly increased, consistent with reports that physical exercise can increase the secretion of GH indirectly through increased secretion of GHRH. This response is closely related to the physical exercise intensity and the higher the intensity, the greater the GH increase. The increase was found immediately after exercise as an acute response and returned to normal after 6 hours, post workout because of the negative feedback mechanism of the axis GH/IGF1. This study measured the levels of GH in response to chronic stimulus so that there are no significant differences, although there is a tendency of decrease in baseline GH in OT group. Decrease in baseline GH in OT group will influence the secretion of IGF-1 and IGFBP-3. As we know, GH stimulates the liver to produce IGF 1. Decrease in GH will correlate with a decrease in IGF-1, but in this study, we found a tendency of IGF-1 increase in the OT group. This increase is suspected to occur because of the initial significant decrease in IGFBP-3 reported in our previous study8. Approximately, 98% of IGF-1 in plasma binds to IGFBP-3. Thus, a decrease in IGFBP-3 will cause the amount of free IGF-1 to increase. Although the reduction mechanism is still unclear, the decrease in IGFBP 3 is suspected to be caused by interference on the axis of GH/IGF1 or interruption in the biosynthesis of IGFBP3 in liver cells. Hibiscus seems to interfere with the GH/IGF-1 axis, as we can assume from the results of C-Hib group with lower GH but higher IGF-1 levels compared to controls.
Table 2: The levels of GH, IGF-1, IGFBP3 and IGFBP3 gene expression
| Group | GH [mIU/L] | IGF-1 [mIU/L] | IGFBP-3 [mIU/L] | IGFBP-3 Gene expr. [arb. units] |
| C | 198.4±26.2 | 4.1±0.9 | 17.4±10.2
* vs. OT-Hib |
2.5±0.3 |
| C-Hib | 182.4±17.4 | 5.1±0.8 | 19.1±11.5 | 2.6±0.4 |
| A-Ex | 211.7±11.3 | 4.2±0.6 | 22.4±9.7 | 2.7±0.3 |
| OT | 176.8±29.2 | 5.2±1.7 | 18.2±9.9 | 3.2±1.2 |
| OT-Hib
|
215.5±27.3
|
5.3±1.3 | 39.7±6.2
* vs. OT |
3.1±0.4 |
| Significance, p<0.05 | n.s. | n.s. | * significant differences | n.s. |
Abbreviations: C, control group; C-Hib, control-H.sabdariffa; A-Ex, mild aerobic exercise; OT, overtraining exercise; OT-Hib, overtraining exercise-H.sabdariffa; n.s, not significant.
IGFBP-3 gene expression (Table 2) is highest in the two overtraining groups, without and with Hib, higher than in control, C-Hib, and A-Ex groups. Obviously, overtraining up-regulates IGFBP-3 gene expression in terms of physical adaptation. However, increased IGFBP-3 gene expression does not match the low IGFBP-3 levels in blood (Table 2), although they were not as rapidly decreased as in our previous study8.
On the first glance, the results of this study seem to show that physical overtraining exercise does not interfere with the process of regulation of IGFBP-3 gene expression, because differences are not significant. However, the strong negative correlation between GPx activity and IGFBP-3 gene expression suggests interference with the regulation of the GH/IGF-1 axis. On the other hand, there is also a strong negative correlation between GPx activity and MDA levels suggesting that antioxidant Hibiscus extract interferes with the oxidative stress in overtraining syndrome as indicated by the highest levels of MDA (Fig. 1). This assumption is supported by the lower MDA levels of the over-trained groups with H. sabdariffa extract (OT-Hib) as compared to groups OT and A-Ex; IGFBP-3 gene expression is slightly (although not significantly) lower and circulating levels of IGFBP-3 are significantly higher than in the over-trained group (OT) and the highest level of all groups.
More research is needed to clarify the exact mechanism of the decrease of IGFBP 3 levels in overtraining syndrome which is associated with oxidative stress. High levels of IGFBP-3 in the OT-Hib group indicate the role of H. sabdariffa as an antioxidant in the counteraction towards overtraining syndrome. On the other hand, it must still be clarified whether (and/or how) H.safdariffa interferes with the GH/IGF-1 axis down to the cellular level to express IGFBP-3 protein.
Antioxidants can prevent or inhibit the oxidative damage and degradation of proteins, lipids (e.g., in cell membranes), and DNA/RNA. H. sabdariffa contains – among other substances – flavonoids, ascorbic acid, protocatechuic acid (PCA), and anthocyanin32. The latter was found to be the most potent antioxidant compound in H.sabdariffa extract raising total antioxidant capacity and especially increasing GPx activity33,34. This was the major reason why we investigated the GPx activity under our experimental conditions. In our study H.sabdariffa increased GPx activity in groups C-Hib versus control and attenuated it to normal control values in the OT group.
In conclusion, H. sabdariffa extract appears to prevent symptoms of overtraining syndrome in rats via antioxidant mechanisms, it decreases MDA levels, normalizes GPx activity, and strongly increases levels of circulating IGFBP-3. The strong negative correlation between GPx activity and IGFBP-3 gene expression needs further clarification with respect to the interference with the GH/IGF-1 axis down to the cellular level.
Acknowledgement
We express our thanks to DRPM UI for having funded this study through a grant from Universitas Indonesia 2013 (Hibah Riset Madya UI 2013).
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
References
- Smith L. Cytokine hypothesis of overtraining: a physiological adaptation to excessive stress? Medicine & Science in Sports & Exercise 2000; 32(2): 317.
CorssRef - Marcello, B.M. Overtraining in sport: physiological, psychological and performance effect of participation in division I competitive softball [doctoral dissertation]. Baylor University; (2006).
- Kenttä G, Hassmén P. Overtraining and recovery. Sports Medicine 1998; 26(1): 1-16.
CorssRef - Bischler T.K. The utility of resting levels of IGF-I and IGFBP-3 as markers of training status in elite athletes [master thesis]. University of Lethbridge (2005).
- Fry A, Schilling B, Weiss L, Chiu L. Beta2-Adrenergic receptor downregulation and performance decrements during high-intensity resistance exercise overtraining. Journal of Applied Physiology 2005; 101(6): 1664-1672.
CorssRef - Gatti R, De Palo F, Antonelli G, Spinella P. IGF-I/IGFBP system: metabolism outline and physical exercise. Journal of Endocrinological Investigation 2012; 35(7): 699-707.
- Elloumi M. El Elj N, Zaouali M, Maso F, Filaire E, Tabka Z, et al. IGFBP-3, a sensitive marker of physical training and overtraining. British Journal of Sports Medicine 2005; 39(9): 604-610.
CorssRef - Ilyas EII, Kartinah NT, Andriani T, Goenarjo RA, Kahanjak DN. Effects of Hibiscus safdariffa Linn. on insulin-like growth factor binding protein 3 (IGFBP-3) to prevent overtraining syndrome. Medical Journal of Indonesia 2014; 23(4): 187-191.
- Finaud J, Lac G, Filaire E. Oxidative stress. Sports Medicine 2006; 36(4): 327-358.
CorssRef - Capoluongo E, Pitocco D, Lulli P, Minucci A, Santonocito C, Manto A et al. Inverse correlation between serum free IGF-I and IGFBP-3 levels and blood pressure in patients affected with type 1 diabetes. Cytokine 2006; 34(5-6): 303-311.
CorssRef - Banerjee A, Mandal A, Chanda D, Chakraborti S. Oxidant, antioxidant and physical exercise. Molecular and Cellular Biochemistry 2003; 253(1): 307-312.
CorssRef - Kanter M. Free radicals, exercise and antioxidant supplementation. Proceedings of the Nutrition Society 1998; 57(01): 9-13.
CorssRef - Urso M, Clarkson P. Oxidative stress, exercise and antioxidant supplementation. Toxicology 2003; 189(1-2): 41-54.
CorssRef - Liu J. Chronically and acutely exercised rats: biomarkers of oxidative stress and endogenous antioxidants. Journal of Applied Physiology 2000; 89(1): 21-28.
- Ali B, Wabel N, Blunden G. Phytochemical, pharmacological and toxicological aspects of Hibiscus sabdariffa L.: a review. Phytotherapy Research 2005; 19(5): 369-375.
CorssRef - Kowalczyk E, Krzesiñski P, Kura M, Szmigiel B, Aszczyk JB. Anthocyanins in medicine. Journal of Pharmacology 2003; 55(5): 699-702.
- Hohl R, Ferraresso RL, De Oliveira RB, Lucco R, Brenzikofer R, De Macedo DV. Development and characterization of an overtraining animal model. Medicine & Science in Sports & Exercise 2009; 41(5): 1155-1163.
CorssRef - Wang S, Lee S, Wang C, Lee C, Lee W, Lee H. Aqueous extract from Hibiscus sabdariffa Linnaeus ameliorate diabetic nephropathy via regulating oxidative status and akt/bad/14-3-3γ in an experimental animal model. Evidence-Based Complementary and Alternative Medicine 2011; 2011: 1-9.
- Yagi K. Simple procedure for specific assay of lipid hydroperoxides in serum or plasma. Methods Mol Biol 1998; 108: 107-110.
CorssRef - Margonis K, Fatourus I, Jamurtas AZ, Nikolaidis MG, Douroudos I, Chatzinikolaou A, et al. Oxidative stress biomarkers responses to physical overtraining: Implications for diagnosis. Free Radical Biology and Medicine 2007; 43: 901-910.
CorssRef - Urhausen A, Kindermann W. Diagnosis of overtraining. Sports Medicine 2002; 32(2): 95-102.
CorssRef - Budgett R. Fatigue and underperformance in athletes: the overtraining syndrome. British Journal of Sports Medicine 1998; 32(2): 107-110.
CorssRef - Kreher JB, Schwartz JB. Overtraining syndrome practical guide. Sports Health 2012; 4: 128-138.
CorssRef - Meeusen R, Duclos M, Foster C, Fry A, Gleeson M, Nieman D, et al. Prevention, diagnosis and treatment of the overtraining syndrome: joint consensus statement of the European College of Sport Science (ECSS) and the American College of Sports Medicine (ACSM). European Journal of Sport Science 2013; 13(1): 1-24.
CorssRef - Hooper S, Mackinnon L, Howard A, Gordon R, Bachmann A. Markers for monitoring overtraining and recovery. Medicine & Science in Sports & Exercise 1995; 27(1): 106-112.
CorssRef - Fry R, Morton A, Keast D. Overtraining in athletes. Sports Medicine 1991; 12(1): 32-65.
CorssRef - Darmono S. Buku Ajar Farmakologi Eksperimental. Penerbit Universitas Indonesia, Jakarta (2001).
- Xiao W, Chen P, Dong J. Effects of overtraining on skeletal muscle growth and gene expression. International Journal of Sports Medicine 2012; 33(10): 846-853.
CorssRef - Nishida Y, Matsubara T, Tobina T, Shindo M, Tokuyama K, Tanaka K, et al. Effect of low-intensity aerobic exercise on insulin-like growth factor-I and insulin-like growth factor-binding proteins in healthy men. International Journal of Endocrinology 2010; 2010: 1-8.
CorssRef - Nindl B, Pierce J. Insulin-like growth factor I as a biomarker of health, fitness and training status. Medicine & Science in Sports & Exercise 2010; 42(1): 39-49.
CorssRef - Roelen C, de Vries W, Koppeschaar H, Vervoorn C, Thijssen J, Blankenstein M. Plasma insulin-like growth factor-I and high affinity growth hormone-binding protein levels increase after two weeks of strenuous physical training. International Journal of Sports Medicine 1997; 18(04): 238-241.
CorssRef - Ali BH, Wabel NA, Blunden G. Phytochemical, pharmacological and toxicological aspects of Hibiscus sabdariffa Linn: a review. Phytotherapy Research 2005; 19(5): 369 – 375.
CorssRef - Tseng TH, Kao ES, Chu CY, Chou FP, Lin Wu HW, Wang CJ. Protective effects of dried flower extracts of Hibiscus sabdariffa L. against oxidative stress in rat primary hepatocytes. Food and Chemical Toxicology 1997; 35(12); 1159-1164.
CorssRef - Wang CJ, Wang JM, Lin WL, Chu CY, Chou FP, Tseng TH. Protective effect of Hibiscus anthocyanins against tert-butyl hydroperoxide-induced hepatic toxicity in rats. Food and Chemical Toxicology 2000; 38(5): 411-416.
CorssRef







